|
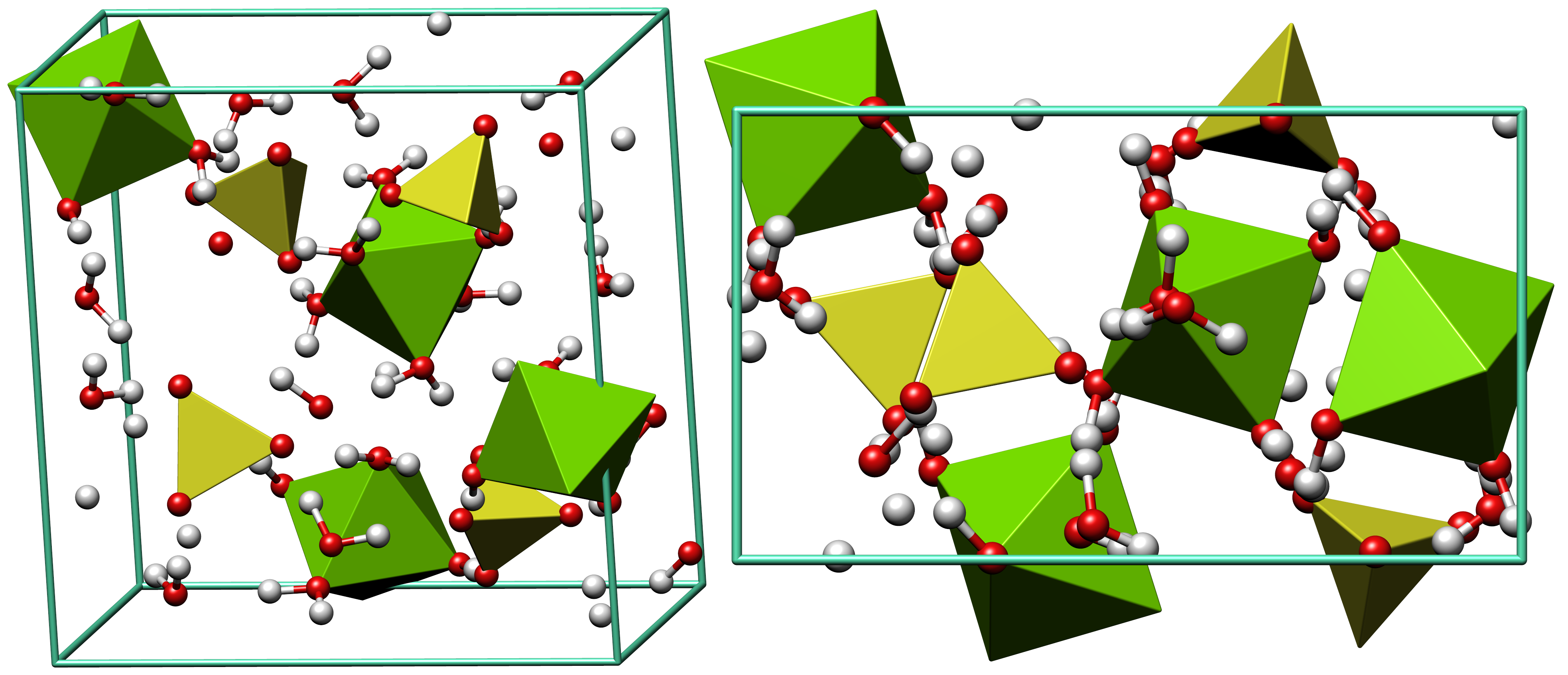
Melanterite
Melanterite is a mineral form of hydrous iron(II) sulfate: FeSO4·7H2O. It is the iron analogue of the copper sulfate chalcanthite. It alters to siderotil by loss of water. It is a secondary sulfate mineral which forms from the oxidation of primary sulfide minerals such as pyrite and marcasite in the near-surface environment. It often occurs as a ''post mine'' encrustation on old underground mine surfaces. It also occurs in coal and lignite seams exposed to humid air and as a rare sublimate phase around volcanic fumaroles. Associated minerals include pisanite, chalcanthite, epsomite, pickeringite, halotrichite Halotrichite, also known as ''feather alum'', is a highly hydrated sulfate of aluminium and iron. Its chemical formula is FeAl2(SO4)4·22H2O. It forms fibrous monoclinic crystals. The crystals are water-soluble. It is formed by the weathering and ... and other sulfate minerals. It was first described in 1850. Gallery File:Melanterite crystal structure.png, Crys ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron(II) Sulfate
Iron(II) sulfate (British English: iron(II) sulphate) or ferrous sulfate denotes a range of salts with the formula Fe SO4·''x''H2O. These compounds exist most commonly as the heptahydrate (''x'' = 7) but several values for x are known. The hydrated form is used medically to treat iron deficiency, and also for industrial applications. Known since ancient times as copperas and as green vitriol (vitriol is an archaic name for sulfate), the blue-green heptahydrate (hydrate with 7 molecules of water) is the most common form of this material. All the iron(II) sulfates dissolve in water to give the same aquo complex e(H2O)6sup>2+, which has octahedral molecular geometry and is paramagnetic. The name copperas dates from times when the copper(II) sulfate was known as blue copperas, and perhaps in analogy, iron(II) and zinc sulfate were known respectively as green and white copperas. It is on the World Health Organization's List of Essential Medicines. In 2020, it was the 1 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfate Mineral
The sulfate minerals are a class of minerals that include the sulfate ion () within their structure. The sulfate minerals occur commonly in primary evaporite depositional environments, as gangue minerals in hydrothermal veins and as secondary minerals in the oxidizing zone of sulfide mineral deposits. The chromate and manganate minerals have a similar structure and are often included with the sulfates in mineral classification systems.Klein, Cornelis and Cornelius S. Hurlbut, 1985, ''Manual of Mineralogy,'' 20th ed., John Wiley and Sons, New York, pp. 347–354 . Sulfate minerals include: *Anhydrous sulfates **Barite BaSO4 **Celestite SrSO4 **Anglesite PbSO4 ** Anhydrite CaSO4 **Hanksite Na22K(SO4)9(CO3)2Cl *Hydroxide and hydrous sulfates **Gypsum CaSO4·2H2O **Chalcanthite CuSO4·5H2O ** Kieserite MgSO4·H2O ** Starkeyite MgSO4·4H2O ** Hexahydrite MgSO4·6H2O ** Epsomite MgSO4·7H2O ** Meridianiite MgSO4·11H2O **Melanterite FeSO4·7H2O ** Antlerite Cu3SO4(OH)4 **Brochantit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chalcanthite
Chalcanthite (, ) is a richly colored blue-green water-soluble sulfate mineral . It is commonly found in the late-stage oxidation zones of copper deposits. Due to its ready solubility, chalcanthite is more common in arid regions. Chalcanthite is a pentahydrate and the most common member of a group of similar hydrated sulfates, the chalcanthite group. These other sulfates are identical in chemical composition to chalcanthite, with the exception of replacement of the copper ion by either manganese as jokokuite, iron as melanterite, or magnesium as pentahydrite. Other names include ''blue stone'', ''blue vitriol'', and ''copper vitriol''. Uses of chalcanthite As chalcanthite is a copper mineral, it can be used as an ore of copper. However, its ready solubility in water means that it tends to crystallize, dissolve, and recrystallize as crusts over any mine surface in more humid regions. Therefore, chalcanthite is only found in the most arid regions in sufficiently large ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Siderotil
Siderotil is an iron(II) sulfate hydrate mineral with formula: FeSO4·5H2O which forms by the dehydration of melanterite. Copper commonly occurs substituting for iron in the structure. It typically occurs as fibrous or powdery encrustations, but may also occur as acicular triclinic crystals. It was first described in 1891 for an occurrence in the Idrija Mine, Idrija, Slovenia Slovenia ( ; sl, Slovenija ), officially the Republic of Slovenia (Slovene: , abbr.: ''RS''), is a country in Central Europe. It is bordered by Italy to the west, Austria to the north, Hungary to the northeast, Croatia to the southeast, an .... Its name derives from the Greek ''sideros'' (iron) and ''tilos'' (fiber) in reference to its iron content and typical fibrous form. However, the material at the Idrija location may not be siderotil, but the mineral has been authenticated from a wide variety of worldwide locations. References Sulfate minerals Iron(II) minerals Triclinic minerals Minerals ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfate Minerals
The sulfate minerals are a class of minerals that include the sulfate ion () within their structure. The sulfate minerals occur commonly in primary evaporite depositional environments, as gangue minerals in hydrothermal veins and as secondary minerals in the oxidizing zone of sulfide mineral deposits. The chromate and manganate minerals have a similar structure and are often included with the sulfates in mineral classification systems.Klein, Cornelis and Cornelius S. Hurlbut, 1985, ''Manual of Mineralogy,'' 20th ed., John Wiley and Sons, New York, pp. 347–354 . Sulfate minerals include: *Anhydrous sulfates **Barite BaSO4 **Celestite SrSO4 ** Anglesite PbSO4 ** Anhydrite CaSO4 ** Hanksite Na22K(SO4)9(CO3)2Cl *Hydroxide and hydrous sulfates **Gypsum CaSO4·2H2O **Chalcanthite CuSO4·5H2O ** Kieserite MgSO4·H2O ** Starkeyite MgSO4·4H2O ** Hexahydrite MgSO4·6H2O ** Epsomite MgSO4·7H2O ** Meridianiite MgSO4·11H2O **Melanterite FeSO4·7H2O ** Antlerite Cu3SO4(OH)4 **Brochant ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfate Mineral
The sulfate minerals are a class of minerals that include the sulfate ion () within their structure. The sulfate minerals occur commonly in primary evaporite depositional environments, as gangue minerals in hydrothermal veins and as secondary minerals in the oxidizing zone of sulfide mineral deposits. The chromate and manganate minerals have a similar structure and are often included with the sulfates in mineral classification systems.Klein, Cornelis and Cornelius S. Hurlbut, 1985, ''Manual of Mineralogy,'' 20th ed., John Wiley and Sons, New York, pp. 347–354 . Sulfate minerals include: *Anhydrous sulfates **Barite BaSO4 **Celestite SrSO4 **Anglesite PbSO4 ** Anhydrite CaSO4 **Hanksite Na22K(SO4)9(CO3)2Cl *Hydroxide and hydrous sulfates **Gypsum CaSO4·2H2O **Chalcanthite CuSO4·5H2O ** Kieserite MgSO4·H2O ** Starkeyite MgSO4·4H2O ** Hexahydrite MgSO4·6H2O ** Epsomite MgSO4·7H2O ** Meridianiite MgSO4·11H2O **Melanterite FeSO4·7H2O ** Antlerite Cu3SO4(OH)4 **Brochantit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pickeringite
Pickeringite is a magnesium aluminium sulfate mineral with formula MgAl2(SO4)4·22(H2O). It forms a series with halotrichite. It forms as an alteration product of pyrite in aluminium rich rocks and in coal seams. It also occurs in pyrite rich hydrothermal ore deposits in arid regions. It forms in fumaroles and in caves. It occurs with kalinite, alunogen, epsomite, melanterite, copiapite and gypsum. It was first described in 1844 for an occurrence in Cerros Pintados, Pampa del Tamarugal, Iquique Province, Tarapacá Region, Chile Chile, officially the Republic of Chile, is a country in the western part of South America. It is the southernmost country in the world, and the closest to Antarctica, occupying a long and narrow strip of land between the Andes to the eas .... It was named for American linguist and philologist John Pickering (1777–1846). References Sulfate minerals Monoclinic minerals Minerals in space group 14 {{sulfate-mineral-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epsomite
Epsomite, Epsom salt, or magnesium sulfate heptahydrate, is a hydrous magnesium sulfate mineral with formula MgSO4·7H2O. Epsomite crystallizes in the orthorhombic system as rarely found acicular or fibrous crystals, the normal form is as massive encrustations. It is colorless to white with tints of yellow, green and pink. The Mohs hardness is 2 to 2.5 and it has a low specific gravity of 1.67. It is readily soluble in water. It absorbs water from the air and converts to hexahydrate with the loss of one water molecule and a switch to monoclinic structure. Etymology It was first systematically described in 1806 for an occurrence near Epsom, Surrey, England, after which it was named. Discovery and occurrence Epsomite forms as encrustations or efflorescences on limestone cavern walls and mine timbers and walls, rarely as volcanic fumarole deposits, and as rare beds in evaporite layers such as those found in certain bodies of salt water. It occurs in association with melanterite, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Marcasite
The mineral marcasite, sometimes called “white iron pyrite”, is iron sulfide (FeS2) with orthorhombic crystal structure. It is physically and crystallographically distinct from pyrite, which is iron sulfide with cubic crystal structure. Both structures do have in common that they contain the disulfide S22− ion, having a short bonding distance between the sulfur atoms. The structures differ in how these di-anions are arranged around the Fe2+ cations. Marcasite is lighter and more brittle than pyrite. Specimens of marcasite often crumble and break up due to the unstable crystal structure. On fresh surfaces, it is pale yellow to almost white and has a bright metallic luster. It tarnishes to a yellowish or brownish color and gives a black streak. It is a brittle material that cannot be scratched with a knife. The thin, flat, tabular crystals, when joined in groups, are called “cockscombs”. In marcasite jewellery, pyrite used as a gemstone is called “marcasite” � ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Volcanic
A volcano is a rupture in the crust of a planetary-mass object, such as Earth, that allows hot lava, volcanic ash, and gases to escape from a magma chamber below the surface. On Earth, volcanoes are most often found where tectonic plates are diverging or converging, and most are found underwater. For example, a mid-ocean ridge, such as the Mid-Atlantic Ridge, has volcanoes caused by divergent tectonic plates whereas the Pacific Ring of Fire has volcanoes caused by convergent tectonic plates. Volcanoes can also form where there is stretching and thinning of the crust's plates, such as in the East African Rift and the Wells Gray-Clearwater volcanic field and Rio Grande rift in North America. Volcanism away from plate boundaries has been postulated to arise from upwelling diapirs from the core–mantle boundary, deep in the Earth. This results in hotspot volcanism, of which the Hawaiian hotspot is an example. Volcanoes are usually not created where two tectoni ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron(II) Minerals
Iron () is a chemical element with symbol Fe (from la, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, right in front of oxygen (32.1% and 30.1%, respectively), forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust. In its metallic state, iron is rare in the Earth's crust, limited mainly to deposition by meteorites. Iron ores, by contrast, are among the most abundant in the Earth's crust, although extracting usable metal from them requires kilns or furnaces capable of reaching or higher, about higher than that required to smelt copper. Humans started to master that process in Eurasia during the 2nd millennium BCE and the use of iron tools and weapons began to displace copper alloys, in some regions, only around 1200 BCE. That event is considered the transition from the Bronze Age to the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halotrichite
Halotrichite, also known as ''feather alum'', is a highly hydrated sulfate of aluminium and iron. Its chemical formula is FeAl2(SO4)4·22H2O. It forms fibrous monoclinic crystals. The crystals are water-soluble. It is formed by the weathering and decomposition of pyrite commonly near or in volcanic vents. The locations of natural occurrences include: the Atacama Desert, Chile; Dresden in Saxony, Germany; San Juan County, Utah; Iceland and Mont Saint-Hilaire, Canada. The name is from Latin: ''halotrichum'' for salt hair which accurately describes the precipitate/evaporite mineral. ;Gallery File:Halotrichite Hydrous iron aluminum sulfate Corral, California 3009.jpg, Halotrichite from California File:Halotrichite-179634.jpg, Halotrichite from the abandoned Golden Queen mine on Soledad Mountain south of Mojave, California Mojave (formerly Mohave) is an unincorporated community in Kern County, California, United States. Mojave is located east of Bakersfield, and north of Lo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |