|
13C
Carbon-13 (13C) is a natural, stable isotope of carbon with a nucleus containing six protons and seven neutrons. As one of the environmental isotopes, it makes up about 1.1% of all natural carbon on Earth. Detection by mass spectrometry A mass spectrum of an organic compound will usually contain a small peak of one mass unit greater than the apparent molecular ion peak (M) of the whole molecule. This is known as the M+1 peak and comes from the few molecules that contain a 13C atom in place of a 12C. A molecule containing one carbon atom will be expected to have an M+1 peak of approximately 1.1% of the size of the M peak, as 1.1% of the molecules will have a 13C rather than a 12C. Similarly, a molecule containing two carbon atoms will be expected to have an M+1 peak of approximately 2.2% of the size of the M peak, as there is double the previous likelihood that any molecule will contain a 13C atom. In the above, the mathematics and chemistry have been simplified, however it can ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopic Signature
An isotopic signature (also isotopic fingerprint) is a ratio of non-radiogenic ' stable isotopes', stable radiogenic isotopes, or unstable radioactive isotopes of particular elements in an investigated material. The ratios of isotopes in a sample material are measured by isotope-ratio mass spectrometry against an isotopic reference material. This process is called isotope analysis. Stable isotopes The atomic mass of different isotopes affect their chemical kinetic behavior, leading to natural isotope separation processes. Carbon isotopes For example, different sources and sinks of methane have different affinity for the 12C and 13C isotopes, which allows distinguishing between different sources by the 13C/12C ratio in methane in the air. In geochemistry, paleoclimatology and paleoceanography this ratio is called δ13C. The ratio is calculated with respect to Pee Dee Belemnite (PDB) standard: :\delta \ce_\mathrm = \left(\frac - 1\right) \cdot 1000 ‰ Similarly, carbo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Urea Breath Test
The urea breath test is a rapid diagnostic procedure used to identify infections by ''Helicobacter pylori'', a spiral bacterium implicated in gastritis, gastric ulcer, and peptic ulcer disease. It is based upon the ability of ''H. pylori'' to convert urea to ammonia and carbon dioxide. Urea breath tests are recommended in leading society guidelines as a preferred non-invasive choice for detecting ''H. pylori'' before and after treatment. Principles and mechanism Patients swallow urea labelled with an uncommon isotope, either radioactive carbon-14 or non-radioactive carbon-13. In the subsequent 10–30 minutes, the detection of isotope-labelled carbon dioxide in exhaled breath indicates that the urea was split; this indicates that urease (the enzyme that ''H. pylori'' uses to metabolize urea) is present in the stomach, and hence that ''H. pylori'' bacteria are present. For the two different forms of urea, different instrumentation is required. Carbon-14 is normally measured by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Carbon
Carbon (6C) has 15 known isotopes, from to , of which and are stable. The longest-lived radioisotope is , with a half-life of years. This is also the only carbon radioisotope found in nature—trace quantities are formed cosmogenically by the reaction + → + . The most stable artificial radioisotope is , which has a half-life of . All other radioisotopes have half-lives under 20 seconds, most less than 200 milliseconds. The least stable isotope is , with a half-life of . List of isotopes , - , , style="text-align:right" , 6 , style="text-align:right" , 2 , , [] , proton emission, 2p , Subsequently decays by double proton emission to for a net reaction of → + 4 , 0+ , , , - , rowspan=3, , rowspan=3 style="text-align:right" , 6 , rowspan=3 style="text-align:right" , 3 , rowspan=3, , rowspan=3, , β+ () , , rowspan=3, 3/2− , rowspan=3, , rowspan=3, , - , β+α () , Immediately decays by proton emission to for a net reaction of → ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon-13 Nuclear Magnetic Resonance
Carbon-13 (C13) nuclear magnetic resonance (most commonly known as carbon-13 NMR spectroscopy or 13C NMR spectroscopy or sometimes simply referred to as carbon NMR) is the application of nuclear magnetic resonance (NMR) spectroscopy to carbon. It is analogous to proton NMR ( NMR) and allows the identification of carbon atoms in an organic molecule just as proton NMR identifies hydrogen atoms. 13C NMR detects only the isotope. The main carbon isotope, is not detected. Although much less sensitive than 1H NMR spectroscopy, 13C NMR spectroscopy is widely used for characterizing organic and organometallic compounds. Chemical shifts 13C NMR chemical shifts follow the same principles as those of 1H, although the typical range of chemical shifts is much larger than for 1H (by a factor of about 20). The chemical shift reference standard for 13C is the carbons in tetramethylsilane (TMS), whose chemical shift is considered to be 0.0 ppm. ImageSize = width:540 height:440 AlignBa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stable Isotope Labeling By Amino Acids In Cell Culture
Stable Isotope Labeling by/with Amino acids in Cell culture (SILAC) is a technique based on mass spectrometry that detects differences in protein abundance among samples using non-radioactive isotopic labeling. It is a popular method for quantitative proteomics. Procedure Two populations of cells are cultivated in cell culture. One of the cell populations is fed with growth medium containing normal amino acids. In contrast, the second population is fed with growth medium containing amino acids labeled with stable (non-radioactive) heavy isotopes. For example, the medium can contain arginine labeled with six carbon-13 atoms (13C) instead of the normal carbon-12 (12C). When the cells are growing in this medium, they incorporate the heavy arginine into all of their proteins. Thereafter, all peptides containing a single arginine are 6 Da heavier than their normal counterparts. Alternatively, uniform labeling with 13C or 15N can be used. Proteins from both cell populations are combin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Longevity
The word " longevity" is sometimes used as a synonym for " life expectancy" in demography. However, the term ''longevity'' is sometimes meant to refer only to especially long-lived members of a population, whereas ''life expectancy'' is always defined statistically as the average number of years remaining at a given age. For example, a population's life expectancy at birth is the same as the average age at death for all people born in the same year (in the case of cohorts). Longevity is best thought of as a term for general audiences meaning 'typical length of life' and specific statistical definitions should be clarified when necessary. Reflections on longevity have usually gone beyond acknowledging the brevity of human life and have included thinking about methods to extend life. Longevity has been a topic not only for the scientific community but also for writers of travel, science fiction, and utopian novels. In fact the legendary fountain of youth appeared in the work of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
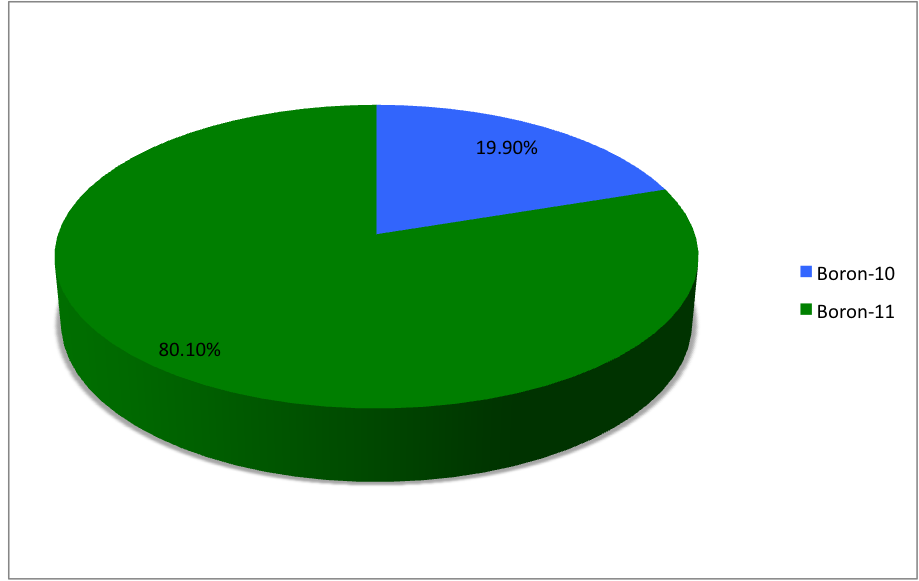
Boron-13
Boron (5B) naturally occurs as isotopes and , the latter of which makes up about 80% of natural boron. There are 13 radioisotopes that have been discovered, with mass numbers from 7 to 21, all with short half-lives, the longest being that of , with a half-life of only and with a half-life of . All other isotopes have half-lives shorter than . Those isotopes with mass below 10 decay into helium (via short-lived isotopes of beryllium for and ) while those with mass above 11 mostly become carbon. List of isotopes , - , ?This isotope has not yet been observed; given data is inferred or estimated from periodic trends. , style="text-align:center" , 5 , style="text-align:center" , 1 , , p-unstable , 2p? , ? , 2−# , , , - , , style="text-align:center" , 5 , style="text-align:center" , 2 , , [] , p , Subsequently decays by double proton emission to for a net reaction of → + 3 , (3/2−) , , , - , Has 1 halo nucleus, halo proton , style="tex ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon-14
Carbon-14, C-14, or radiocarbon, is a radioactive isotope of carbon with an atomic nucleus containing 6 protons and 8 neutrons. Its presence in organic materials is the basis of the radiocarbon dating method pioneered by Willard Libby and colleagues (1949) to date archaeological, geological and hydrogeological samples. Carbon-14 was discovered on February 27, 1940, by Martin Kamen and Sam Ruben at the University of California Radiation Laboratory in Berkeley, California. Its existence had been suggested by Franz Kurie in 1934. There are three naturally occurring isotopes of carbon on Earth: carbon-12 (), which makes up 99% of all carbon on Earth; carbon-13 (), which makes up 1%; and carbon-14 (), which occurs in trace amounts, making up about 1 or 1.5 atoms per 1012 atoms of carbon in the atmosphere. Carbon-12 and carbon-13 are both stable, while carbon-14 is unstable and has a half-life of 5,730 ± 40 years. Carbon-14 decays into nitrogen-14 () through beta decay. A gra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotope Fractionation
Isotope fractionation describes fractionation processes that affect the relative abundance of isotopes, phenomena which are taken advantage of in isotope geochemistry and other fields. Normally, the focus is on stable isotopes of the same element. Isotopic fractionation can be measured by isotope analysis, using isotope-ratio mass spectrometry or cavity ring-down spectroscopy to measure ratios of isotopes, an important tool to understand geochemical and biological systems. For example, biochemical processes cause changes in ratios of stable carbon isotopes incorporated into biomass. Definition Stable isotopes partitioning between two substances ''A'' and ''B'' can be expressed by the use of the isotopic fractionation factor (alpha): where ''R'' is the ratio of the heavy to light isotope (e.g., 2H/1H or 18O/16O). Values for alpha tend to be very close to 1. McGlynn, Shawn E.; "Biological Isotope Fractionation and Earth History: From Enzymes to Cells to Ecosystems" pp 59-79 in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon Monoxide
Carbon monoxide (chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simplest molecule of the oxocarbon family. In coordination complexes the carbon monoxide ligand is called carbonyl. It is a key ingredient in many processes in industrial chemistry. The most common source of carbon monoxide is the partial combustion of carbon-containing compounds, when insufficient oxygen or heat is present to produce carbon dioxide. There are also numerous environmental and biological sources that generate and emit a significant amount of carbon monoxide. It is important in the production of many compounds, including drugs, fragrances, and fuels. Upon emission into the atmosphere, carbon monoxide affects several processes that contribute to climate change. Carbon monoxide has important biological roles across phylogenetic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane on Earth makes it an economically attractive fuel, although capturing and storing it poses technical challenges due to its gaseous state under normal conditions for temperature and pressure. Naturally occurring methane is found both below ground and under the seafloor and is formed by both geological and biological processes. The largest reservoir of methane is under the seafloor in the form of methane clathrates. When methane reaches the surface and the atmosphere, it is known as atmospheric methane. The Earth's atmospheric methane concentration has increased by about 150% since 1750, and it accounts for 20% of the total radiative forcing from all of the long-lived and globally mixed greenhouse gases. It has also been detected on other ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helicobacter Pylori
''Helicobacter pylori'', previously known as ''Campylobacter pylori'', is a gram-negative, microaerophilic, spiral (helical) bacterium usually found in the stomach. Its helical shape (from which the genus name, helicobacter, derives) is thought to have evolved in order to penetrate the mucoid lining of the stomach and thereby establish infection. The bacterium was first identified in 1982 by the Australian doctors Barry Marshall and Robin Warren. ''H. pylori'' has been associated with cancer of the mucosa-associated lymphoid tissue in the stomach, esophagus, colon, rectum, or tissues around the eye (termed extranodal marginal zone B-cell lymphoma of the cited organ), and of lymphoid tissue in the stomach (termed diffuse large B-cell lymphoma). ''H. pylori'' infection usually has no symptoms but sometimes causes gastritis (stomach inflammation) or ulcers of the stomach or first part of the small intestine. The infection is also associated with the development of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |