Sakurai Reaction on:
[Wikipedia]
[Google]
[Amazon]
The Sakurai reaction (also known as the Hosomi–Sakurai reaction) is the  Lewis acid activation is essential for complete reaction. Strong Lewis acids such as
Lewis acid activation is essential for complete reaction. Strong Lewis acids such as
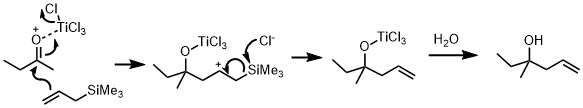
 The Hosomi-Sakurai reactions are allylation reactions which involve use of allyl silanes as allylmetal reagents. This section demonstrates examples of allylation of diverse ketones. In figure 1, allylation of a carbonyl ketone (compound containing a ketone group and two different functional groups) has been shown. In the given reaction, the electrophilic compound (carbon with a ketone group) is treated with titanium tetrachloride, a strong Lewis acid and allyltrimethylsilane. According to the general principle, the Lewis acid first activates the electrophilic carbon in presence of allyltrimethylsilane which then undergoes nucleophilic attack from electrons on the allylic silane. The silicon plays the key role in stabilizing the carbocation of carbon at the β-position. Hosomi-Sakurai reaction is also applicable for other functional groups such as enones, where conjugate addition is usually seen. In figure 2, the Hosomi- Sakurai reaction has been shown using a cinnamoyl ketone. This reaction follows the same mechanism as the previous reaction shown here.
The Hosomi-Sakurai reactions are allylation reactions which involve use of allyl silanes as allylmetal reagents. This section demonstrates examples of allylation of diverse ketones. In figure 1, allylation of a carbonyl ketone (compound containing a ketone group and two different functional groups) has been shown. In the given reaction, the electrophilic compound (carbon with a ketone group) is treated with titanium tetrachloride, a strong Lewis acid and allyltrimethylsilane. According to the general principle, the Lewis acid first activates the electrophilic carbon in presence of allyltrimethylsilane which then undergoes nucleophilic attack from electrons on the allylic silane. The silicon plays the key role in stabilizing the carbocation of carbon at the β-position. Hosomi-Sakurai reaction is also applicable for other functional groups such as enones, where conjugate addition is usually seen. In figure 2, the Hosomi- Sakurai reaction has been shown using a cinnamoyl ketone. This reaction follows the same mechanism as the previous reaction shown here.
Link
Akira Hosomi HP
Addition reactions Carbon-carbon bond forming reactions Name reactions
chemical reaction
A chemical reaction is a process that leads to the chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and break ...
of carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes ...
electrophile
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that ca ...
s (such as a ketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bon ...
shown here) with allyltrimethylsilane
Allyltrimethylsilane is the organosilicon compound with the formula (CH3)3SiCH2CH=CH2. The molecule consists of the trimethylsilyl group attached to allyl group. This colorless liquid is used in organic synthesis
Organic synthesis is a spe ...
catalyzed by strong Lewis acid
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any sp ...
s.
 Lewis acid activation is essential for complete reaction. Strong Lewis acids such as
Lewis acid activation is essential for complete reaction. Strong Lewis acids such as titanium tetrachloride
Titanium tetrachloride is the inorganic compound with the formula . It is an important intermediate in the production of titanium metal and the pigment titanium dioxide. is a volatile liquid. Upon contact with humid air, it forms thick clouds ...
, boron trifluoride
Boron trifluoride is the inorganic compound with the formula BF3. This pungent, colourless, and toxic gas forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds.
Structure and bondin ...
, tin tetrachloride
Tin(IV) chloride, also known as tin tetrachloride or stannic chloride, is an inorganic compound with the formula Sn Cl4. It is a colorless hygroscopic liquid, which fumes on contact with air. It is used as a precursor to other tin compounds. It w ...
, and AlCl(Et)2 are all effective in promoting the Hosomi reaction. The reaction is a type of electrophilic allyl shift with formation of an intermediate beta-silyl carbocation. Driving force is the stabilization of said carbocation by the beta-silicon effect
The beta-silicon effect also called silicon hyperconjugation in organosilicon chemistry is a special type of hyperconjugation that describes the stabilizing influence of a silicon atom on the development of positive charge at a carbon atom one po ...
.
The Hosomi-Sakurai reaction can be performed on a number of functional groups. An electrophilic carbon, activated by a Lewis acid, is required. Below is a list of different functional groups that can be used in the Hosomi–Sakurai reaction. The reaction achieves results similar to the addition of an allyl Grignard reagent to the carbonyl.
Mechanism

 The Hosomi-Sakurai reactions are allylation reactions which involve use of allyl silanes as allylmetal reagents. This section demonstrates examples of allylation of diverse ketones. In figure 1, allylation of a carbonyl ketone (compound containing a ketone group and two different functional groups) has been shown. In the given reaction, the electrophilic compound (carbon with a ketone group) is treated with titanium tetrachloride, a strong Lewis acid and allyltrimethylsilane. According to the general principle, the Lewis acid first activates the electrophilic carbon in presence of allyltrimethylsilane which then undergoes nucleophilic attack from electrons on the allylic silane. The silicon plays the key role in stabilizing the carbocation of carbon at the β-position. Hosomi-Sakurai reaction is also applicable for other functional groups such as enones, where conjugate addition is usually seen. In figure 2, the Hosomi- Sakurai reaction has been shown using a cinnamoyl ketone. This reaction follows the same mechanism as the previous reaction shown here.
The Hosomi-Sakurai reactions are allylation reactions which involve use of allyl silanes as allylmetal reagents. This section demonstrates examples of allylation of diverse ketones. In figure 1, allylation of a carbonyl ketone (compound containing a ketone group and two different functional groups) has been shown. In the given reaction, the electrophilic compound (carbon with a ketone group) is treated with titanium tetrachloride, a strong Lewis acid and allyltrimethylsilane. According to the general principle, the Lewis acid first activates the electrophilic carbon in presence of allyltrimethylsilane which then undergoes nucleophilic attack from electrons on the allylic silane. The silicon plays the key role in stabilizing the carbocation of carbon at the β-position. Hosomi-Sakurai reaction is also applicable for other functional groups such as enones, where conjugate addition is usually seen. In figure 2, the Hosomi- Sakurai reaction has been shown using a cinnamoyl ketone. This reaction follows the same mechanism as the previous reaction shown here.
Beta-silicon effec] stabilization
As displayed in the mechanism, the Hosomi–Sakurai reaction goes through a secondary carbocation intermediate. Secondary carbocations are inherently unstable, however the β-silicon effect from the silicon atom stabilizes the carbocation. Silicon is able to donate into an empty p-orbital, and the silicon orbital is shared between the two carbons. This stabilizes the positive charge over 3 orbitals. Another term for the β-silicon effect is silicon-hyperconjugation. This interaction is essential for the reaction to go to completion.Literature of historic interest
* * *( *References
{{reflistExternal links
* Hosomi-Sakurai reaction @ www.organic-chemistry.orLink
Akira Hosomi HP
Addition reactions Carbon-carbon bond forming reactions Name reactions