|
Milligram Per Cent
Milligram per cent is a traditional symbol used to denote a unit of measure of concentration. The traditional use of the 'mg%' symbol was meant to indicate the mass (in milligrams) of that chemical in 100 milliliters of solution (e.g., blood). The meaning of the symbol 'percent' is 'divided by 100', therefore the accurate meaning of the notation 'mg%' is 'milligrams divided by 100', which is a unit of mass, not a concentration. Therefore, for dimensional analysis purposes, when denoting a concentration of mass divided by volume, it has largely been replaced by units such as 'mg/dL' or the International System of Units equivalent. For example, a plasma ethanol concentration incorrectly denoted as 0.1 mg% can be written as '0.1 mg/dL' (or '1 mg/L', etc.) meaning a mass of 0.1 milligrams of ethyl alcohol per 100 milliliter volume Volume is a measure of occupied three-dimensional space. It is often quantified numerically using SI derived units (such as the cubi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Concentration
In chemistry, concentration is the abundance of a constituent divided by the total volume of a mixture. Several types of mathematical description can be distinguished: '' mass concentration'', ''molar concentration'', '' number concentration'', and '' volume concentration''. The concentration can refer to any kind of chemical mixture, but most frequently refers to solutes and solvents in solutions. The molar (amount) concentration has variants, such as normal concentration and osmotic concentration. Etymology The term concentration comes from the word concentrate, from the French , from con– + center, meaning “to put at the center”. Qualitative description Often in informal, non-technical language, concentration is described in a qualitative way, through the use of adjectives such as "dilute" for solutions of relatively low concentration and "concentrated" for solutions of relatively high concentration. To concentrate a solution, one must add more solute (for exam ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Milligrams
The kilogram (also kilogramme) is the unit of mass in the International System of Units (SI), having the unit symbol kg. It is a widely used measure in science, engineering and commerce worldwide, and is often simply called a kilo colloquially. It means 'one thousand grams'. The kilogram is defined in terms of the second and the metre, both of which are based on fundamental physical constants. This allows a properly equipped metrology laboratory to calibrate a mass measurement instrument such as a Kibble balance as the primary standard to determine an exact kilogram mass. The kilogram was originally defined in 1795 as the mass of one litre of water. The current definition of a kilogram agrees with this original definition to within 30 parts per million. In 1799, the platinum '' Kilogramme des Archives'' replaced it as the standard of mass. In 1889, a cylinder of platinum-iridium, the International Prototype of the Kilogram (IPK), became the standard of the unit of mass for th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Litre
The litre (international spelling) or liter (American English spelling) (SI symbols L and l, other symbol used: ℓ) is a metric unit of volume. It is equal to 1 cubic decimetre (dm3), 1000 cubic centimetres (cm3) or 0.001 cubic metre (m3). A cubic decimetre (or litre) occupies a volume of (see figure) and is thus equal to one-thousandth of a cubic metre. The original French metric system used the litre as a base unit. The word ''litre'' is derived from an older French unit, the '' litron'', whose name came from Byzantine Greek—where it was a unit of weight, not volume—via Late Medieval Latin, and which equalled approximately 0.831 litres. The litre was also used in several subsequent versions of the metric system and is accepted for use with the SI,Bureau International des Poids et ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mass
Mass is an intrinsic property of a body. It was traditionally believed to be related to the quantity of matter in a physical body, until the discovery of the atom and particle physics. It was found that different atoms and different elementary particles, theoretically with the same amount of matter, have nonetheless different masses. Mass in modern physics has multiple definitions which are conceptually distinct, but physically equivalent. Mass can be experimentally defined as a measure of the body's inertia, meaning the resistance to acceleration (change of velocity) when a net force is applied. The object's mass also determines the strength of its gravitational attraction to other bodies. The SI base unit of mass is the kilogram (kg). In physics, mass is not the same as weight, even though mass is often determined by measuring the object's weight using a spring scale, rather than balance scale comparing it directly with known masses. An object on the Moon woul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimensional Analysis
In engineering and science, dimensional analysis is the analysis of the relationships between different physical quantities by identifying their base quantities (such as length, mass, time, and electric current) and units of measure (such as miles vs. kilometres, or pounds vs. kilograms) and tracking these dimensions as calculations or comparisons are performed. The conversion of units from one dimensional unit to another is often easier within the metric or the SI than in others, due to the regular 10-base in all units. ''Commensurable'' physical quantities are of the same kind and have the same dimension, and can be directly compared to each other, even if they are expressed in differing units of measure, e.g. yards and metres, pounds (mass) and kilograms, seconds and years. ''Incommensurable'' physical quantities are of different kinds and have different dimensions, and can not be directly compared to each other, no matter what units they are expressed in, e.g. metres and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Volume
Volume is a measure of occupied three-dimensional space. It is often quantified numerically using SI derived units (such as the cubic metre and litre) or by various imperial or US customary units (such as the gallon, quart, cubic inch). The definition of length (cubed) is interrelated with volume. The volume of a container is generally understood to be the capacity of the container; i.e., the amount of fluid (gas or liquid) that the container could hold, rather than the amount of space the container itself displaces. In ancient times, volume is measured using similar-shaped natural containers and later on, standardized containers. Some simple three-dimensional shapes can have its volume easily calculated using arithmetic formulas. Volumes of more complicated shapes can be calculated with integral calculus if a formula exists for the shape's boundary. Zero-, one- and two-dimensional objects have no volume; in fourth and higher dimensions, an analogous concept to the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
International System Of Units
The International System of Units, known by the international abbreviation SI in all languages and sometimes pleonastically as the SI system, is the modern form of the metric system and the world's most widely used system of measurement. Established and maintained by the General Conference on Weights and Measures (CGPM), it is the only system of measurement with an official status in nearly every country in the world, employed in science, technology, industry, and everyday commerce. The SI comprises a coherent system of units of measurement starting with seven base units, which are the second (symbol s, the unit of time), metre (m, length), kilogram (kg, mass), ampere (A, electric current), kelvin (K, thermodynamic temperature), mole (mol, amount of substance), and candela (cd, luminous intensity). The system can accommodate coherent units for an unlimited number of additional quantities. These are called coherent derived units, which can always be represented ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Blood Alcohol Content
Blood alcohol content (BAC), also called blood alcohol concentration or blood alcohol level, is a measurement of alcohol intoxication used for legal or medical purposes; it is expressed as mass of alcohol per volume or mass of blood. For example, a BAC of 0.10 by (0.10% or one tenth of one percent) means that there is 0.10 g of alcohol for every 100 mL of blood, which is the same as 21.7 mmol/L. A BAC of 0.10 by (0.10%) is 0.10 g of alcohol per 100 g of blood (23 mmol/L). A BAC of 0.0 is sober; in different countries the maximum permitted BAC when driving ranges from about 0.04% to 0.08%; BAC levels over 0.08% are considered very impaired; above 0.4% is potentially fatal. Effects by alcohol level Estimation by intake Blood alcohol content can be estimated by a method developed by Swedish professor in the 1920s: :EBAC = \frac\times100\%-\beta\times T where: * is the mass of alcohol consumed. * is the ratio of body water to total weight. It v ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethyl Alcohol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a hydroxyl group). Ethanol is a volatile, flammable, colorless liquid with a characteristic wine-like odor and pungent taste. It is a psychoactive recreational drug, the active ingredient in alcoholic drinks. Ethanol is naturally produced by the fermentation process of sugars by yeasts or via petrochemical processes such as ethylene hydration. It has medical applications as an antiseptic and disinfectant. It is used as a chemical solvent and in the synthesis of organic compounds, and as a fuel source. Ethanol also can be dehydrated to make ethylene, an important chemical feedstock. As of 2006, world production of ethanol was , coming mostly from Brazil and the U.S. Etymology ''Ethanol'' is the systematic name defined by the International Un ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Parts-per-million
In science and engineering, the parts-per notation is a set of pseudo-units to describe small values of miscellaneous dimensionless quantities, e.g. mole fraction or mass fraction. Since these fractions are quantity-per-quantity measures, they are pure numbers with no associated units of measurement. Commonly used are parts-per-million (ppm, ), parts-per-billion (ppb, ), parts-per-trillion (ppt, ) and parts-per-quadrillion (ppq, ). This notation is not part of the International System of Units (SI) system and its meaning is ambiguous. Overview Parts-per notation is often used describing dilute solutions in chemistry, for instance, the relative abundance of dissolved minerals or pollutants in water. The quantity "1 ppm" can be used for a mass fraction if a water-borne pollutant is present at one-millionth of a gram per gram of sample solution. When working with aqueous solutions, it is common to assume that the density of water is 1.00 g/mL. Therefore, it is common to equ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Properties
A chemical property is any of a material's properties that becomes evident during, or after, a chemical reaction; that is, any quality that can be established only by changing a substance's chemical identity.William L. Masterton, Cecile N. Hurley, "Chemistry: Principles and Reactions", 6th edition. Brooks/Cole Cengage Learning, 2009, p.1(Google books)/ref> Simply speaking, chemical properties cannot be determined just by viewing or touching the substance; the substance's internal structure must be affected greatly for its chemical properties to be investigated. When a substance goes under a chemical reaction, the properties will change drastically, resulting in chemical change. However, a catalytic property would also be a chemical property. Chemical properties can be contrasted with physical properties, which can be discerned without changing the substance's structure. However, for many properties within the scope of physical chemistry, and other disciplines at the boundary betwe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Physiology
Physiology (; ) is the scientific study of functions and mechanisms in a living system. As a sub-discipline of biology, physiology focuses on how organisms, organ systems, individual organs, cells, and biomolecules carry out the chemical and physical functions in a living system. According to the classes of organisms, the field can be divided into medical physiology, animal physiology, plant physiology, cell physiology, and comparative physiology. Central to physiological functioning are biophysical and biochemical processes, homeostatic control mechanisms, and communication between cells. ''Physiological state'' is the condition of normal function. In contrast, ''pathological state'' refers to abnormal conditions, including human diseases. The Nobel Prize in Physiology or Medicine is awarded by the Royal Swedish Academy of Sciences for exceptional scientific achievements in physiology related to the field of medicine. Foundations Cells Although there are di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |



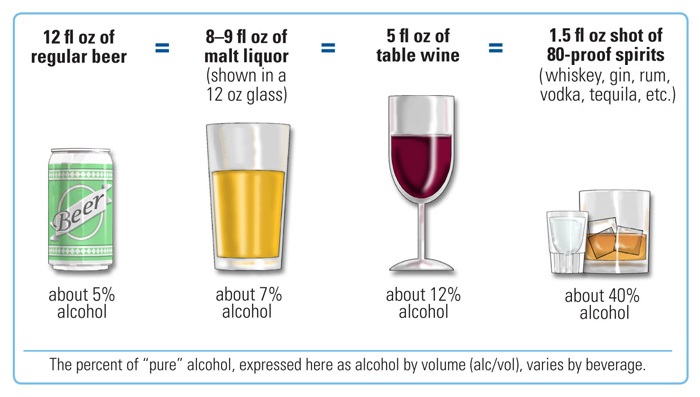

.jpg)