|
Pauli Exclusion Principle
In quantum mechanics, the Pauli exclusion principle (German: Pauli-Ausschlussprinzip) states that two or more identical particles with half-integer spins (i.e. fermions) cannot simultaneously occupy the same quantum state within a system that obeys the laws of quantum mechanics. This principle was formulated by Austrian physicist Wolfgang Pauli in 1925 for electrons, and later extended to all fermions with his spin–statistics theorem of 1940. In the case of electrons in atoms, the exclusion principle can be stated as follows: in a poly-electron atom it is impossible for any two electrons to have the same two values of ''all'' four of their quantum numbers, which are: ''n'', the principal quantum number; ', the azimuthal quantum number; ''m'', the magnetic quantum number; and ''ms'', the spin quantum number. For example, if two electrons reside in the same orbital, then their values of ''n'', ', and ''m'' are equal. In that case, the two values of ''m''s (spin) pair must be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Wolfgang Pauli Young
Wolfgang is a German male given name traditionally popular in Germany, Austria and Switzerland. The name is a combination of the Old High German words ''wolf'', meaning "wolf", and ''gang'', meaning "path", "journey", "travel". Besides the regular "wolf", the first element also occurs in Old High German as the combining form "-olf". The earliest reference of the name being used was in the 8th century. The name was also attested as "Vulfgang" in the in the 9th century. The earliest recorded famous bearer of the name was a tenth-century Saint Wolfgang of Regensburg. Due to the lack of conflict with the pagan reference in the name with Catholicism, it is likely a much more ancient name whose meaning had already been lost by the tenth century. Grimm (''Teutonic Mythology'' p. 1093) interpreted the name as that of a hero in front of whom walks the "wolf of victory". A Latin gloss by Arnold of St Emmeram interprets the name as ''Lupambulus''.E. Förstemann, ''Altdeutsches Namenb ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
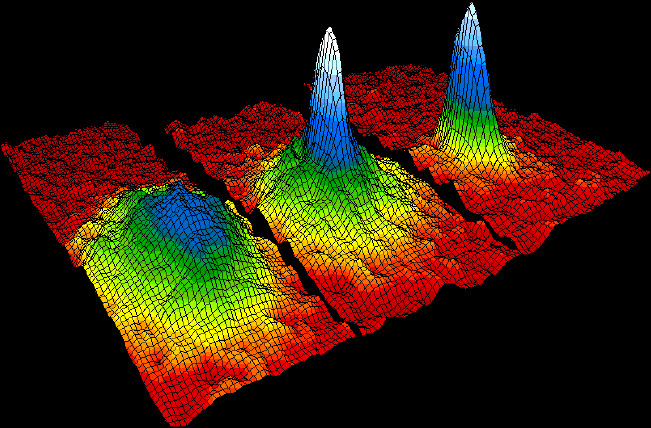
Bose–Einstein Condensate
In condensed matter physics, a Bose–Einstein condensate (BEC) is a state of matter that is typically formed when a gas of bosons at very low Density, densities is cooled to temperatures very close to absolute zero#Relation with Bose–Einstein condensate, absolute zero, i.e. . Under such conditions, a large fraction of bosons occupy the lowest quantum state, at which microscopic Quantum mechanics, quantum-mechanical phenomena, particularly wave interference#Quantum interference, wavefunction interference, become apparent Macroscopic quantum phenomena, macroscopically. More generally, condensation refers to the appearance of macroscopic occupation of one or several states: for example, in BCS theory, a superconductor is a condensate of Cooper pairs. As such, condensation can be associated with phase transition, and the macroscopic occupation of the state is the order parameter. Bose–Einstein condensate was first predicted, generally, in 1924–1925 by Albert Einstein, credit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Helium-4
Helium-4 () is a stable isotope of the element helium. It is by far the more abundant of the two naturally occurring isotopes of helium, making up about 99.99986% of the helium on Earth. Its nucleus is identical to an alpha particle, and consists of two protons and two neutrons. Helium-4 makes up about one quarter of the ordinary matter in the universe by mass, with almost all of the rest being hydrogen. While nuclear fusion in stars also produces helium-4, most of the helium-4 in the Sun and in the universe is thought to have been produced during the Big Bang, known as " primordial helium". However, primordial helium-4 is largely absent from the Earth, having escaped during the high-temperature phase of Earth's formation. On Earth, most naturally occurring helium-4 is produced by the alpha decay of heavy elements in the Earth's crust, after the planet cooled and solidified. When liquid helium-4 is cooled to below , it becomes a superfluid, with properties very different from ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Helium-3
Helium-3 (3He see also helion) is a light, stable isotope of helium with two protons and one neutron. (In contrast, the most common isotope, helium-4, has two protons and two neutrons.) Helium-3 and hydrogen-1 are the only stable nuclides with more protons than neutrons. It was discovered in 1939. Helium-3 atoms are fermionic and become a superfluid at the temperature of 2.491 mK. Helium-3 occurs as a primordial nuclide, escaping from Earth's crust into its atmosphere and into outer space over millions of years. It is also thought to be a natural nucleogenic and cosmogenic nuclide, one produced when lithium is bombarded by natural neutrons, which can be released by spontaneous fission and by nuclear reactions with cosmic rays. Some found in the terrestrial atmosphere is a remnant of atmospheric and underwater nuclear weapons testing. Nuclear fusion using helium-3 has long been viewed as a desirable future energy source. The fusion of two of its atoms would be aneut ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Atom
Atoms are the basic particles of the chemical elements. An atom consists of a atomic nucleus, nucleus of protons and generally neutrons, surrounded by an electromagnetically bound swarm of electrons. The chemical elements are distinguished from each other by the number of protons that are in their atoms. For example, any atom that contains 11 protons is sodium, and any atom that contains 29 protons is copper. Atoms with the same number of protons but a different number of neutrons are called isotopes of the same element. Atoms are extremely small, typically around 100 picometers across. A human hair is about a million carbon atoms wide. Atoms are smaller than the shortest wavelength of visible light, which means humans cannot see atoms with conventional microscopes. They are so small that accurately predicting their behavior using classical physics is not possible due to quantum mechanics, quantum effects. More than 99.94% of an atom's mass is in the nucleus. Protons hav ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Subatomic Particle
In physics, a subatomic particle is a particle smaller than an atom. According to the Standard Model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a baryon, like a proton or a neutron, composed of three quarks; or a meson, composed of two quarks), or an elementary particle, which is not composed of other particles (for example, quarks; or electrons, muons, and tau particles, which are called leptons). Particle physics and nuclear physics study these particles and how they interact. Most force-carrying particles like photons or gluons are called bosons and, although they have quanta of energy, do not have rest mass or discrete diameters (other than pure energy wavelength) and are unlike the former particles that have rest mass and cannot overlap or combine which are called fermions. The W and Z bosons, however, are an exception to this rule and have relatively large rest masses at approxim ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Neutron
The neutron is a subatomic particle, symbol or , that has no electric charge, and a mass slightly greater than that of a proton. The Discovery of the neutron, neutron was discovered by James Chadwick in 1932, leading to the discovery of nuclear fission in 1938, the first self-sustaining nuclear reactor (Chicago Pile-1, 1942) and the first nuclear weapon (Trinity (nuclear test), Trinity, 1945). Neutrons are found, together with a similar number of protons in the atomic nucleus, nuclei of atoms. Atoms of a chemical element that differ only in neutron number are called isotopes. Free neutrons are produced copiously in nuclear fission and nuclear fusion, fusion. They are a primary contributor to the nucleosynthesis of chemical elements within stars through fission, fusion, and neutron capture processes. Neutron stars, formed from massive collapsing stars, consist of neutrons at the density of atomic nuclei but a total mass more than the Sun. Neutron properties and interactions ar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Proton
A proton is a stable subatomic particle, symbol , Hydron (chemistry), H+, or 1H+ with a positive electric charge of +1 ''e'' (elementary charge). Its mass is slightly less than the mass of a neutron and approximately times the mass of an electron (the proton-to-electron mass ratio). Protons and neutrons, each with a mass of approximately one Dalton (unit), dalton, are jointly referred to as ''nucleons'' (particles present in atomic nuclei). One or more protons are present in the Atomic nucleus, nucleus of every atom. They provide the attractive electrostatic central force which binds the atomic electrons. The number of protons in the nucleus is the defining property of an element, and is referred to as the atomic number (represented by the symbol ''Z''). Since each chemical element, element is identified by the number of protons in its nucleus, each element has its own atomic number, which determines the number of atomic electrons and consequently the chemical characteristi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Baryon
In particle physics, a baryon is a type of composite particle, composite subatomic particle that contains an odd number of valence quarks, conventionally three. proton, Protons and neutron, neutrons are examples of baryons; because baryons are composed of quarks, they belong to the hadron list of particles, family of particles. Baryons are also classified as fermions because they have half-integer Spin (physics), spin. The name "baryon", introduced by Abraham Pais, comes from the Ancient Greek, Greek word for "heavy" (βαρύς, ''barýs''), because, at the time of their naming, most known elementary particles had lower masses than the baryons. Each baryon has a corresponding antiparticle (antibaryon) where their corresponding antiquarks replace quarks. For example, a proton is made of two up quarks and one down quark; and its corresponding antiparticle, the antiproton, is made of two up antiquarks and one down antiquark. Baryons participate in the residual strong force, which ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Neutrino
A neutrino ( ; denoted by the Greek letter ) is an elementary particle that interacts via the weak interaction and gravity. The neutrino is so named because it is electrically neutral and because its rest mass is so small ('' -ino'') that it was long thought to be zero. The rest mass of the neutrino is much smaller than that of the other known elementary particles (excluding massless particles). The weak force has a very short range, the gravitational interaction is extremely weak due to the very small mass of the neutrino, and neutrinos do not participate in the electromagnetic interaction or the strong interaction. Consequently, neutrinos typically pass through normal matter unimpeded and with no detectable effect. Weak interactions create neutrinos in one of three leptonic flavors: # electron neutrino, # muon neutrino, # tau neutrino, Each flavor is associated with the correspondingly named charged lepton. Although neutrinos were long believed to be mas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Quark
A quark () is a type of elementary particle and a fundamental constituent of matter. Quarks combine to form composite particles called hadrons, the most stable of which are protons and neutrons, the components of atomic nucleus, atomic nuclei. All commonly observable matter is composed of up quarks, down quarks and electrons. Owing to a phenomenon known as ''color confinement'', quarks are never found in isolation; they can be found only within hadrons, which include baryons (such as protons and neutrons) and mesons, or in quark–gluon plasmas. There is also the theoretical possibility of #Other_phases_of_quark_matter, more exotic phases of quark matter. For this reason, much of what is known about quarks has been drawn from observations of hadrons. Quarks have various Intrinsic and extrinsic properties, intrinsic physical property, properties, including electric charge, mass, color charge, and Spin (physics), spin. They are the only elementary particles in the Standard Mode ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Elementary Particle
In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles. The Standard Model presently recognizes seventeen distinct particles—twelve fermions and five bosons. As a consequence of flavor and color combinations and antimatter, the fermions and bosons are known to have 48 and 13 variations, respectively. Among the 61 elementary particles embraced by the Standard Model number: electrons and other leptons, quarks, and the fundamental bosons. Subatomic particles such as protons or neutrons, which contain two or more elementary particles, are known as composite particles. Ordinary matter is composed of atoms, themselves once thought to be indivisible elementary particles. The name ''atom'' comes from the Ancient Greek word ''ἄτομος'' ( atomos) which means ''indivisible'' or ''uncuttable''. Despite the theories about atoms that had existed for thousands of years, the factual existence of ato ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |