|
Bancroft Point
A Bancroft point is the temperature where an azeotrope occurs in a binary system. Although vapor liquid azeotropy is impossible for binary systems which are rigorously described by Raoult's law, for real systems, azeotropy is inevitable at temperatures where the saturation vapor pressure of the components are equal. Such a temperature is called a Bancroft point. However, not all azeotropic binary systems exhibit such a point. Also, a Bancroft point must lie in the valid temperature ranges of the Antoine equation. Bancroft point is named after Wilder Dwight Bancroft. See also * Raoult's law * Vapor–liquid equilibrium In thermodynamics and chemical engineering, the vapor–liquid equilibrium (VLE) describes the distribution of a chemical species between the vapor phase and a liquid phase. The concentration of a vapor in contact with its liquid, especially a ... * Bancroft rule External links Separation of Azeotropic Mixtures Phase transitions Thermodynamics Distillatio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Azeotrope
An azeotrope () or a constant heating point mixture is a mixture of two or more liquids whose proportions cannot be altered or changed by simple distillation.Moore, Walter J. ''Physical Chemistry'', 3rd e Prentice-Hall 1962, pp. 140–142 This happens when an azeotrope is boiled, the vapour has the same proportions of constituents as the unboiled mixture. Because their composition is unchanged by distillation, azeotropes are also called (especially in older texts) ''constant boiling point mixtures''. Some azeotropic mixtures of pairs of compounds are known, and many azeotropes of three or more compounds are also known. In such a case it is not possible to separate the components by fractional distillation and azeotropic distillation is usually used instead. There are two types of azeotropes: minimum boiling azeotrope and maximum boiling azeotrope. A solution that shows greater positive deviation from Raoult's law forms a minimum boiling azeotrope at a specific composition. For ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mixture
In chemistry, a mixture is a material made up of two or more different chemical substances which are not chemically bonded. A mixture is the physical combination of two or more substances in which the identities are retained and are mixed in the form of solutions, suspensions and colloids. Mixtures are one product of mechanically blending or mixing chemical substances such as elements and compounds, without chemical bonding or other chemical change, so that each ingredient substance retains its own chemical properties and makeup. Despite the fact that there are no chemical changes to its constituents, the physical properties of a mixture, such as its melting point, may differ from those of the components. Some mixtures can be separated into their components by using physical (mechanical or thermal) means. Azeotropes are one kind of mixture that usually poses considerable difficulties regarding the separation processes required to obtain their constituents (physical or chemic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
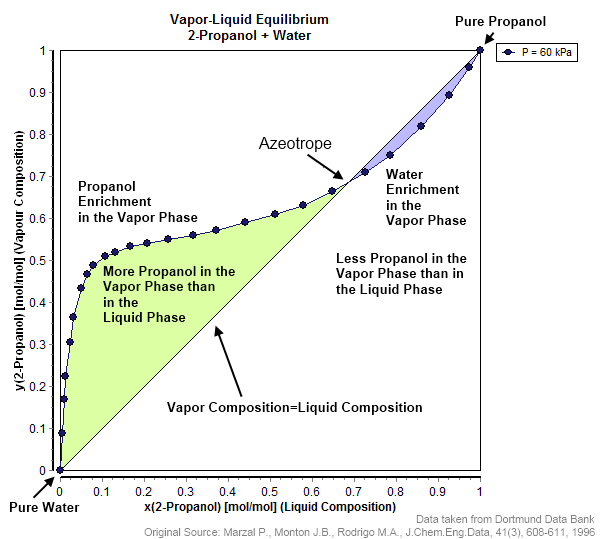
Vapor–liquid Equilibrium
In thermodynamics and chemical engineering, the vapor–liquid equilibrium (VLE) describes the distribution of a chemical species between the vapor phase and a liquid phase. The concentration of a vapor in contact with its liquid, especially at equilibrium, is often expressed in terms of vapor pressure, which will be a partial pressure (a part of the total gas pressure) if any other gas(es) are present with the vapor. The equilibrium vapor pressure of a liquid is in general strongly dependent on temperature. At vapor–liquid equilibrium, a liquid with individual components in certain concentrations will have an equilibrium vapor in which the concentrations or partial pressures of the vapor components have certain values depending on all of the liquid component concentrations and the temperature. The converse is also true: if a vapor with components at certain concentrations or partial pressures is in vapor–liquid equilibrium with its liquid, then the component concentrati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vapor Pressure
Vapor pressure (or vapour pressure in English-speaking countries other than the US; see spelling differences) or equilibrium vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases (solid or liquid) at a given temperature in a closed system. The equilibrium vapor pressure is an indication of a liquid's evaporation rate. It relates to the tendency of particles to escape from the liquid (or a solid). A substance with a high vapor pressure at normal temperatures is often referred to as '' volatile''. The pressure exhibited by vapor present above a liquid surface is known as vapor pressure. As the temperature of a liquid increases, the kinetic energy of its molecules also increases. As the kinetic energy of the molecules increases, the number of molecules transitioning into a vapor also increases, thereby increasing the vapor pressure. The vapor pressure of any substance increases non-linearly with temperature accord ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antoine Equation
The Antoine equation is a class of semi-empirical correlations describing the relation between vapor pressure and temperature for pure substances. The Antoine equation is derived from the Clausius–Clapeyron relation. The equation was presented in 1888 by the French engineer (1825–1897). Equation The Antoine equation is :\log_ p = A-\frac. where ''p'' is the vapor pressure, is temperature (in °C or in K according to the value of C) and , and are component-specific constants. The simplified form with set to zero: :\log_ p = A-\frac is the August equation, after the German physicist Ernst Ferdinand August (1795–1870). The August equation describes a linear relation between the logarithm of the pressure and the reciprocal temperature. This assumes a temperature-independent heat of vaporization. The Antoine equation allows an improved, but still inexact description of the change of the heat of vaporization with the temperature. The Antoine equation can also be transfo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wilder Dwight Bancroft
Wilder Dwight Bancroft (October 1, 1867 – February 7, 1953) was an American physical chemist. Biography Born in Middletown, Rhode Island, he was the grandson of historian and statesman George Bancroft and great-grandson of Aaron Bancroft. He received a B.A. from Harvard University in 1888, and a Ph.D. from University of Leipzig in 1892, as well as honorary SCDs from Lafayette College (in 1919) and Cambridge University (in 1923). He was an assistant chemistry instructor at Harvard University from 1888–1889 and 1893–1894, then a full instructor from 1894-1895. He then became an assistant professor at Cornell University in 1895, then a full professor (at Cornell) in 1903. He was elected a Fellow of the American Academy of Arts and Sciences in 1913, and was elected to the National Academy of Sciences in 1920.http://www.nasonline.org/publications/biographical-memoirs/memoir-pdfs/bancroft-wilder-d.pdf Bancroft was trained by Wilhelm Ostwald and Jacobus Henricus van 't ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bancroft Rule
The Bancroft rule in colloidal chemistry states: "The phase in which an emulsifier is more soluble constitutes the continuous phase." This means that water-soluble surfactants tend to give oil-in-water emulsions and oil-soluble surfactants give water-in-oil emulsions. It is a general rule of thumb, still used, but regarded as inferior to HLD theory (Hydrophilic Lipophilic Difference), which takes many more factors into consideration. It was named after Wilder Dwight Bancroft, an American physical chemist, who proposed the rule in the 1910s. Technical details In all of the typical emulsions, there are tiny particles (discrete phase) suspended in a liquid (continuous phase). In an oil-in-water emulsion, oil is the discrete phase, while water is the continuous phase. What the Bancroft rule states is that contrary to common sense, what makes an emulsion oil-in-water or water-in-oil is not the relative percentages of oil or water, but which phase the emulsifier is more soluble in. S ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phase Transitions
In chemistry, thermodynamics, and other related fields, a phase transition (or phase change) is the physical process of transition between one state of a medium and another. Commonly the term is used to refer to changes among the basic states of matter: solid, liquid, and gas, and in rare cases, plasma. A phase of a thermodynamic system and the states of matter have uniform physical properties. During a phase transition of a given medium, certain properties of the medium change as a result of the change of external conditions, such as temperature or pressure. This can be a discontinuous change; for example, a liquid may become gas upon heating to its boiling point, resulting in an abrupt change in volume. The identification of the external conditions at which a transformation occurs defines the phase transition point. Types of phase transition At the phase transition point for a substance, for instance the boiling point, the two phases involved - liquid and vapor, have identic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thermodynamics
Thermodynamics is a branch of physics that deals with heat, work, and temperature, and their relation to energy, entropy, and the physical properties of matter and radiation. The behavior of these quantities is governed by the four laws of thermodynamics which convey a quantitative description using measurable macroscopic physical quantities, but may be explained in terms of microscopic constituents by statistical mechanics. Thermodynamics applies to a wide variety of topics in science and engineering, especially physical chemistry, biochemistry, chemical engineering and mechanical engineering, but also in other complex fields such as meteorology. Historically, thermodynamics developed out of a desire to increase the efficiency of early steam engines, particularly through the work of French physicist Sadi Carnot (1824) who believed that engine efficiency was the key that could help France win the Napoleonic Wars. Scots-Irish physicist Lord Kelvin was the first to formula ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Distillation
Distillation, or classical distillation, is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation, usually inside an apparatus known as a still. Dry distillation is the heating of solid materials to produce gaseous products (which may condense into liquids or solids); this may involve chemical changes such as destructive distillation or cracking. Distillation may result in essentially complete separation (resulting in nearly pure components), or it may be a partial separation that increases the concentration of selected components; in either case, the process exploits differences in the relative volatility of the mixture's components. In industrial applications, distillation is a unit operation of practically universal importance, but is a physical separation process, not a chemical reaction. An installation used for distillation, especially of distilled beverages, is a distillery. Distillation includes th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |