|
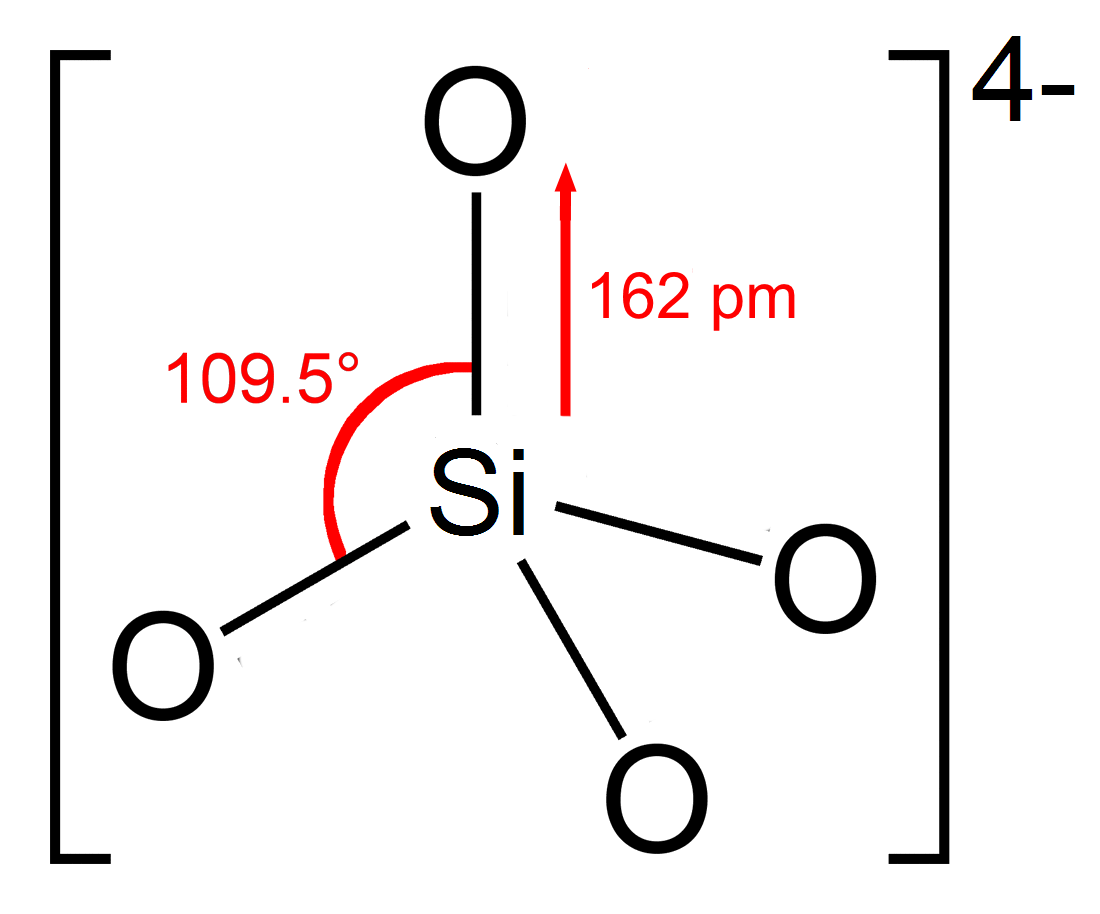
Barium Orthosilicate
In chemistry, orthosilicate is the anion , or any of its salts and esters. It is one of the silicate anions. It is occasionally called the silicon tetroxide anion or group.C. A. Kumins, and A. E. Gessler (1953), "Short-Cycle Syntheses of Ultramarine Blue". ''Indunstrial & Engineering Chemistry'', volume 45, issue 3, pages 567–572. Orthosilicate salts, like sodium orthosilicate, are stable, and occur widely in nature as silicate minerals, being the defining feature of the nesosilicates. Olivine, a magnesium or iron(II) orthosilicate, is the most abundant mineral in the upper mantle. The orthosilicate anion is a strong base, the conjugate base of the extremely weak orthosilicic acid (p''K''a2 = 13.2 at 25 °C). This equilibrium is difficult to study since the acid tends to decompose into a hydrated silica condensate. Structure The orthosilicate ion or group has tetrahedral shape, with one silicon atom surrounded by four oxygen atoms. In the anion, each oxygen carri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrahedron
In geometry, a tetrahedron (: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular Face (geometry), faces, six straight Edge (geometry), edges, and four vertex (geometry), vertices. The tetrahedron is the simplest of all the ordinary convex polytope, convex polyhedra. The tetrahedron is the three-dimensional case of the more general concept of a Euclidean geometry, Euclidean simplex, and may thus also be called a 3-simplex. The tetrahedron is one kind of pyramid (geometry), pyramid, which is a polyhedron with a flat polygon base and triangular faces connecting the base to a common point. In the case of a tetrahedron, the base is a triangle (any of the four faces can be considered the base), so a tetrahedron is also known as a "triangular pyramid". Like all convex polyhedra, a tetrahedron can be folded from a single sheet of paper. It has two such net (polyhedron), nets. For any tetrahedron there exists a sphere (called th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methanol
Methanol (also called methyl alcohol and wood spirit, amongst other names) is an organic chemical compound and the simplest aliphatic Alcohol (chemistry), alcohol, with the chemical formula (a methyl group linked to a hydroxyl group, often abbreviated as MeOH). It is a light, Volatility (chemistry), volatile, colorless and flammable liquid with a distinctive alcoholic odor similar to that of ethanol (potable alcohol), but is more acutely toxic than the latter. Methanol acquired the name wood alcohol because it was once produced through destructive distillation of wood. Today, methanol is mainly produced industrially by hydrogenation of carbon monoxide. Methanol consists of a methyl group linked to a polar hydroxyl group. With more than 20 million tons produced annually, it is used as a Precursor (chemistry), precursor to other commodity chemicals, including formaldehyde, acetic acid, methyl tert-butyl ether, methyl ''tert''-butyl ether, methyl benzoate, anisole, peroxyacids, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetramethyl Orthosilicate
Tetramethyl orthosilicate (TMOS) is the chemical compound with the formula Si(OCH3)4. This molecule consists of four methoxy groups bonded to a silicon atom. The basic properties are similar to the more popular tetraethyl orthosilicate, which is usually preferred because the product of hydrolysis, ethanol, is less toxic than methanol. Tetramethyl orthosilicate hydrolyzes to SiO2: :Si(OCH3)4 + 2 H2O → SiO2 + 4 CH3OH In organic synthesis, Si(OCH3)4 has been used to convert ketones and aldehydes to the corresponding ketals and acetal In organic chemistry, an acetal is a functional group with the connectivity . Here, the R groups can be organic fragments (a carbon atom, with arbitrary other atoms attached to that) or hydrogen, while the R' groups must be organic fragments n ...s, respectively.Sakurai, H. "Silicon(IV) Methoxide" in Encyclopedia of Reagents for Organic Synthesis 2001 John Wiley & Sons. Safety The hydrolysis of Si(OCH3)4 produces insoluble SiO2 and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetraethyl Orthosilicate
Tetraethyl orthosilicate, formally named tetraethoxysilane (TEOS), ethyl silicate is the organic chemical compound with the formula Si(OC2H5)4. TEOS is a colorless liquid. It degrades in water. TEOS is the of orthosilicic acid, Si(OH)4. It is the most prevalent alkoxide of silicon. TEOS is a tetrahedral molecule. Like its many analogues, it is prepared by alcoholysis of silicon tetrachloride: :SiCl4 + 4 EtOH → Si(OEt)4 + 4 HCl where Et is the ethyl group, C2H5, and thus EtOH is ethanol. Applications TEOS is mainly used as a crosslinking agent in silicone polymers and as a precursor to silicon dioxide in the semiconductor industry. TEOS is also used as the silica source for synthesis of some zeolites. Other applications include coatings for carpets and other objects. TEOS is used in the production of aerogel. These applications exploit the reactivity of the Si-OR bonds. TEOS has historically been used as an additive to alcohol based rocket fuels to decrease the heat f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Synthesis
Organic synthesis is a branch of chemical synthesis concerned with the construction of organic compounds. Organic compounds are molecules consisting of combinations of covalently-linked hydrogen, carbon, oxygen, and nitrogen atoms. Within the general subject of organic synthesis, there are many different types of synthetic routes that can be completed including total synthesis, Enantioselective synthesis, stereoselective synthesis, automated synthesis, and many more. Additionally, in understanding organic synthesis it is necessary to be familiar with the methodology, techniques, and applications of the subject. Total synthesis A total synthesis refers to the complete chemical synthesis of molecules from simple, Precursor (chemistry), natural precursors. Total synthesis is accomplished either via a linear or convergent approach. In a Linear synthesis, ''linear'' synthesis—often adequate for simple structures—several steps are performed sequentially until the molecule is com ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vacuum Tube
A vacuum tube, electron tube, thermionic valve (British usage), or tube (North America) is a device that controls electric current flow in a high vacuum between electrodes to which an electric voltage, potential difference has been applied. It takes the form of an evacuated tubular envelope of glass or sometimes metal containing electrodes connected to external connection pins. The type known as a thermionic tube or thermionic valve utilizes thermionic emission of electrons from a hot cathode for fundamental Electronics, electronic functions such as signal amplifier, amplification and current Rectifier, rectification. Non-thermionic types such as vacuum phototubes achieve electron emission through the photoelectric effect, and are used for such purposes as the detection of light and measurement of its intensity. In both types the electrons are accelerated from the cathode to the anode by the electric field in the tube. The first, and simplest, vacuum tube, the diode or Flem ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cathode Poisoning
In vacuum tubes and gas-filled tubes, a hot cathode or thermionic cathode is a cathode electrode which is heated to make it emit electrons due to thermionic emission. This is in contrast to a cold cathode, which does not have a heating element. The heating element is usually an electrical filament heated by a separate electric current passing through it. Hot cathodes typically achieve much higher power density than cold cathodes, emitting significantly more electrons from the same surface area. Cold cathodes rely on field electron emission or secondary electron emission from positive ion bombardment, and do not require heating. There are two types of hot cathode. In a ''directly heated cathode'', the filament is the cathode and emits the electrons. In an ''indirectly heated cathode'', the filament or ''heater'' heats a separate metal cathode electrode which emits the electrons. From the 1920s to the 1960s, a wide variety of electronic devices used hot-cathode vacuum tubes. Today, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Strontium
Strontium is a chemical element; it has symbol Sr and atomic number 38. An alkaline earth metal, it is a soft silver-white yellowish metallic element that is highly chemically reactive. The metal forms a dark oxide layer when it is exposed to air. Strontium has physical and chemical properties similar to those of its two vertical neighbors in the periodic table, calcium and barium. It occurs naturally mainly in the minerals celestine and strontianite, and is mostly mined from these. Both strontium and strontianite are named after Strontian, a village in Scotland near which the mineral was discovered in 1790 by Adair Crawford and William Cruickshank; it was identified as a new element the next year from its crimson-red flame test color. Strontium was first isolated as a metal in 1808 by Humphry Davy using the then newly discovered process of electrolysis. During the 19th century, strontium was mostly used in the production of sugar from sugar beets (see strontian proces ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Light-emitting Diode
A light-emitting diode (LED) is a semiconductor device that emits light when current flows through it. Electrons in the semiconductor recombine with electron holes, releasing energy in the form of photons. The color of the light (corresponding to the energy of the photons) is determined by the energy required for electrons to cross the band gap of the semiconductor. White light is obtained by using multiple semiconductors or a layer of light-emitting phosphor on the semiconductor device. Appearing as practical electronic components in 1962, the earliest LEDs emitted low-intensity infrared (IR) light. Infrared LEDs are used in remote-control circuits, such as those used with a wide variety of consumer electronics. The first visible-light LEDs were of low intensity and limited to red. Early LEDs were often used as indicator lamps, replacing small incandescent bulbs, and in seven-segment displays. Later developments produced LEDs available in visible, ultraviolet (U ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphor
A phosphor is a substance that exhibits the phenomenon of luminescence; it emits light when exposed to some type of radiant energy. The term is used both for fluorescent or phosphorescent substances which glow on exposure to ultraviolet or visible light, and cathodoluminescent substances which glow when struck by an electron beam ( cathode rays) in a cathode-ray tube. When a phosphor is exposed to radiation, the orbital electrons in its molecules are excited to a higher energy level; when they return to their former level they emit the energy as light of a certain color. Phosphors can be classified into two categories: fluorescent substances which emit the energy immediately and stop glowing when the exciting radiation is turned off, and phosphorescent substances which emit the energy after a delay, so they keep glowing after the radiation is turned off, decaying in brightness over a period of milliseconds to days. Fluorescent materials are used in applications in wh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Europium
Europium is a chemical element; it has symbol Eu and atomic number 63. It is a silvery-white metal of the lanthanide series that reacts readily with air to form a dark oxide coating. Europium is the most chemically reactive, least dense, and softest of the lanthanides. It is soft enough to be cut with a knife. Europium was discovered in 1896, provisionally designated as Σ; in 1901, it was named after the continent of Europe. Europium usually assumes the oxidation state +3, like other members of the lanthanide series, but compounds having oxidation state +2 are also common. All europium compounds with oxidation state +2 are slightly reducing. Europium has no significant biological role and is relatively non-toxic compared to other heavy metals. Most applications of europium exploit the phosphorescence of europium compounds. Europium is one of the rarest of the rare-earth elements on Earth.Stwertka, Albert. ''A Guide to the Elements'', Oxford University Press, 1996, p. 156. Ety ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |