|
Low Blood Potassium
Hypokalemia is a low level of potassium (K+) in the blood serum. Mild low potassium does not typically cause symptoms. Symptoms may include feeling tired, leg cramps, weakness, and constipation. Low potassium also increases the risk of an abnormal heart rhythm, which is often too slow and can cause cardiac arrest. Causes of hypokalemia include vomiting, diarrhea, medications like furosemide and steroids, dialysis, diabetes insipidus, hyperaldosteronism, hypomagnesemia, and not enough intake in the diet. Normal potassium levels are between 3.5 and 5.0 mmol/L (3.5 and 5.0 mEq/L) with levels below 3.5 mmol/L defined as hypokalemia. It is classified as severe when levels are less than 2.5 mmol/L. Low levels may also be suspected based on an electrocardiogram (ECG). Hyperkalemia is a high level of potassium in the blood serum. The speed at which potassium should be replaced depends on whether or not there are symptoms or abnormalities on an electrocardiogram. Potassi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ST Segment
In electrocardiography, the ST segment connects the QRS complex and the T wave and has a duration of 0.005 to 0.150 sec (5 to 150 ms). It starts at the J point (junction between the QRS complex and ST segment) and ends at the beginning of the T wave. However, since it is usually difficult to determine exactly where the ST segment ends and the T wave begins, the relationship between the ST segment and T wave should be examined together. The typical ST segment duration is usually around 0.08 sec (80 ms). It should be essentially level with the PR and TP segments. The ST segment represents the isoelectric period when the ventricles are in between depolarization and repolarization. Interpretation * The normal ST segment has a slight upward concavity. * Flat, downsloping, or depressed ST segments may indicate coronary ischemia. * ST elevation may indicate transmural myocardial infarction. An elevation of >1mm and longer than 80 milliseconds following the J-point. This measure h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium
Potassium is the chemical element with the symbol K (from Neo-Latin ''kalium'') and atomic number19. Potassium is a silvery-white metal that is soft enough to be cut with a knife with little force. Potassium metal reacts rapidly with atmospheric oxygen to form flaky white potassium peroxide in only seconds of exposure. It was first isolated from potash, the ashes of plants, from which its name derives. In the periodic table, potassium is one of the alkali metals, all of which have a single valence electron in the outer electron shell, that is easily removed to create an ion with a positive charge – a cation, that combines with anions to form salts. Potassium in nature occurs only in ionic salts. Elemental potassium reacts vigorously with water, generating sufficient heat to ignite hydrogen emitted in the reaction, and burning with a lilac- colored flame. It is found dissolved in sea water (which is 0.04% potassium by weight), and occurs in many minerals such as orthocl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic table) it occurs naturally only in combination with other elements and it almost always has an oxidation state of +2. It reacts readily with air to form a thin passivation coating of magnesium oxide that inhibits further corrosion of the metal. The free metal burns with a brilliant-white light. The metal is obtained mainly by electrolysis of magnesium salts obtained from brine. It is less dense than aluminium and is used primarily as a component in strong and lightweight alloys that contain aluminium. In the cosmos, magnesium is produced in large, aging stars by the sequential addition of three helium nuclei to a carbon nucleus. When such stars explode as supernovas, much of the magnesium is expelled into the interstellar medium where it m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
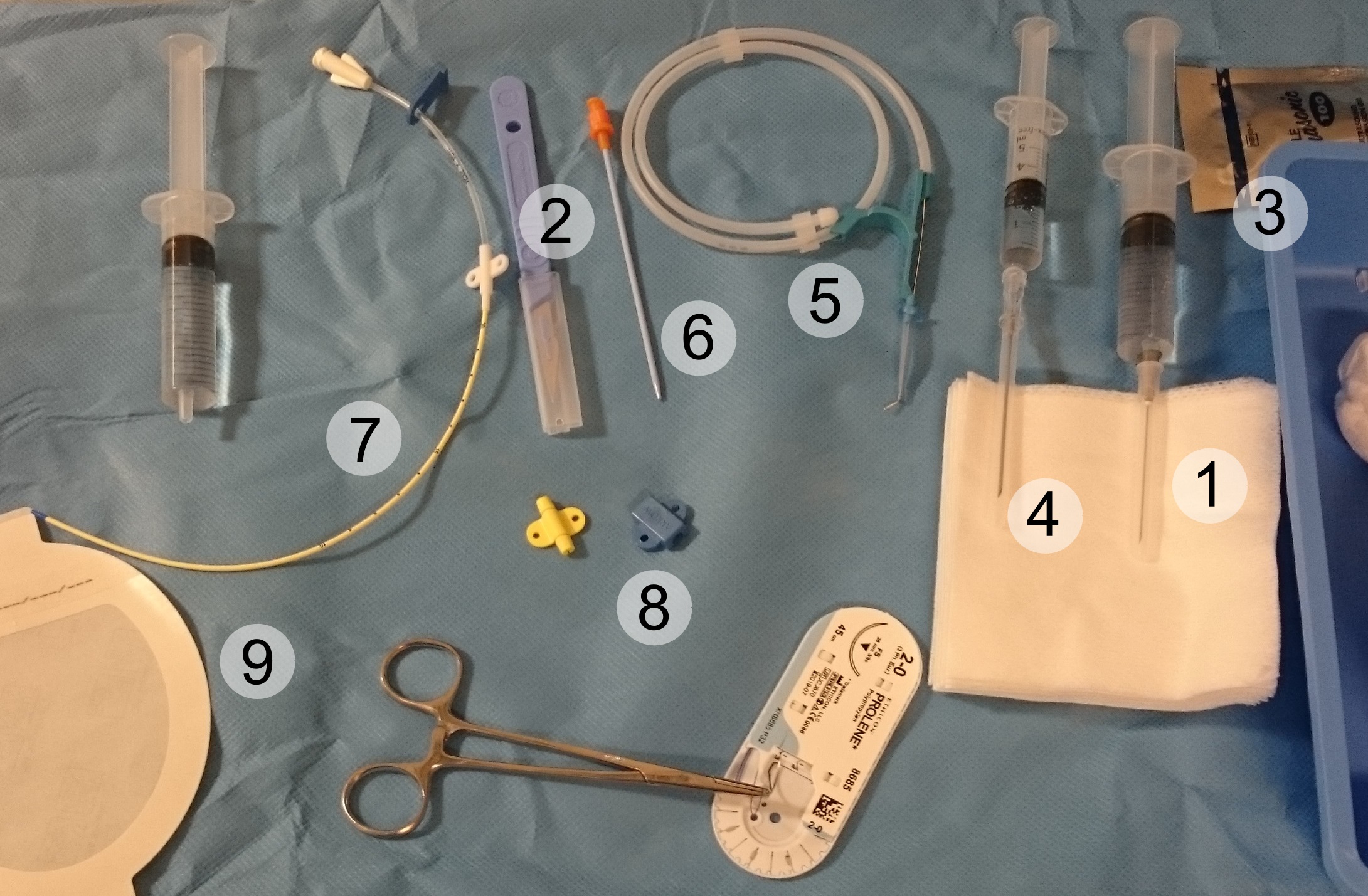
Central Venous Catheter
A central venous catheter (CVC), also known as a central line(c-line), central venous line, or central venous access catheter, is a catheter placed into a large vein. It is a form of venous access. Placement of larger catheters in more centrally located veins is often needed in critically ill patients, or in those requiring prolonged intravenous therapies, for more reliable vascular access. These catheters are commonly placed in veins in the neck (internal jugular vein), chest ( subclavian vein or axillary vein), groin (femoral vein), or through veins in the arms (also known as a PICC line, or peripherally inserted central catheters). Central lines are used to administer medication or fluids that are unable to be taken by mouth or would harm a smaller peripheral vein, obtain blood tests (specifically the "central venous oxygen saturation"), administer fluid or blood products for large volume resuscitation, and measure central venous pressure. The catheters used are commonly ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Intravenous Therapy
Intravenous therapy (abbreviated as IV therapy) is a medical technique that administers fluids, medications and nutrients directly into a person's vein. The intravenous route of administration is commonly used for rehydration or to provide nutrients for those who cannot, or will not—due to reduced mental states or otherwise—consume food or water by mouth. It may also be used to administer medications or other medical therapy such as blood products or electrolytes to correct electrolyte imbalances. Attempts at providing intravenous therapy have been recorded as early as the 1400s, but the practice did not become widespread until the 1900s after the development of techniques for safe, effective use. The intravenous route is the fastest way to deliver medications and fluid replacement throughout the body as they are introduced directly into the circulatory system and thus quickly distributed. For this reason, the intravenous route of administration is also used for the cons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrocardiography
Electrocardiography is the process of producing an electrocardiogram (ECG or EKG), a recording of the heart's electrical activity. It is an electrogram of the heart which is a graph of voltage versus time of the electrical activity of the heart using electrodes placed on the skin. These electrodes detect the small electrical changes that are a consequence of cardiac muscle depolarization followed by repolarization during each cardiac cycle (heartbeat). Changes in the normal ECG pattern occur in numerous cardiac abnormalities, including cardiac rhythm disturbances (such as atrial fibrillation and ventricular tachycardia), inadequate coronary artery blood flow (such as myocardial ischemia and myocardial infarction), and electrolyte disturbances (such as hypokalemia and hyperkalemia). Traditionally, "ECG" usually means a 12-lead ECG taken while lying down as discussed below. However, other devices can record the electrical activity of the heart such as a Holter monitor but al ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hyperkalemia
Hyperkalemia is an elevated level of potassium (K+) in the blood. Normal potassium levels are between 3.5 and 5.0mmol/L (3.5 and 5.0mEq/L) with levels above 5.5mmol/L defined as hyperkalemia. Typically hyperkalemia does not cause symptoms. Occasionally when severe it can cause palpitations, muscle pain, muscle weakness, or numbness. Hyperkalemia can cause an abnormal heart rhythm which can result in cardiac arrest and death. Common causes of hyperkalemia include kidney failure, hypoaldosteronism, and rhabdomyolysis. A number of medications can also cause high blood potassium including spironolactone, NSAIDs, and angiotensin converting enzyme inhibitors. The severity is divided into mild (5.5–5.9mmol/L), moderate (6.0–6.4mmol/L), and severe (>6.5mmol/L). High levels can be detected on an electrocardiogram (ECG). Pseudohyperkalemia, due to breakdown of cells during or after taking the blood sample, should be ruled out. Initial treatment in those with ECG changes is sa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
MEq/L
An equivalent (symbol: officially equiv; unofficially but often Eq) is the amount of a substance that reacts with (or is ''equivalent'' to) an arbitrary amount (typically one mole) of another substance in a given chemical reaction. It is an archaic unit of measurement that was used in chemistry and the biological sciences (see '). The mass of an equivalent is called its equivalent weight. Formula : ''milligrams to = mg x V / MW'' :: Example for elemental compounds: (mg element / elemental compound mass fraction) x V / MV : '' to milligrams = mEq x MW / V'' Common examples mEq to milligram Milligram to mEq Formal definition In a more formal definition, the ''equivalent'' is the amount of a substance needed to do one of the following: * react with or supply one mole of hydrogen ions () in an acid–base reaction * react with or supply one mole of electrons in a redox reaction. The "hydrogen ion" and the "electron" in these examples are respectively called the "react ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mmol/L
Molar concentration (also called molarity, amount concentration or substance concentration) is a measure of the concentration of a chemical species, in particular of a solute in a solution, in terms of amount of substance per unit volume of solution. In chemistry, the most commonly used unit for molarity is the number of moles per liter, having the unit symbol mol/L or mol/ dm3 in SI unit. A solution with a concentration of 1 mol/L is said to be 1 molar, commonly designated as 1 M. Definition Molar concentration or molarity is most commonly expressed in units of moles of solute per litre of solution. For use in broader applications, it is defined as amount of substance of solute per unit volume of solution, or per unit volume available to the species, represented by lowercase c: :c = \frac = \frac = \frac. Here, n is the amount of the solute in moles, N is the number of constituent particles present in volume V (in litres) of the solution, and N_\text is the Avo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diarrhea
Diarrhea, also spelled diarrhoea, is the condition of having at least three loose, liquid, or watery bowel movements each day. It often lasts for a few days and can result in dehydration due to fluid loss. Signs of dehydration often begin with loss of the normal stretchiness of the skin and irritable behaviour. This can progress to decreased urination, loss of skin color, a fast heart rate, and a decrease in responsiveness as it becomes more severe. Loose but non-watery stools in babies who are exclusively breastfed, however, are normal. The most common cause is an infection of the intestines due to either a virus, bacterium, or parasite—a condition also known as gastroenteritis. These infections are often acquired from food or water that has been contaminated by feces, or directly from another person who is infected. The three types of diarrhea are: short duration watery diarrhea, short duration bloody diarrhea, and persistent diarrhea (lasting more than two weeks ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cardiac Arrest
Cardiac arrest is when the heart suddenly and unexpectedly stops beating. It is a medical emergency that, without immediate medical intervention, will result in sudden cardiac death within minutes. Cardiopulmonary resuscitation (CPR) and possibly defibrillation are needed until further treatment can be provided. Cardiac arrest results in a rapid loss of consciousness, and breathing may be abnormal or absent. While cardiac arrest may be caused by heart attack or heart failure, these are not the same, and in 15 to 25% of cases, there is a non-cardiac cause. Some individuals may experience chest pain, shortness of breath, nausea, an elevated heart rate, and a light-headed feeling immediately before entering cardiac arrest. The most common cause of cardiac arrest is an underlying heart problem like coronary artery disease that decreases the amount of oxygenated blood supplying the heart muscle. This, in turn, damages the structure of the muscle, which can alter its fun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |