|
Pregnanolone
Pregnanolone, also known as eltanolone, is an endogenous inhibitory neurosteroid which is produced in the body from progesterone. It is closely related to allopregnanolone, which has similar properties. Biological activity Pregnanolone is a positive allosteric modulator of the GABAA receptor, as well as a negative allosteric modulator of the glycine receptor. Biological function Pregnanolone has sedative, anxiolytic, anesthetic, and anticonvulsant effects. During pregnancy, pregnanolone and allopregnanolone are involved in sedation and anesthesia of the fetus. Biochemistry Pregnanolone is synthesized from progesterone via the enzymes 5β-reductase and 3α-hydroxysteroid dehydrogenase, with 5β-dihydroprogesterone occurring as a metabolic intermediate. The elimination half-life of pregnanolone is between 0.9 and 3.5 hours. Chemistry Pregnanolone, also known as 3α,5β-tetrahydroprogesterone (3α,5β-THP) or as 5β-pregnan-3α-ol-20-one, is a naturally occurring pregn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allopregnanolone
Allopregnanolone is a natural product, naturally occurring neurosteroid which is made in the body from the hormone progesterone. As a medication, allopregnanolone is referred to as brexanolone, sold under the brand name Zulresso, and used to treat postpartum depression. It is given by intravenous, injection into a vein. Side effects of brexanolone may include sedation, sleepiness, dry mouth, hot flashes, and loss of consciousness. It is a neurosteroid and acts as a positive allosteric modulator of the GABAA receptor, GABAA receptor, the major biological target of the inhibitory postsynaptic potential, inhibitory neurotransmitter γ-aminobutyric acid (GABA). Brexanolone was approved for medical use in the United States in 2019. The U.S. Food and Drug Administration (FDA) considers it to be a first-in-class medication. The long administration time, as well as the cost for a one-time treatment, have raised concerns about accessibility for many women. Medical uses Brexanolon ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isopregnanolone
Isopregnanolone, also known as isoallopregnanolone and epiallopregnanolone, as well as sepranolone (), and as 3β-hydroxy-5α-pregnan-20-one or 3β,5α-tetrahydroprogesterone (3β,5α-THP), is an endogenous neurosteroid and a natural 3β-epimer of allopregnanolone.Öfverman, C., Strömberg, J., Birzniece, V., Turkmen, S., Hill, M., Lundgren, P., ... & Johansson, I. M. (2009). The progesterone metabolite isoallopregnanolone is a subunit-selective antagonist of the GABA-A receptor. Chicago It has been reported to act as a subunit-selective negative allosteric modulator of the GABAA receptor, and antagonizes in animals and humans some but not all of the GABAA receptor-mediated effects of allopregnanolone, such as anesthesia, sedation, and reduced saccadic eye movements, but not learning impairment. Isopregnanolone has no hormonal effects and appears to have no effect on the GABAA receptor by itself; it selectively antagonizes allopregnanolone and does not affect the effects of othe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neurosteroid
Neurosteroids, also known as neuroactive steroids, are endogenous or exogenous steroids that rapidly alter neuronal excitability through interaction with ligand-gated ion channels and other cell surface receptors. The term ''neurosteroid'' was coined by the French physiologist Étienne-Émile Baulieu and refers to steroids synthesized in the brain. The term, ''neuroactive steroid'' refers to steroids that can be synthesized in the brain, or are synthesized by an endocrine gland, that then reach the brain through the bloodstream and have effects on brain function. The term neuroactive steroids was first coined in 1992 by Steven Paul and Robert Purdy. In addition to their actions on neuronal membrane receptors, some of these steroids may also exert effects on gene expression via nuclear steroid hormone receptors. Neurosteroids have a wide range of potential clinical applications from sedation to treatment of epilepsy and traumatic brain injury. Ganaxolone, a synthetic analog of the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epipregnanolone
