|
4-Aminobiphenyl
4-Aminobiphenyl (4-APB) is an organic compound with the formula C6H5C6H4NH2. It is an amine derivative of biphenyl. It is a colorless solid, although aged samples can appear colored. 4-Aminobiphenyl was commonly used in the past as a rubber antioxidant and an intermediate for dyes. Exposure to this aryl-amine can happen through contact with chemical dyes and from inhalation of cigarette smoke. Researches showed that 4-aminobiphenyl is responsible for bladder cancer in humans and dogs by damaging DNA. Due to its carcinogenic effects, commercial production of 4-aminobiphenyl ceased in the United States in the 1950s. Synthesis and reactivity Like other aniline derivatives, 4-aminobiphenyl is weakly basic. It is prepared by reduction of 4-nitrobiphenyl, which, together with the 2-nitro derivatives, is obtained by nitration of biphenyl. Another reaction to synthesize 4-aminobiphenyl can be obtained by using 4-azidobiphenyl. This can be done by reacting 4-azidobiphenyl with diphosph ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glucuronidation
Glucuronidation is often involved in drug metabolism of substances such as drugs, pollutants, bilirubin, androgens, estrogens, mineralocorticoids, glucocorticoids, fatty acid derivatives, retinoids, and bile acids. These linkages involve glycosidic bonds. Mechanism Glucuronidation consists of transfer of the glucuronic acid component of uridine diphosphate glucuronic acid to a substrate by any of several types of UDP-glucuronosyltransferase. UDP-glucuronic acid (glucuronic acid linked via a glycosidic bond to uridine diphosphate) is an intermediate in the process and is formed in the liver. One example is the N-glucuronidation of an aromatic amine, 4-aminobiphenyl, by UGT1A4 or UGT1A9 from human, rat, or mouse liver. : The substances resulting from glucuronidation are known as glucuronides (or glucuronosides) and are typically much more water-soluble than the non-glucuronic acid-containing substances from which they were originally synthesised. The human body uses glucuronidation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CRC Press
The CRC Press, LLC is an American publishing group that specializes in producing technical books. Many of their books relate to engineering, science and mathematics. Their scope also includes books on business, forensics and information technology. CRC Press is now a division of Taylor & Francis, itself a subsidiary of Informa. History The CRC Press was founded as the Chemical Rubber Company (CRC) in 1903 by brothers Arthur, Leo and Emanuel Friedman in Cleveland, Ohio, based on an earlier enterprise by Arthur, who had begun selling rubber laboratory aprons in 1900. The company gradually expanded to include sales of laboratory equipment to chemists. In 1913 the CRC offered a short (116-page) manual called the ''Rubber Handbook'' as an incentive for any purchase of a dozen aprons. Since then the ''Rubber Handbook'' has evolved into the CRC's flagship book, the '' CRC Handbook of Chemistry and Physics''. In 1964, Chemical Rubber decided to focus on its publishing ventures, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glucuronide
A glucuronide, also known as glucuronoside, is any substance produced by linking glucuronic acid to another substance via a glycosidic bond. The glucuronides belong to the glycosides. Glucuronidation, the conversion of chemical compounds to glucuronides, is a method that animals use to assist in the excretion of toxic substances, drugs or other substances that cannot be used as an energy source. Glucuronic acid is attached via a glycosidic bond to the substance, and the resulting glucuronide, which has a much higher water solubility than the original substance, is eventually excreted by the kidneys. Enzymes that cleave the glycosidic bond of a glucuronide are called glucuronidases. Examples * Miquelianin ( Quercetin 3-O-glucuronide) * Morphine-6-glucuronide * Scutellarein-7-glucuronide Scutellarin is a flavone, a type of phenolic chemical compound. It can be found in the plants '' Scutellaria barbata'' and '' S. lateriflora'' which have been used in traditional medicin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetylation
: In organic chemistry, acetylation is an organic esterification reaction with acetic acid. It introduces an acetyl group into a chemical compound. Such compounds are termed ''acetate esters'' or simply ''acetates''. Deacetylation is the opposite reaction, the removal of an acetyl group from a chemical compound. Organic synthesis Acetate esters and acetamides are generally prepared by acetylations. Acetylations are often used in making C-acetyl bonds in Friedel-Crafts reactions. Carbanions and their equivalents are susceptible to acetylations. Acetylation reagents Many acetylations are achieved using these three reagents: *Acetic anhydride. This reagent is common in the laboratory; its use cogenerates acetic acid. *Acetyl chloride. This reagent is also common in the laboratory, but its use cogenerates hydrogen chloride, which can be undesirable. *Ketene. At one time acetic anhydride was prepared by the reaction of ketene with acetic acid: :H2C=C=O + CH3COOH -> (CH3CO)2O\De ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
N-acetyltransferase 2
N-acetyltransferase 2 (arylamine N-acetyltransferase), also known as NAT2, is an enzyme which in humans is encoded by the ''NAT2'' gene. Function This gene encodes a type of N-acetyltransferase N-acetyltransferase (NAT) is an enzyme that catalyzes the transfer of acetyl groups from acetyl-CoA to arylamines, arylhydroxylamines and arylhydrazines. They have wide specificity for aromatic amines, particularly serotonin, and can also catalyz .... The NAT2 isozyme functions to both activate and deactivate arylamine and hydrazine drugs and carcinogens. Polymorphisms in this gene are responsible for the N-acetylation polymorphism in which human populations segregate into rapid, intermediate, and slow acetylator phenotypes. Polymorphisms in NAT2 are also associated with higher incidences of cancer and drug toxicity. A second arylamine N-acetyltransferase gene ( NAT1) is located near NAT2. Phenotype prediction The NAT2 acetylator phenotype can be inferred from NAT2 genotype ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
N-acetyltransferase 1
N-acetyltransferase 1 (arylamine N-acetyltransferase) is a protein that in humans is encoded by the NAT1 gene. This gene is one of two arylamine N-acetyltransferase (NAT) genes in the human genome, and is orthologous to the mouse and rat NAT2 genes. The enzyme encoded by this gene catalyzes the transfer of an acetyl group from acetyl-CoA to various arylamine and hydrazine substrates. This enzyme helps metabolize drugs and other xenobiotics, and functions in folate catabolism. Multiple transcript variants encoding different isoform A protein isoform, or "protein variant", is a member of a set of highly similar proteins that originate from a single gene or gene family and are the result of genetic differences. While many perform the same or similar biological roles, some is ...s have been found for this gene. rovided by RefSeq, Aug 2011 References Further reading * * * * * * * * * * {{gene-8-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glucuronosyltransferase
Uridine 5'-diphospho-glucuronosyltransferase ( UDP-glucuronosyltransferase, UGT) is a microsomal glycosyltransferase () that catalyzes the transfer of the glucuronic acid component of UDP-glucuronic acid to a small hydrophobic molecule. This is a glucuronidation reaction. ''Alternative names:'' *glucuronyltransferase *UDP-glucuronyl transferase *UDP-GT Function Glucuronosyltransferases are responsible for the process of glucuronidation, a major part of phase II metabolism. Arguably the most important of the Phase II (conjugative) enzymes, UGTs have been the subject of increasing scientific inquiry since the mid-to-late 1990s. The reaction catalyzed by the UGT enzyme involves the addition of a glucuronic acid moiety to xenobiotics and is the most important pathway for the human body's elimination of the most frequently prescribed drugs. It is also the major pathway for foreign chemical (dietary, environmental, pharmaceutical) removal for most drugs, dietary substances, toxins ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
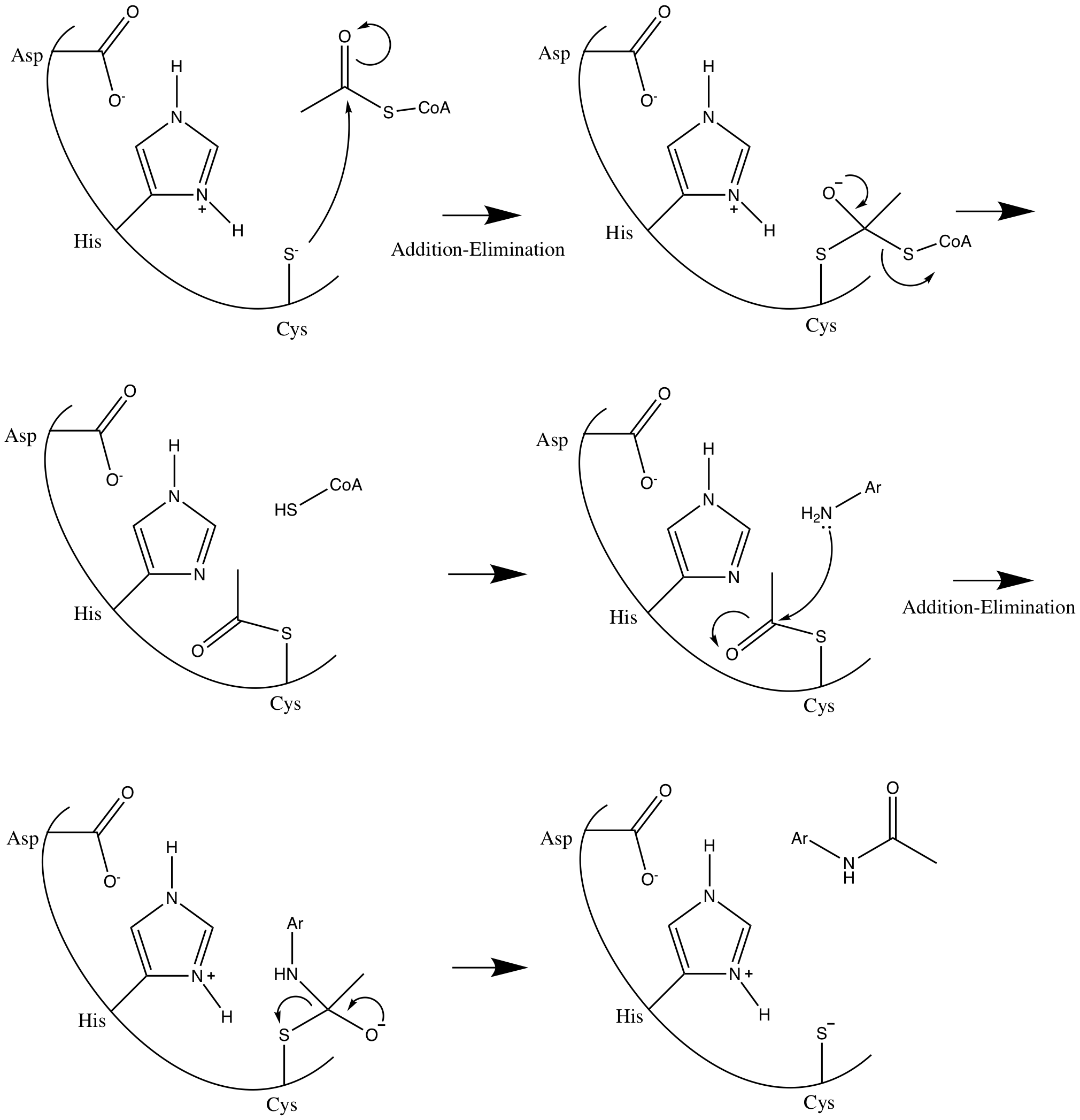
N-acetyltransferase
N-acetyltransferase (NAT) is an enzyme that catalyzes the transfer of acetyl groups from acetyl-CoA to arylamines, arylhydroxylamines and arylhydrazines. They have wide specificity for aromatic amines, particularly serotonin, and can also catalyze acetyl transfer between arylamines without CoA. N-acetyltransferases are cytosolic enzymes found in the liver and many tissues of most mammalian species, except the dog and fox, which cannot acetylate xenobiotics. Acetyl groups are important in the conjugation of metabolites from the liver, to allow excretion of the byproducts ( phase II metabolism). This is especially important in the metabolism and excretion of drug products ( drug metabolism). __TOC__ Enzyme Mechanism NAT enzymes are differentiated by the presence of a conserved catalytic triad that favors aromatic amine and hydrazine substrates. NATs catalyze the acetylation of small molecules through a double displacement reaction called the ping pong bi bi reaction. The mech ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cytochrome P450
Cytochromes P450 (CYPs) are a superfamily of enzymes containing heme as a cofactor that functions as monooxygenases. In mammals, these proteins oxidize steroids, fatty acids, and xenobiotics, and are important for the clearance of various compounds, as well as for hormone synthesis and breakdown. In 1963, Estabrook, Cooper, and Rosenthal described the role of CYP as a catalyst in steroid hormone synthesis and drug metabolism. In plants, these proteins are important for the biosynthesis of defensive compounds, fatty acids, and hormones. CYP enzymes have been identified in all kingdoms of life: animals, plants, fungi, protists, bacteria, and archaea, as well as in viruses. However, they are not omnipresent; for example, they have not been found in ''Escherichia coli''. , more than 300,000 distinct CYP proteins are known. CYPs are, in general, the terminal oxidase enzymes in electron transfer chains, broadly categorized as P450-containing systems. The term "P450" is derived ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetylation Reactions
: In organic chemistry, acetylation is an organic esterification reaction with acetic acid. It introduces an acetyl group into a chemical compound. Such compounds are termed ''acetate esters'' or simply ''acetates''. Deacetylation is the opposite reaction, the removal of an acetyl group from a chemical compound. Organic synthesis Acetate esters and acetamides are generally prepared by acetylations. Acetylations are often used in making C-acetyl bonds in Friedel-Crafts reactions. Carbanions and their equivalents are susceptible to acetylations. Acetylation reagents Many acetylations are achieved using these three reagents: *Acetic anhydride. This reagent is common in the laboratory; its use cogenerates acetic acid. *Acetyl chloride. This reagent is also common in the laboratory, but its use cogenerates hydrogen chloride, which can be undesirable. * Ketene. At one time acetic anhydride was prepared by the reaction of ketene with acetic acid: :H2C=C=O + CH3COOH -> (CH3CO)2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Codons
The genetic code is the set of rules used by living cells to translate information encoded within genetic material ( DNA or RNA sequences of nucleotide triplets, or codons) into proteins. Translation is accomplished by the ribosome, which links proteinogenic amino acids in an order specified by messenger RNA (mRNA), using transfer RNA (tRNA) molecules to carry amino acids and to read the mRNA three nucleotides at a time. The genetic code is highly similar among all organisms and can be expressed in a simple table with 64 entries. The codons specify which amino acid will be added next during protein biosynthesis. With some exceptions, a three-nucleotide codon in a nucleic acid sequence specifies a single amino acid. The vast majority of genes are encoded with a single scheme (see the RNA codon table). That scheme is often referred to as the canonical or standard genetic code, or simply ''the'' genetic code, though variant codes (such as in mitochondria) exist. History Effort ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |