Rubidium on:
[Wikipedia]
[Google]
[Amazon]
Rubidium is a
 Rubidium is a very soft,
Rubidium is a very soft,

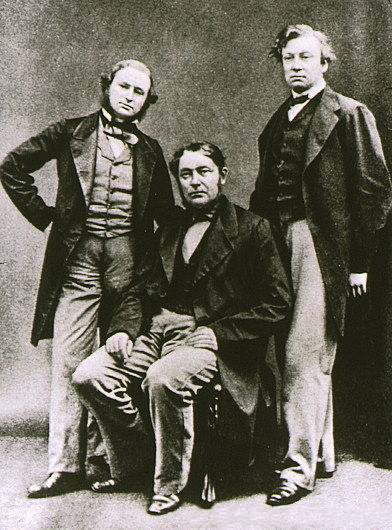 Rubidium was discovered in 1861 by
Rubidium was discovered in 1861 by
 Rubidium compounds are sometimes used in
Rubidium compounds are sometimes used in
Rubidium
at ''
chemical element
A chemical element is a chemical substance whose atoms all have the same number of protons. The number of protons is called the atomic number of that element. For example, oxygen has an atomic number of 8: each oxygen atom has 8 protons in its ...
; it has symbol
A symbol is a mark, Sign (semiotics), sign, or word that indicates, signifies, or is understood as representing an idea, physical object, object, or wikt:relationship, relationship. Symbols allow people to go beyond what is known or seen by cr ...
Rb and atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of its atomic nucleus. For ordinary nuclei composed of protons and neutrons, this is equal to the proton number (''n''p) or the number of pro ...
37. It is a very soft, whitish-grey solid in the alkali metal
The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K),The symbols Na and K for sodium and potassium are derived from their Latin names, ''natrium'' and ''kalium''; these are still the origins of the names ...
group, similar to potassium
Potassium is a chemical element; it has Symbol (chemistry), symbol K (from Neo-Latin ) and atomic number19. It is a silvery white metal that is soft enough to easily cut with a knife. Potassium metal reacts rapidly with atmospheric oxygen to ...
and caesium
Caesium (IUPAC spelling; also spelled cesium in American English) is a chemical element; it has Symbol (chemistry), symbol Cs and atomic number 55. It is a soft, silvery-golden alkali metal with a melting point of , which makes it one of only f ...
. Rubidium is the first alkali metal
The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K),The symbols Na and K for sodium and potassium are derived from their Latin names, ''natrium'' and ''kalium''; these are still the origins of the names ...
in the group to have a density higher than water
Water is an inorganic compound with the chemical formula . It is a transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance. It is the main constituent of Earth's hydrosphere and the fluids of all known liv ...
. On Earth, natural rubidium comprises two isotope
Isotopes are distinct nuclear species (or ''nuclides'') of the same chemical element. They have the same atomic number (number of protons in their Atomic nucleus, nuclei) and position in the periodic table (and hence belong to the same chemica ...
s: 72% is a stable isotope Rb, and 28% is slightly radioactive
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is conside ...
Rb, with a half-life Half-life is a mathematical and scientific description of exponential or gradual decay.
Half-life, half life or halflife may also refer to:
Film
* Half-Life (film), ''Half-Life'' (film), a 2008 independent film by Jennifer Phang
* ''Half Life: ...
of 48.8 billion years – more than three times as long as the estimated age of the universe
In physical cosmology, the age of the universe is the cosmological time, time elapsed since the Big Bang: 13.79 billion years.
Astronomers have two different approaches to determine the age of the universe. One is based on a particle physics ...
.
German chemists Robert Bunsen
Robert Wilhelm Eberhard Bunsen (;
30 March 1811
– 16 August 1899) was a German chemist. He investigated emission spectra of heated elements, and discovered caesium (in 1860) and rubidium (in 1861) with the physicist Gustav Kirchhoff. The Bu ...
and Gustav Kirchhoff
Gustav Robert Kirchhoff (; 12 March 1824 – 17 October 1887) was a German chemist, mathematician, physicist, and spectroscopist who contributed to the fundamental understanding of electrical circuits, spectroscopy and the emission of black-body ...
discovered rubidium in 1861 by the newly developed technique, flame spectroscopy. The name comes from the Latin
Latin ( or ) is a classical language belonging to the Italic languages, Italic branch of the Indo-European languages. Latin was originally spoken by the Latins (Italic tribe), Latins in Latium (now known as Lazio), the lower Tiber area aroun ...
word , meaning deep red, the color of its emission spectrum. Rubidium's compounds have various chemical and electronic applications. Rubidium metal is easily vaporized and has a convenient spectral absorption range, making it a frequent target for laser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word ''laser'' originated as an acronym for light amplification by stimulated emission of radi ...
manipulation of atom
Atoms are the basic particles of the chemical elements. An atom consists of a atomic nucleus, nucleus of protons and generally neutrons, surrounded by an electromagnetically bound swarm of electrons. The chemical elements are distinguished fr ...
s. Rubidium is not a known nutrient for any living organisms
An organism is any living thing that functions as an individual. Such a definition raises more problems than it solves, not least because the concept of an individual is also difficult. Many criteria, few of them widely accepted, have been pro ...
. However, rubidium ion
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convent ...
s have similar properties and the same charge as potassium ions, and are actively taken up and treated by animal cell
The cell is the basic structural and functional unit of all life, forms of life. Every cell consists of cytoplasm enclosed within a Cell membrane, membrane; many cells contain organelles, each with a specific function. The term comes from the ...
s in similar ways.
Characteristics
Physical properties
ductile
Ductility refers to the ability of a material to sustain significant plastic deformation before fracture. Plastic deformation is the permanent distortion of a material under applied stress, as opposed to elastic deformation, which is reversi ...
, silvery-white metal. It has a melting point of and a boiling point of . It forms amalgams
Amalgam most commonly refers to:
* Amalgam (chemistry), mercury alloy
* Amalgam (dentistry), material of silver tooth fillings
** Bonded amalgam, used in dentistry
Amalgam may also refer to:
* Amalgam Comics, a publisher
* Amalgam Digital, an in ...
with mercury and alloy
An alloy is a mixture of chemical elements of which in most cases at least one is a metal, metallic element, although it is also sometimes used for mixtures of elements; herein only metallic alloys are described. Metallic alloys often have prop ...
s with gold
Gold is a chemical element; it has chemical symbol Au (from Latin ) and atomic number 79. In its pure form, it is a brightness, bright, slightly orange-yellow, dense, soft, malleable, and ductile metal. Chemically, gold is a transition metal ...
, iron
Iron is a chemical element; it has symbol Fe () and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, forming much of Earth's o ...
, caesium
Caesium (IUPAC spelling; also spelled cesium in American English) is a chemical element; it has Symbol (chemistry), symbol Cs and atomic number 55. It is a soft, silvery-golden alkali metal with a melting point of , which makes it one of only f ...
, sodium
Sodium is a chemical element; it has Symbol (chemistry), symbol Na (from Neo-Latin ) and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 element, group 1 of the peri ...
, and potassium
Potassium is a chemical element; it has Symbol (chemistry), symbol K (from Neo-Latin ) and atomic number19. It is a silvery white metal that is soft enough to easily cut with a knife. Potassium metal reacts rapidly with atmospheric oxygen to ...
, but not lithium
Lithium (from , , ) is a chemical element; it has chemical symbol, symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard temperature and pressure, standard conditions, it is the least dense metal and the ...
(despite rubidium and lithium being in the same periodic group). Rubidium and potassium show a very similar purple color in the flame test
A flame test is relatively quick test for the presence of some elements in a sample. The technique is archaic and of questionable reliability, but once was a component of qualitative inorganic analysis. The phenomenon is related to pyrotechnics ...
, and distinguishing the two elements requires more sophisticated analysis, such as spectroscopy.
Chemical properties
Rubidium is the second mostelectropositive
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the d ...
of the stable alkali metals and has a very low first ionization energy
In physics and chemistry, ionization energy (IE) is the minimum energy required to remove the most loosely bound electron of an isolated gaseous atom, Ion, positive ion, or molecule. The first ionization energy is quantitatively expressed as
: ...
of only 403 kJ/mol. It has an electron configuration of rs1 and is photosensitive. Due to its strong electropositive nature, rubidium reacts explosively with water to produce rubidium hydroxide and hydrogen gas. As with all the alkali metals, the reaction is usually vigorous enough to ignite metal or the hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter ...
gas produced by the reaction, potentially causing an explosion. Rubidium, being denser than potassium, sinks in water, reacting violently; caesium explodes on contact with water. However, the reaction rates of all alkali metals depend upon surface area of metal in contact with water, with small metal droplets giving explosive rates. Rubidium has also been reported to ignite spontaneously in air.
Compounds

Rubidium chloride
Rubidium chloride is the chemical compound with the formula RbCl. This alkali metal halide salt is composed of rubidium and chlorine, and finds diverse uses ranging from electrochemistry to molecular biology.
Structure
In its gas phase, RbCl is d ...
(RbCl) is probably the most used rubidium compound: among several other chlorides, it is used to induce living cells to take up DNA
Deoxyribonucleic acid (; DNA) is a polymer composed of two polynucleotide chains that coil around each other to form a double helix. The polymer carries genetic instructions for the development, functioning, growth and reproduction of al ...
; it is also used as a biomarker, because in nature, it is found only in small quantities in living organisms and when present, replaces potassium. Other common rubidium compounds are the corrosive rubidium hydroxide (RbOH), the starting material for most rubidium-based chemical processes; rubidium carbonate (Rb2CO3), used in some optical glasses, and rubidium copper sulfate, Rb2SO4·CuSO4·6H2O. Rubidium silver iodide (RbAg4I5) has the highest room temperature
Room temperature, colloquially, denotes the range of air temperatures most people find comfortable indoors while dressed in typical clothing. Comfortable temperatures can be extended beyond this range depending on humidity, air circulation, and ...
conductivity of any known ionic crystal, a property exploited in thin film batteries and other applications.
Rubidium forms a number of oxides
An oxide () is a chemical compound containing at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion (anion bearing a net charge of −2) of oxygen, an O2− ion with oxygen in the oxidation state o ...
when exposed to air, including rubidium monoxide (Rb2O), Rb6O, and Rb9O2; rubidium in excess oxygen gives the superoxide
In chemistry, a superoxide is a compound that contains the superoxide ion, which has the chemical formula . The systematic name of the anion is dioxide(1−). The reactive oxygen ion superoxide is particularly important as the product of t ...
RbO2. Rubidium forms salts with halogens, producing rubidium fluoride
Rubidium fluoride (RbF) is the fluoride salt of rubidium. It is a cubic crystal with rock-salt structure.
Synthesis
There are several methods for synthesising rubidium fluoride. One involves reacting rubidium hydroxide with hydrofluoric acid: ...
, rubidium chloride
Rubidium chloride is the chemical compound with the formula RbCl. This alkali metal halide salt is composed of rubidium and chlorine, and finds diverse uses ranging from electrochemistry to molecular biology.
Structure
In its gas phase, RbCl is d ...
, rubidium bromide, and rubidium iodide.
Isotopes
Rubidium in the Earth's crust is composed of two isotopes: the stable 85Rb (72.2%) and theradioactive
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is conside ...
87Rb (27.8%). Natural rubidium is radioactive, with specific activity of about 670 Bq/g, enough to significantly expose a photographic film
Photographic film is a strip or sheet of transparent film base coated on one side with a gelatin photographic emulsion, emulsion containing microscopically small light-sensitive silver halide crystals. The sizes and other characteristics of the ...
in 110 days. Thirty additional rubidium isotopes have been synthesized with half-lives of less than 3 months; most are highly radioactive and have few uses.
Rubidium-87 has a half-life Half-life is a mathematical and scientific description of exponential or gradual decay.
Half-life, half life or halflife may also refer to:
Film
* Half-Life (film), ''Half-Life'' (film), a 2008 independent film by Jennifer Phang
* ''Half Life: ...
of years, which is more than three times the age of the universe
In physical cosmology, the age of the universe is the cosmological time, time elapsed since the Big Bang: 13.79 billion years.
Astronomers have two different approaches to determine the age of the universe. One is based on a particle physics ...
of years, making it a primordial nuclide
In geochemistry, geophysics and nuclear physics, primordial nuclides, also known as primordial isotopes, are nuclides found on Earth that have existed in their current form since before Earth was formed. Primordial nuclides were present in the ...
. It readily substitutes for potassium
Potassium is a chemical element; it has Symbol (chemistry), symbol K (from Neo-Latin ) and atomic number19. It is a silvery white metal that is soft enough to easily cut with a knife. Potassium metal reacts rapidly with atmospheric oxygen to ...
in mineral
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed. (2011): Mi ...
s, and is therefore fairly widespread. Rb has been used extensively in dating rocks; 87Rb beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which an atomic nucleus emits a beta particle (fast energetic electron or positron), transforming into an isobar of that nuclide. For example, beta decay of a neutron ...
s to stable 87Sr. During fractional crystallization, Sr tends to concentrate in plagioclase
Plagioclase ( ) is a series of Silicate minerals#Tectosilicates, tectosilicate (framework silicate) minerals within the feldspar group. Rather than referring to a particular mineral with a specific chemical composition, plagioclase is a continu ...
, leaving Rb in the liquid phase. Hence, the Rb/Sr ratio in residual magma
Magma () is the molten or semi-molten natural material from which all igneous rocks are formed. Magma (sometimes colloquially but incorrectly referred to as ''lava'') is found beneath the surface of the Earth, and evidence of magmatism has also ...
may increase over time, and the progressing differentiation results in rocks with elevated Rb/Sr ratios. The highest ratios (10 or more) occur in pegmatite
A pegmatite is an igneous rock showing a very coarse texture, with large interlocking crystals usually greater in size than and sometimes greater than . Most pegmatites are composed of quartz, feldspar, and mica, having a similar silicic c ...
s. If the initial amount of Sr is known or can be extrapolated, then the age can be determined by measurement of the Rb and Sr concentrations and of the 87Sr/86Sr ratio. The dates indicate the true age of the minerals only if the rocks have not been subsequently altered (see rubidium–strontium dating
The rubidium–strontium dating method (Rb–Sr) is a radiometric dating technique, used by scientists to determine the age of rocks and minerals from their content of specific isotopes of rubidium (87Rb) and strontium (87Sr, 86Sr). One of the t ...
).
Rubidium-82
Rubidium-82 (82Rb) is a radioactive isotope of rubidium. 82Rb is widely used in myocardial perfusion imaging. This isotope undergoes rapid uptake by myocardiocytes, which makes it a valuable tool for identifying myocardial ischemia in Positron Em ...
, one of the element's non-natural isotopes, is produced by electron-capture decay of strontium-82 with a half-life of 25.36 days. With a half-life of 76 seconds, rubidium-82 decays by positron emission to stable krypton-82.
Occurrence
Rubidium is not abundant, being one of 56 elements that combined make up 0.05% of the Earth's crust; at roughly the 23rd most abundant element in the Earth's crust it is more abundant thanzinc
Zinc is a chemical element; it has symbol Zn and atomic number 30. It is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodic tabl ...
or copper
Copper is a chemical element; it has symbol Cu (from Latin ) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orang ...
. It occurs naturally in the minerals leucite
Leucite (from the Greek word ''leukos'' meaning white) is a rock-forming mineral of the feldspathoid group, silica-undersaturated and composed of potassium and aluminium tectosilicate KAlSi2O6. Crystals have the form of cubic icositetrahedra b ...
, pollucite
Pollucite is a zeolite mineral with the formula with iron, calcium, rubidium and potassium as common substituting elements. It is important as a significant ore of caesium and sometimes rubidium. It forms a solid solution series with analcime. I ...
, carnallite
Carnallite (also carnalite) is an evaporite mineral, a hydrated potassium magnesium chloride with formula KCl.MgCl2·6(H2O). It is variably colored yellow to white, reddish, and sometimes colorless or blue. It is usually massive to fibrous with r ...
, and zinnwaldite, which contain as much as 1% rubidium oxide
An oxide () is a chemical compound containing at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion (anion bearing a net charge of −2) of oxygen, an O2− ion with oxygen in the oxidation st ...
. Lepidolite
Lepidolite is the common name for a lilac-gray or rose-colored series of minerals in the mica group. The mineralogical name for this series is the polylithionite-trilithionite series. Lepidolite has a chemical formula of . It is the most abundan ...
contains between 0.3% and 3.5% rubidium, and is the commercial source of the element. Some potassium
Potassium is a chemical element; it has Symbol (chemistry), symbol K (from Neo-Latin ) and atomic number19. It is a silvery white metal that is soft enough to easily cut with a knife. Potassium metal reacts rapidly with atmospheric oxygen to ...
minerals and potassium chloride
Potassium chloride (KCl, or potassium salt) is a metal halide salt composed of potassium and chlorine. It is odorless and has a white or colorless vitreous crystal appearance. The solid dissolves readily in water, and its solutions have a sa ...
s also contain the element in commercially significant quantities.
Seawater
Seawater, or sea water, is water from a sea or ocean. On average, seawater in the world's oceans has a salinity of about 3.5% (35 g/L, 35 ppt, 600 mM). This means that every kilogram (roughly one liter by volume) of seawater has approximat ...
contains an average of 125 μg/L of rubidium compared to the much higher value for potassium of 408 mg/L and the much lower value of 0.3 μg/L for caesium. Rubidium is the 18th most abundant element in seawater.
Because of its large ionic radius
Ionic radius, ''r''ion, is the radius of a monatomic ion in an ionic crystal structure. Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cati ...
, rubidium is one of the "incompatible element
In petrology and geochemistry, an incompatible element is one that is unsuitable in size and/or charge to the cation sites of the minerals in which it is included. It is defined by a partition coefficient between rock-forming minerals and melt ...
s". During magma crystallization, rubidium is concentrated together with its heavier analogue caesium in the liquid phase and crystallizes last. Therefore, the largest deposits of rubidium and caesium are zone pegmatite
A pegmatite is an igneous rock showing a very coarse texture, with large interlocking crystals usually greater in size than and sometimes greater than . Most pegmatites are composed of quartz, feldspar, and mica, having a similar silicic c ...
ore bodies formed by this enrichment process. Because rubidium substitutes for potassium
Potassium is a chemical element; it has Symbol (chemistry), symbol K (from Neo-Latin ) and atomic number19. It is a silvery white metal that is soft enough to easily cut with a knife. Potassium metal reacts rapidly with atmospheric oxygen to ...
in the crystallization of magma, the enrichment is far less effective than that of caesium. Zone pegmatite ore bodies containing mineable quantities of caesium as pollucite
Pollucite is a zeolite mineral with the formula with iron, calcium, rubidium and potassium as common substituting elements. It is important as a significant ore of caesium and sometimes rubidium. It forms a solid solution series with analcime. I ...
or the lithium minerals lepidolite
Lepidolite is the common name for a lilac-gray or rose-colored series of minerals in the mica group. The mineralogical name for this series is the polylithionite-trilithionite series. Lepidolite has a chemical formula of . It is the most abundan ...
are also a source for rubidium as a by-product.
Two notable sources of rubidium are the rich deposits of pollucite
Pollucite is a zeolite mineral with the formula with iron, calcium, rubidium and potassium as common substituting elements. It is important as a significant ore of caesium and sometimes rubidium. It forms a solid solution series with analcime. I ...
at Bernic Lake
Bernic Lake is a lake in the eastern part of the province of Manitoba, Canada. It is located just southwest of Nopiming Provincial Park, and just north of Whiteshell Provincial Park.
The Tanco mine is located on the northwestern shore of the ri ...
, Manitoba
Manitoba is a Provinces and territories of Canada, province of Canada at the Centre of Canada, longitudinal centre of the country. It is Canada's Population of Canada by province and territory, fifth-most populous province, with a population ...
, Canada, and the rubicline found as impurities in pollucite on the Italian island of Elba
Elba (, ; ) is a Mediterranean Sea, Mediterranean island in Tuscany, Italy, from the coastal town of Piombino on the Italian mainland, and the largest island of the Tuscan Archipelago. It is also part of the Arcipelago Toscano National Park, a ...
, with a rubidium content of 17.5%. Both of those deposits are also sources of caesium.
Production
Although rubidium is more abundant in Earth's crust than caesium, the limited applications and the lack of a mineral rich in rubidium limits the production of rubidium compounds to 2 to 4tonne
The tonne ( or ; symbol: t) is a unit of mass equal to 1,000 kilograms. It is a non-SI unit accepted for use with SI. It is also referred to as a metric ton in the United States to distinguish it from the non-metric units of the s ...
s per year. Several methods are available for separating potassium, rubidium, and caesium. The fractional crystallization of a rubidium and caesium alum yields after 30 subsequent steps pure rubidium alum. Two other methods are reported, the chlorostannate process and the ferrocyanide process.
For several years in the 1950s and 1960s, a by-product of potassium production called Alkarb was a main source for rubidium. Alkarb contained 21% rubidium, with the rest being potassium and a small amount of caesium. Today the largest producers of caesium produce rubidium as a by-product from pollucite.
History
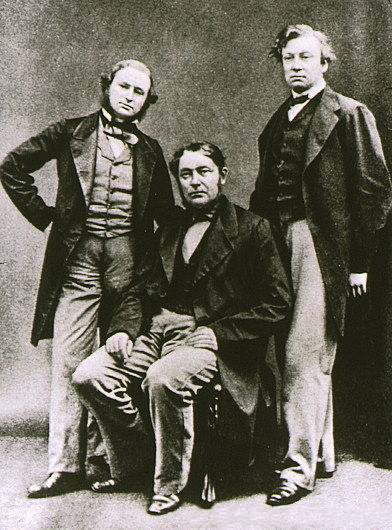 Rubidium was discovered in 1861 by
Rubidium was discovered in 1861 by Robert Bunsen
Robert Wilhelm Eberhard Bunsen (;
30 March 1811
– 16 August 1899) was a German chemist. He investigated emission spectra of heated elements, and discovered caesium (in 1860) and rubidium (in 1861) with the physicist Gustav Kirchhoff. The Bu ...
and Gustav Kirchhoff
Gustav Robert Kirchhoff (; 12 March 1824 – 17 October 1887) was a German chemist, mathematician, physicist, and spectroscopist who contributed to the fundamental understanding of electrical circuits, spectroscopy and the emission of black-body ...
, in Heidelberg, Germany, in the mineral lepidolite
Lepidolite is the common name for a lilac-gray or rose-colored series of minerals in the mica group. The mineralogical name for this series is the polylithionite-trilithionite series. Lepidolite has a chemical formula of . It is the most abundan ...
through flame spectroscopy. Because of the bright red lines in its emission spectrum
The emission spectrum of a chemical element or chemical compound is the Spectrum (physical sciences), spectrum of frequencies of electromagnetic radiation emitted due to electrons making a atomic electron transition, transition from a high energ ...
, they chose a name derived from the Latin
Latin ( or ) is a classical language belonging to the Italic languages, Italic branch of the Indo-European languages. Latin was originally spoken by the Latins (Italic tribe), Latins in Latium (now known as Lazio), the lower Tiber area aroun ...
word , meaning "deep red".
Rubidium is a minor component in lepidolite
Lepidolite is the common name for a lilac-gray or rose-colored series of minerals in the mica group. The mineralogical name for this series is the polylithionite-trilithionite series. Lepidolite has a chemical formula of . It is the most abundan ...
. Kirchhoff and Bunsen processed 150 kg of a lepidolite containing only 0.24% rubidium monoxide (Rb2O). Both potassium and rubidium form insoluble salts with chloroplatinic acid, but those salts show a slight difference in solubility in hot water. Therefore, the less soluble rubidium hexachloroplatinate (Rb2PtCl6) could be obtained by fractional crystallization. After reduction of the hexachloroplatinate with hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter ...
, the process yielded 0.51 grams of rubidium chloride
Rubidium chloride is the chemical compound with the formula RbCl. This alkali metal halide salt is composed of rubidium and chlorine, and finds diverse uses ranging from electrochemistry to molecular biology.
Structure
In its gas phase, RbCl is d ...
(RbCl) for further studies. Bunsen and Kirchhoff began their first large-scale isolation of caesium and rubidium compounds with of mineral water, which yielded 7.3 grams of caesium chloride
Caesium chloride or cesium chloride is the inorganic compound with the formula Caesium, CsChloride, Cl. This colorless salt is an important source of caesium ions in a variety of niche applications. Its crystal structure forms a major structural ...
and 9.2 grams of rubidium chloride
Rubidium chloride is the chemical compound with the formula RbCl. This alkali metal halide salt is composed of rubidium and chlorine, and finds diverse uses ranging from electrochemistry to molecular biology.
Structure
In its gas phase, RbCl is d ...
. Rubidium was the second element, shortly after caesium, to be discovered by spectroscopy, just one year after the invention of the spectroscope
An optical spectrometer (spectrophotometer, spectrograph or spectroscope) is an instrument used to measure properties of light over a specific portion of the electromagnetic spectrum, typically used in spectroscopic analysis to identify mate ...
by Bunsen and Kirchhoff.
The two scientists used the rubidium chloride to estimate that the atomic weight
Relative atomic mass (symbol: ''A''; sometimes abbreviated RAM or r.a.m.), also known by the deprecated synonym atomic weight, is a dimensionless physical quantity defined as the ratio of the average mass of atoms of a chemical element in a giv ...
of the new element was 85.36 (the currently accepted value is 85.47). They tried to generate elemental rubidium by electrolysis of molten rubidium chloride, but instead of a metal, they obtained a blue homogeneous substance, which "neither under the naked eye nor under the microscope showed the slightest trace of metallic substance". They presumed that it was a subchloride (); however, the product was probably a colloid
A colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout another substance. Some definitions specify that the particles must be dispersed in a liquid, while others exte ...
al mixture of the metal and rubidium chloride. In a second attempt to produce metallic rubidium, Bunsen was able to reduce rubidium by heating charred rubidium tartrate. Although the distilled rubidium was pyrophoric
A substance is pyrophoric (from , , 'fire-bearing') if it ignites spontaneously in air at or below (for gases) or within 5 minutes after coming into contact with air (for liquids and solids). Examples are organolithium compounds and triethylb ...
, they were able to determine the density and the melting point. The quality of this research in the 1860s can be appraised by the fact that their determined density differs by less than 0.1 g/cm3 and the melting point by less than 1 °C from the presently accepted values.
The slight radioactivity of rubidium was discovered in 1908, but that was before the theory of isotopes was established in 1910, and the low level of activity (half-life greater than 1010 years) made interpretation complicated. The now proven decay of 87Rb to stable 87Sr through beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which an atomic nucleus emits a beta particle (fast energetic electron or positron), transforming into an isobar of that nuclide. For example, beta decay of a neutron ...
was still under discussion in the late 1940s.
Rubidium had minimal industrial value before the 1920s. Since then, the most important use of rubidium is research and development, primarily in chemical and electronic applications. In 1995, rubidium-87 was used to produce a Bose–Einstein condensate
In condensed matter physics, a Bose–Einstein condensate (BEC) is a state of matter that is typically formed when a gas of bosons at very low Density, densities is cooled to temperatures very close to absolute zero#Relation with Bose–Einste ...
, for which the discoverers, Eric Allin Cornell, Carl Edwin Wieman and Wolfgang Ketterle
Wolfgang Ketterle (; born 21 October 1957) is a German physicist and professor of physics at the Massachusetts Institute of Technology (MIT). His research has focused on experiments that trap and cool atoms to temperatures close to absolute zer ...
, won the 2001 Nobel Prize in Physics
The Nobel Prize in Physics () is an annual award given by the Royal Swedish Academy of Sciences for those who have made the most outstanding contributions to mankind in the field of physics. It is one of the five Nobel Prizes established by the ...
.
Applications
 Rubidium compounds are sometimes used in
Rubidium compounds are sometimes used in fireworks
Fireworks are Explosive, low explosive Pyrotechnics, pyrotechnic devices used for aesthetic and entertainment purposes. They are most commonly used in fireworks displays (also called a fireworks show or pyrotechnics), combining a large numbe ...
to give them a purple color. Rubidium has also been considered for use in a thermoelectric generator
A thermoelectric generator (TEG), also called a Seebeck generator, is a solid state device that converts heat (driven by temperature differences) directly into electrical energy through a phenomenon called the '' Seebeck effect'' (a form of the ...
using the magnetohydrodynamic principle, whereby hot rubidium ions are passed through a magnetic field
A magnetic field (sometimes called B-field) is a physical field that describes the magnetic influence on moving electric charges, electric currents, and magnetic materials. A moving charge in a magnetic field experiences a force perpendicular ...
. These conduct electricity and act like an armature of a generator, thereby generating an electric current
An electric current is a flow of charged particles, such as electrons or ions, moving through an electrical conductor or space. It is defined as the net rate of flow of electric charge through a surface. The moving particles are called charge c ...
. Rubidium, particularly vaporized 87Rb, is one of the most commonly used atomic species employed for laser cooling
Laser cooling includes several techniques where atoms, molecules, and small mechanical systems are cooled with laser light. The directed energy of lasers is often associated with heating materials, e.g. laser cutting, so it can be counterintuit ...
and Bose–Einstein condensation. Its desirable features for this application include the ready availability of inexpensive diode laser
The laser diode chip removed and placed on the eye of a needle for scale
A laser diode (LD, also injection laser diode or ILD or semiconductor laser or diode laser) is a semiconductor device similar to a light-emitting diode in which a diode p ...
light at the relevant wavelength
In physics and mathematics, wavelength or spatial period of a wave or periodic function is the distance over which the wave's shape repeats.
In other words, it is the distance between consecutive corresponding points of the same ''phase (waves ...
and the moderate temperatures required to obtain substantial vapor pressures. For cold-atom applications requiring tunable interactions, 85Rb is preferred for its rich Feshbach spectrum.
Rubidium has been used for polarizing 3He, producing volumes of magnetized 3He gas, with the nuclear spins aligned rather than random. Rubidium vapor is optically pumped by a laser, and the polarized Rb polarizes 3He through the hyperfine interaction. Such spin-polarized 3He cells are useful for neutron polarization measurements and for producing polarized neutron beams for other purposes.
The resonant element in atomic clock
An atomic clock is a clock that measures time by monitoring the resonant frequency of atoms. It is based on atoms having different energy levels. Electron states in an atom are associated with different energy levels, and in transitions betwee ...
s utilizes the hyperfine structure
In atomic physics, hyperfine structure is defined by small shifts in otherwise degenerate electronic energy levels and the resulting splittings in those electronic energy levels of atoms, molecules, and ions, due to electromagnetic multipole int ...
of rubidium's energy levels, and rubidium is useful for high-precision timing. It is used as the main component of secondary frequency references (rubidium oscillators) in cell site transmitters and other electronic transmitting, networking, and test equipment. These rubidium standards are often used with GNSS
A satellite navigation or satnav system is a system that uses satellites to provide autonomous geopositioning. A satellite navigation system with global coverage is termed global navigation satellite system (GNSS). , four global systems are op ...
to produce a "primary frequency standard" that has greater accuracy and is less expensive than caesium standards. Such rubidium standards are often mass-produced for the telecommunications industry
The telecommunications industry within the sector of information and communication technology comprises all telecommunication/ telephone companies and Internet service providers, and plays a crucial role in the evolution of mobile communications ...
.
Other potential or current uses of rubidium include a working fluid in vapor turbines, as a getter
A getter is a deposit of reactive material that is placed inside a vacuum system to complete and maintain the vacuum. When gas molecules strike the getter material, they combine with it chemically or by adsorption. Thus the getter removes small ...
in vacuum tube
A vacuum tube, electron tube, thermionic valve (British usage), or tube (North America) is a device that controls electric current flow in a high vacuum between electrodes to which an electric voltage, potential difference has been applied. It ...
s, and as a photocell
Photodetectors, also called photosensors, are devices that detect light or other forms of electromagnetic radiation and convert it into an electrical signal. They are essential in a wide range of applications, from digital imaging and optical c ...
component. Rubidium is also used as an ingredient in special types of glass, in the production of superoxide
In chemistry, a superoxide is a compound that contains the superoxide ion, which has the chemical formula . The systematic name of the anion is dioxide(1−). The reactive oxygen ion superoxide is particularly important as the product of t ...
by burning in oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), non ...
, in the study of potassium
Potassium is a chemical element; it has Symbol (chemistry), symbol K (from Neo-Latin ) and atomic number19. It is a silvery white metal that is soft enough to easily cut with a knife. Potassium metal reacts rapidly with atmospheric oxygen to ...
ion channel
Ion channels are pore-forming membrane proteins that allow ions to pass through the channel pore. Their functions include establishing a resting membrane potential, shaping action potentials and other electrical signals by Gating (electrophysiol ...
s in biology, and as the vapor in atomic magnetometer
A magnetometer is a device that measures magnetic field or magnetic dipole moment. Different types of magnetometers measure the direction, strength, or relative change of a magnetic field at a particular location. A compass is one such device, ...
s. In particular, 87Rb is used with other alkali metals in the development of spin-exchange relaxation-free (SERF) magnetometers.
Rubidium-82
Rubidium-82 (82Rb) is a radioactive isotope of rubidium. 82Rb is widely used in myocardial perfusion imaging. This isotope undergoes rapid uptake by myocardiocytes, which makes it a valuable tool for identifying myocardial ischemia in Positron Em ...
is used for positron emission tomography
Positron emission tomography (PET) is a functional imaging technique that uses radioactive substances known as radiotracers to visualize and measure changes in metabolic processes, and in other physiological activities including blood flow, r ...
. Rubidium is very similar to potassium, and tissue with high potassium content will also accumulate the radioactive rubidium. One of the main uses is myocardial perfusion imaging
Myocardial perfusion imaging or scanning (also referred to as MPI or MPS) is a nuclear medicine procedure that illustrates the function of the heart muscle (myocardium).
It evaluates many heart conditions, such as coronary artery disease (CAD), ...
. As a result of changes in the blood–brain barrier
The blood–brain barrier (BBB) is a highly selective semipermeable membrane, semipermeable border of endothelium, endothelial cells that regulates the transfer of solutes and chemicals between the circulatory system and the central nervous system ...
in brain tumors, rubidium collects more in brain tumors than normal brain tissue, allowing the use of radioisotope rubidium-82 in nuclear medicine
Nuclear medicine (nuclear radiology, nucleology), is a medical specialty involving the application of radioactivity, radioactive substances in the diagnosis and treatment of disease. Nuclear imaging is, in a sense, ''radiology done inside out'', ...
to locate and image brain tumors. Rubidium-82 has a very short half-life of 76 seconds, and the production from decay of strontium-82 must be done close to the patient.
Rubidium was tested for the influence on manic depression and depression. Dialysis patients suffering from depression show a depletion in rubidium, and therefore a supplementation may help during depression. In some tests the rubidium was administered as rubidium chloride with up to 720 mg per day for 60 days.
Precautions and biological effects
Rubidium reacts violently with water and can cause fires. To ensure safety and purity, this metal is usually kept under drymineral oil
Mineral oil is any of various colorless, odorless, light mixtures of higher alkanes from a mineral source, particularly a distillate of petroleum, as distinct from usually edible vegetable oils.
The name 'mineral oil' by itself is imprecise, ...
or sealed in glass ampoules in an inert atmosphere. Rubidium forms peroxide
In chemistry, peroxides are a group of Chemical compound, compounds with the structure , where the R's represent a radical (a portion of a complete molecule; not necessarily a free radical) and O's are single oxygen atoms. Oxygen atoms are joined ...
s on exposure even to a small amount of air diffused into the oil, and storage is subject to similar precautions as the storage of metallic potassium
Potassium is a chemical element; it has Symbol (chemistry), symbol K (from Neo-Latin ) and atomic number19. It is a silvery white metal that is soft enough to easily cut with a knife. Potassium metal reacts rapidly with atmospheric oxygen to ...
.
Rubidium, like sodium and potassium, almost always has +1 oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical Electrical charge, charge of an atom if all of its Chemical bond, bonds to other atoms are fully Ionic bond, ionic. It describes the degree of oxidation (loss of electrons ...
when dissolved in water, even in biological contexts. The human body tends to treat Rb+ ions as if they were potassium ions, and therefore concentrates rubidium in the body's intracellular fluid
The human body and even its individual body fluids may be conceptually divided into various fluid compartments, which, although not literally fascial compartment, anatomic compartments, do represent a real division in terms of how portions of the ...
(i.e., inside cells). The ions are not particularly toxic; a 70 kg person contains on average 0.36 g of rubidium, and an increase in this value by 50 to 100 times did not show negative effects in test persons. The biological half-life
Biological half-life (elimination half-life, pharmacological half-life) is the time taken for concentration of a drug, biological substance (such as a medication) to decrease from its maximum concentration (chemistry), concentration (Cmax (pharm ...
of rubidium in humans measures 31–46 days. Although a partial substitution of potassium by rubidium is possible, when more than 50% of the potassium in the muscle tissue of rats was replaced with rubidium, the rats died.
References
Further reading
* Meites, Louis (1963). ''Handbook of Analytical Chemistry'' (New York: McGraw-Hill Book Company, 1963) *External links
*Rubidium
at ''
The Periodic Table of Videos
''Periodic Videos'' (also known as ''The Periodic Table of Videos'') is a video project and YouTube channel on chemistry. It consists of a series of videos about chemical elements and the periodic table, with additional videos on other topics i ...
'' (University of Nottingham)
{{Authority control
Chemical elements
Alkali metals
Reducing agents
Chemical elements with body-centered cubic structure
Pyrophoric materials