Epipregnanolone, also known as 3β-hydroxy-5β-pregnan-20-one, 3β,5β-tetrahydroprogesterone, or 3β,5β-THP, is an endogenous neurosteroid. It acts as a negative allosteric modulator of the GABAA receptor and reverses the effects of potentiators like allopregnanolone. Epipregnanolone is biosynthesized from progesterone by the actions of 5β-reductase and 3β-hydroxysteroid dehydrogenase, with 5β-dihydroprogesterone as the intermediate in this two-step transformation. Epipregnanolone is not a progestogen itself, but via metabolization into other steroids, behaves indirectly as one. The sulfate of epipreganolone, epipregnanolone sulfate, is a negative allosteric modulator of the NMDA and GABAA receptors and also acts as a TRPM3 channel activator. Chemistry See also * Isopregnanolone * 3β-Dihydroprogesterone * Pregnanolone Pregnanolone, also known as eltanolone, is an endogenous inhibitory neurosteroid which is produced in the body from progesterone. It is closel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Progesterone
Progesterone (; P4) is an endogenous steroid and progestogen sex hormone involved in the menstrual cycle, pregnancy, and embryogenesis of humans and other species. It belongs to a group of steroid hormones called the progestogens and is the major progestogen in the body. Progesterone has a variety of important functions in the body. It is also a crucial metabolic intermediate in the production of other endogenous steroids, including the sex hormones and the corticosteroids, and plays an important role in brain function as a neurosteroid. In addition to its role as a natural hormone, progesterone is also used as a medication, such as in combination with estrogen for contraception, to reduce the risk of Uterine cancer, uterine or cervical cancer, in hormone replacement therapy, and in feminizing hormone therapy. It was first prescribed in 1934. Biological activity Progesterone is the most important progestogen in the body. As a potent agonist of the progesterone receptor, nu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Renanolone
Renanolone (International Nonproprietary Name, INN), or 11-ketopregnanolone, also known as 5β-pregnan-3α-ol-11,20-dione, is a synthetic compound, synthetic neuroactive steroid which is described as a general anesthetic but was never introduced for clinical use. Its isomers, alfaxolone and alfadolone, are also general anesthetics, and are known to act as positive allosteric modulators of the GABAA receptor, GABAA receptor, a property which is likely the case for renanolone as well. Chemistry See also * Alfadolone * Alfaxolone * Ganaxolone * Hydroxydione * Minaxolone * Pregnanolone References 5β-Pregnanes General anesthetics Neurosteroids Secondary alcohols Diketones GABAA receptor positive allosteric modulators {{Nervous-system-drug-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxydione
Hydroxydione, as hydroxydione sodium succinate (, , ) (brand names Viadril, Predion, and Presuren), also known as 21-Hydroxy-5β-pregnane-3,20-dione, is a neuroactive steroid which was formerly used as a general anesthetic, but was discontinued due to incidence of thrombophlebitis in patients. It was introduced in 1957, and was the first neuroactive steroid general anesthetic to be introduced for clinical use, an event which was shortly preceded by the observation in 1954 of the sedative properties of progesterone in mice. Chemistry Related compounds include alfadolone, alfaxolone, dihydrodeoxycorticosterone, ganaxolone, minaxolone, pregnanolone, and renanolone. References {{GABAA receptor positive modulators 5β-Pregnanes Primary alcohols Diketones GABAA receptor positive allosteric modulators General anesthetics Neurosteroids ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
GABAA Receptor
The GABAA receptor (GABAAR) is an ionotropic receptor and ligand-gated ion channel. Its endogenous Ligand (biochemistry), ligand is γ-aminobutyric acid (GABA), the major inhibitory neurotransmitter in the central nervous system. Accurate regulation of GABAergic transmission through appropriate developmental processes, specificity to neural cell types, and responsiveness to activity is crucial for the proper functioning of nearly all aspects of the central nervous system (CNS). Upon opening, the GABAA receptor on the Chemical synapse, postsynaptic cell is selectively permeable to Chloride, chloride ions () and, to a lesser extent, Bicarbonate, bicarbonate ions (). GABAAR are members of the ligand-gated ion channel receptor superfamily, which is a chloride channel family with a dozen or more heterotetrametric subtypes and 19 distinct subunits. These subtypes have distinct brain regional and subcellular localization, age-dependent expression, and the ability to undergo plastic alt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sedative
A sedative or tranquilliser is a substance that induces sedation by reducing irritability or Psychomotor agitation, excitement. They are central nervous system (CNS) Depressant, depressants and interact with brain activity, causing its deceleration. Various kinds of sedatives can be distinguished, but the majority of them affect the neurotransmitter Gamma-Aminobutyric acid, gamma-aminobutyric acid (GABA). Most sedatives produce relaxing effects by increasing GABA activity. This group is related to hypnotics. The term ''sedative'' describes drugs that serve to calm or Anxiolytic, relieve anxiety, whereas the term ''hypnotic'' describes drugs whose main purpose is to initiate, sustain, or lengthen sleep. Because these two functions frequently overlap, and because drugs in this class generally produce dose-dependent effects (ranging from anxiolysis to loss of consciousness), they are often referred to collectively as ''sedative–hypnotic'' drugs. Terminology There is some overlap ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Steroid
A steroid is an organic compound with four fused compound, fused rings (designated A, B, C, and D) arranged in a specific molecular configuration. Steroids have two principal biological functions: as important components of cell membranes that alter membrane fluidity; and as signal transduction, signaling molecules. Examples include the lipid cholesterol, sex hormones estradiol and testosterone, anabolic steroids, and the anti-inflammatory corticosteroid drug dexamethasone. Hundreds of steroids are found in Fungus, fungi, plants, and animals. All steroids are manufactured in cells from a sterols, sterol: Cholesterol, cholesterol (animals), lanosterol (opisthokonts), or cycloartenol (plants). All three of these molecules are produced via Cyclic compound, cyclization of the triterpene squalene. Structure The steroid nucleus (parent structure, core structure) is called gonane (cyclopentanoperhydrophenanthrene). It is typically composed of seventeen carbon atoms, bonded in fou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
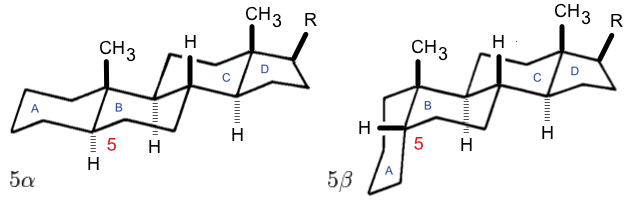
Pregnane
Pregnane, also known as 17β-ethylandrostane or as 10β,13β-dimethyl-17β-ethylgonane, is a C21 steroid and, indirectly, a parent of progesterone. It is a parent hydrocarbon for two series of steroids stemming from 5α-pregnane (originally allopregnane) and 5β-pregnane (17β-ethyl etiocholane). It has a gonane core. 5β-Pregnane is the parent of pregnanediones, pregnanolones, and pregnanediols, and is found largely in urine as a metabolic product of 5β-pregnane compounds. Pregnanes Pregnanes are steroid derivatives with carbons present at positions 1 through 21. Most biologically significant pregnane derivatives fall into one of two groups: pregnenes and pregnadienes. Another class is pregnatrienes. Pregnenes Pregnenes have a double bond. Examples include: * Cortisone * Hydrocortisone * Progesterone Pregnadienes Pregnadienes have two double bonds. Examples include: * Cyproterone acetate * Danazol Danazol, sold as Danocrine and other brand names, is a me ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metabolic Intermediate
Metabolic intermediates are compounds produced during the conversion of substrates (starting molecules) into final products in biochemical reactions within cells. Although these intermediates are of relatively minor direct importance to cellular function, they can play important roles in the allosteric regulation of enzymes, glycolysis, the citric acid cycle, and amino acid synthesis. Metabolic pathways consist of a series of enzymatically catalyzed reactions where each step transforms a substrate into a product that serves as the substrate for the next reaction. Metabolic intermediates are compounds that form during these steps, and they are neither the starting substrate nor the final product of the pathway. These intermediates are crucial because they allow for regulation, energy storage, and extraction of chemical energy in a controlled manner. Types of Metabolic Intermediates Metabolic intermediates can belong to different biochemical classes based on the type o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |