|
Ledipasvir
Ledipasvir is a drug for the treatment of hepatitis C that was developed by Gilead Sciences. After completing Phase III clinical trials, on February 10, 2014, Gilead filed for U.S. approval of a ledipasvir/sofosbuvir fixed-dose combination tablet for genotype 1 hepatitis C. The ledipasvir/sofosbuvir combination is a direct-acting antiviral agent that interferes with HCV replication and can be used to treat patients with genotypes 1a or 1b without PEG-interferon or ribavirin. Ledipasvir is an inhibitor of NS5A, a hepatitis C virus protein. Data presented at the 20th Conference on Retroviruses and Opportunistic Infections in March 2013 showed that a triple regimen of the nucleotide analog inhibitor sofosbuvir, ledipasvir, and ribavirin produced a 12-week post-treatment sustained virological response (SVR12) rate of 100% for both treatment-naive patients and prior non-responders with HCV genotype 1. The sofosbuvir/ledipasvir coformulation is being tested with and without rib ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ledipasvir/sofosbuvir
Ledipasvir/sofosbuvir, sold under the trade name Harvoni among others, is a medication used to treat hepatitis C. It is a fixed-dose combination of ledipasvir and sofosbuvir. Cure rates are 94% to 99% in people infected with hepatitis C virus (HCV) genotype 1. Some evidence also supports use in HCV genotype 3 and 4. It is taken daily by mouth for 8–24 weeks. It is generally well tolerated. Common side effects include muscle pains, headache, nausea, rash, and cough. It is unclear if use in pregnancy is safe for the baby. Ledipasvir works by decreasing the activity of NS5A and sofosbuvir works by decreasing the activity of NS5B polymerase. Ledipasvir/sofosbuvir was approved for medical use in the United States, in the European Union, and in Canada in 2014. It is on the World Health Organization's List of Essential Medicines. Medical uses Cure rates are 94% to 99% in people infected with genotype 1 (46% of HCV cases). It has also been evaluated for the treatment of infecti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sofosbuvir
Sofosbuvir, sold under the brand name Sovaldi among others, is a medication used to treat hepatitis C. It is taken by mouth. Common side effects include fatigue, headache, nausea, and trouble sleeping. Side effects are generally more common in interferon-containing regimens. Sofosbuvir may reactivate hepatitis B in those who have been previously infected. In combination with ledipasvir, daclatasvir or simeprevir, it is not recommended with amiodarone due to the risk of an abnormally slow heartbeat. Sofosbuvir is in the nucleotide analog family of medications and works by blocking the hepatitis C NS5B protein. Sofosbuvir was discovered in 2007 and approved for medical use in the United States in 2013. It is on the World Health Organization's List of Essential Medicines. Medical uses Initial HCV treatment In 2016, the American Association for the Study of Liver Diseases and the Infectious Diseases Society of America jointly published a recommendation for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hepatitis C
Hepatitis C is an infectious disease caused by the hepatitis C virus (HCV) that primarily affects the liver; it is a type of viral hepatitis. During the initial infection period, people often have mild or no symptoms. Early symptoms can include fever, dark urine, abdominal pain, and jaundice, yellow tinged skin. The virus persists in the liver, becoming Chronic condition, chronic, in about 70% of those initially infected. Early on, chronic infection typically has no symptoms. Over many years however, it often leads to liver disease and occasionally cirrhosis. In some cases, those with cirrhosis will develop serious complications such as liver failure, hepatocellular carcinoma, liver cancer, or esophageal varices, dilated blood vessels in the esophagus and gastric varices, stomach. HCV is spread primarily by blood-to-blood contact associated with injection drug use, poorly sterilized medical equipment, needlestick injuries in healthcare, and blood transfusions, transfusions. In r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gilead Sciences
Gilead Sciences, Inc. () is an American biopharmaceutical company headquartered in Foster City, California, that focuses on researching and developing antiviral drugs used in the treatment of HIV/AIDS, hepatitis B, hepatitis C, influenza, and COVID-19, including ledipasvir/sofosbuvir and sofosbuvir. Gilead is a member of the Nasdaq-100 and the S&P 100. Gilead was founded in 1987 under the name Oligogen by Michael L. Riordan. The original name was a reference to oligonucleotides, small strands of DNA used to target genetic sequences. Gilead held its initial public offering in 1992, and successfully developed drugs like Tamiflu and Vistide that decade. In the 2000s, Gilead received approval for drugs including Viread and Hepsera, among others. It began evolving from a biotechnology company into a pharmaceutical company, acquiring several subsidiaries, though it still relied heavily on contracting to manufacture its drugs. The company continued its growth in the 2010s. Howev ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
NS5B
Nonstructural protein 5B (NS5B) is a viral protein found in the hepatitis C virus (HCV). It is an RNA-dependent RNA polymerase, having the key function of replicating HCV's viral RNA by using the viral positive RNA strand as a template to catalyze the polymerization of ribonucleoside triphosphates (rNTP) during RNA replication. Several crystal structures of NS5B polymerase in several crystalline forms have been determined based on the same consensus sequence BK (HCV-BK, genotype 1). The structure can be represented by a right hand shape with fingers, palm, and thumb. The encircled active site, unique to NS5B, is contained within the palm structure of the protein. Recent studies on NS5B protein genotype 1b strain J4's (HC-J4) structure indicate a presence of an active site where possible control of nucleotide binding occurs and initiation of de-novo RNA synthesis. De-novo adds necessary primers for initiation of RNA replication. Drugs targeting NS5B Several drugs are either on the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
P-glycoprotein
P-glycoprotein 1 (permeability glycoprotein, abbreviated as P-gp or Pgp) also known as multidrug resistance protein 1 (MDR1) or ATP-binding cassette sub-family B member 1 (ABCB1) or cluster of differentiation 243 (CD243) is an important protein of the cell membrane that pumps many foreign substances out of cells. More formally, it is an ATP-dependent efflux pump with broad substrate specificity. It exists in animals, fungi, and bacteria, and it likely evolved as a defense mechanism against harmful substances. P-gp is extensively distributed and expressed in the intestinal epithelium where it pumps xenobiotics (such as toxins or drugs) back into the intestinal lumen, in liver cells where it pumps them into bile ducts, in the cells of the proximal tubule of the kidney where it pumps them into urinary filtrate (in the proximal tubule), and in the capillary endothelial cells composing the blood–brain barrier and blood–testis barrier, where it pumps them back into the ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclopropanes
Cyclopropanes are a family of organic compounds containing the cyclopropyl group. The parent is cyclopropane (). Synthesis and reactions Most cyclopropanes are not prepared from the parent cyclopropane, which is somewhat inert. Instead, yclopropyl groups are often prepared by cyclization of 1,3-difunctional alkanes. An example of the former, cyclopropyl cyanide is prepared by the reaction of 4- chlorobutyronitrile with a strong base. Phenylcyclopropane is produced analogously from the 1,3-dibromide. A second major route to cyclopropanes entails addition of methylene (or its substituted derivatives) to an alkene, a process called cyclopropanation. Substituted cyclopropanes undergo the reactions associated with the cyclopropyl ring or the substituents. Vinylcyclopropanes are a special case as they undergo vinylcyclopropane rearrangement. Simple substituted cyclopropanes * Chlorocyclopropane * Cyclopropane carboxylic acid * Cyclopropyl amine * Cyclopropyl cyanide * Cycloprop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzimidazoles
Benzimidazole is a heterocyclic aromatic organic compound. This bicyclic compound may be viewed as fused rings of the aromatic compounds benzene and imidazole. It is a white solid that appears in form of tabular crystals. Preparation Benzimidazole was discovered during research on vitamin B12. The benzimidazole nucleus was found to be a stable platform on which drugs could be developed. Benzimidazole is produced by condensation of o-phenylenediamine with formic acid, or the equivalent trimethyl orthoformate: :C6H4(NH2)2 + HC(OCH3)3 → C6H4N(NH)CH + 3 CH3OH 2-Substituted derivatives are obtained when the condensation is conducted with aldehydes in place of formic acid, followed by oxidation. Reactions Benzimidazole is a base: :C6H4N(NH)CH + H+ → 6H4(NH)2CHsup>+ It can also be deprotonated with stronger bases: :C6H4N(NH)CH + LiH → Li 6H4N2CH + H2 The imine can be alkylated and also serves as a ligand in coordination chemistry. The most pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
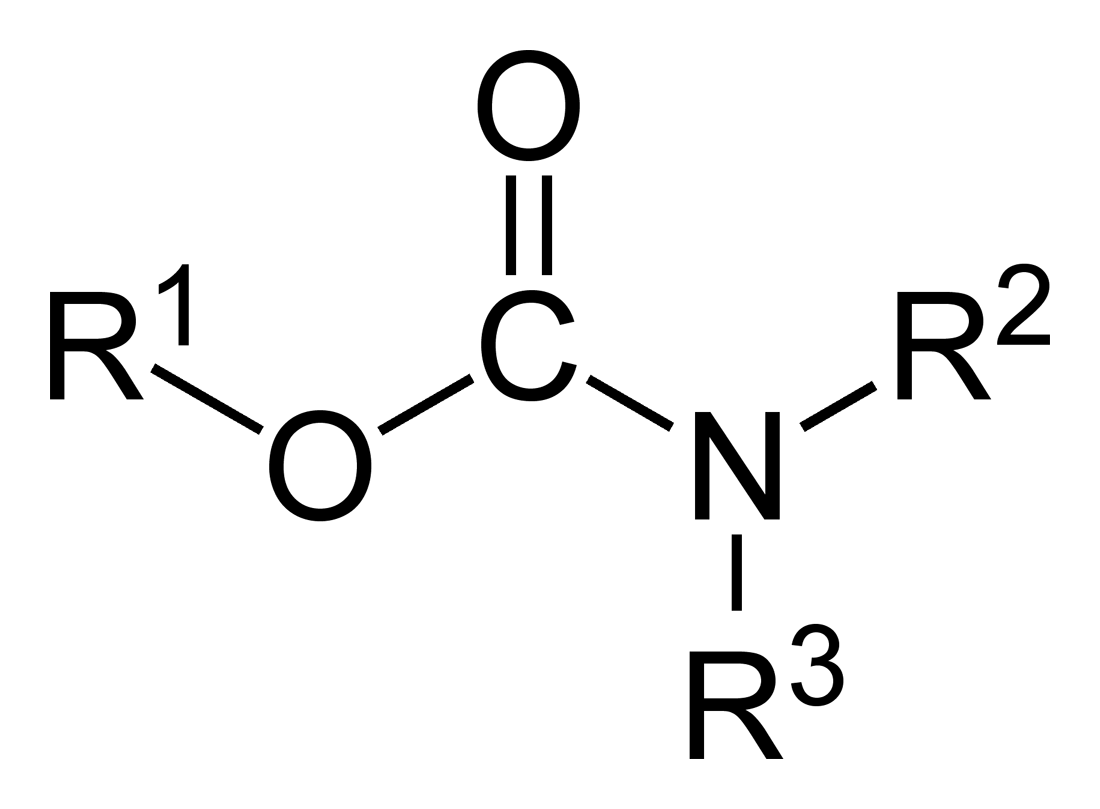
Carbamates
In organic chemistry, a carbamate is a category of organic compounds with the general formula and structure , which are formally derived from carbamic acid (). The term includes organic compounds (e.g., the ester ethyl carbamate), formally obtained by replacing one or more of the hydrogen atoms by other organic functional groups; as well as salts with the carbamate anion (e.g. ammonium carbamate). Polymers whose repeat units are joined by carbamate like groups are an important family of plastics, the polyurethanes. See for clarification. Properties While carbamic acids are unstable, many carbamate esters and salts are stable and well known. Equilibrium with carbonate and bicarbonate In water solutions, the carbamate anion slowly equilibrates with the ammonium cation and the carbonate or bicarbonate anions: : : Calcium carbamate is soluble in water, whereas calcium carbonate is not. Adding a calcium salt to an ammonium carbamate/carbonate solution will precipitate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorenes
Fluorene , or 9''H''-fluorene is an organic compound with the formula (C6H4)2CH2. It forms white crystals that exhibit a characteristic, aromatic odor similar to that of naphthalene. Despite its name, it does not contain the element fluorine, but rather it comes from the violet fluorescence it exhibits. For commercial purposes it is obtained from coal tar, where it was discovered and named by Marcellin Berthelot in 1867. It is insoluble in water and soluble in many organic solvents. Although sometimes classified as a polycyclic aromatic hydrocarbon, the five-membered ring has no aromatic properties. Fluorene is mildly acidic. Synthesis, structure, and reactivity Although fluorene is obtained from coal tar, it can also be prepared by dehydrogenation of diphenylmethane. Alternatively, it can be prepared by the reduction of fluorenone with zinc or hypophosphorous acid–iodine. The fluorene molecule is nearly planar,D. M. Burns, John Iball (1954), ''Molecular Structure of Fluorene ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
NS5A Inhibitors
Nonstructural protein 5A (NS5A) is a Zinc-binding protein, zinc-binding and proline-rich Hydrophile, hydrophilic phosphoprotein that plays a key role in Hepatitis C virus RNA replication. It appears to be a dimeric form without ''trans''-membrane helices. Structure NS5A is derived from a large polyprotein that is translated from the HCV genome, and undergoes post-translation processing by NS3 (HCV), nonstructural protein 3 (NS3) viral protease. Despite no inherent enzymatic activity being attributed to NS5A, its function is mediated through interaction with other nonstructural (NS) viral and cellular proteins. NS5A has two phosphorylated forms: p56 and p58, which differ in the electrophoretic mobility. p56 is basally phosphorylated by host cellular protein kinase at the center and near the C terminus, whereas p58 is a form of hyper-phosphorylated NS5A at the center of the serine-rich region. Protein mass spectrometry identified several phosphorylated serine residues in this region ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |