Metal ions in aqueous solution on:
[Wikipedia]
[Google]
[Amazon]
A metal ion in aqueous solution or aqua ion is a
* No experimental information regarding aqua ion structures
Most
Al3+, Y3+, La3+, , , - , Li+, Na+, K+
Be2+, Mn2+, Fe2+, Co2+, Ni2+, Cu2+, Zn2+, Cd2+
Sc3+, Ti3+, V3+, Cr3+, Fe3+, Rh3+, Ga3+, In3+
Ce4+, Th4+, Pa4+, U4+, Np4+, Pu4+, , , , - , Ag+, Tl+
Pb2+
Ti3+, Bi3+, , , , - , Sn2+, Hg2+, Pd2+ , , ca. 12 The cations most resistant to hydrolysis for their size and charge are hard pre-transition metal ions or lanthanide ions. The slightly less resistant group includes the transition metal ions. The third group contains mostly soft ions ion of post-transition metals. The ions which show the strongest tendency to hydrolyze for their charge and size are Pd2+, Sn2+ and Hg2+. This is because of the low coordination numbers of ions in this part of the periodic table (also including Ag+ and Au+), so that fewer water molecules are present around the cation and they experience more electrostatic force than normal. A similar situation affects Be2+, the smallest aqua cation, which is also more acidic than would normally be expected. The standard enthalpy change for the first hydrolysis step is generally not very different from that of the dissociation of pure water. Consequently, the standard enthalpy change for the substitution reaction : (H2O)nsup>z+ +OH− : (H2O)n-1(OH)sup>(z-1)+ + H2O is close to zero. This is typical of reactions between a hard cation and a hard anion, such as the hydroxide ion. It means that the standard entropy charge is the major contributor to the standard free energy change and hence the equilibrium constant. : The change in ionic charge is responsible for the effect as the aqua ion has a greater ordering effect on the solution than the less highly charged hydroxo complex.
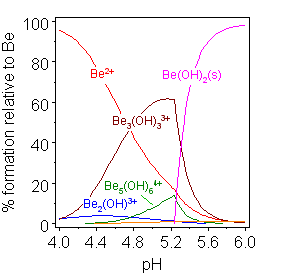

 The hydrolysis of beryllium shows many of the characteristics typical of multiple hydrolysis reactions. The concentrations of various species, including polynuclear species with bridging hydroxide ions, change as a function of pH up to the precipitation of an insoluble hydroxide. Beryllium hydrolysis is unusual in that the concentration of e(H2O)3(OH)sup>+ is too low to be measured. Instead a trimer ([Be3(H2O)6(OH3))3+ is formed, whose structure has been confirmed in solid salts. The formation of polynuclear species is driven by the reduction in charge density within the molecule as a whole. The local environment of the beryllium ions approximates to [Be(H2O)2(OH)2]+. The reduction in effective charge releases free energy in the form of a decrease of the entropy of ordering at the charge centers.
:{, class="wikitable"
, +Some polynuclear hydrolysis products
! Species formula!! cations!! ! scope="col" width="300" , structure
, -
, M2(OH)+, , Be2+, Mn2+, Co2+, Ni2+
The hydrolysis of beryllium shows many of the characteristics typical of multiple hydrolysis reactions. The concentrations of various species, including polynuclear species with bridging hydroxide ions, change as a function of pH up to the precipitation of an insoluble hydroxide. Beryllium hydrolysis is unusual in that the concentration of e(H2O)3(OH)sup>+ is too low to be measured. Instead a trimer ([Be3(H2O)6(OH3))3+ is formed, whose structure has been confirmed in solid salts. The formation of polynuclear species is driven by the reduction in charge density within the molecule as a whole. The local environment of the beryllium ions approximates to [Be(H2O)2(OH)2]+. The reduction in effective charge releases free energy in the form of a decrease of the entropy of ordering at the charge centers.
:{, class="wikitable"
, +Some polynuclear hydrolysis products
! Species formula!! cations!! ! scope="col" width="300" , structure
, -
, M2(OH)+, , Be2+, Mn2+, Co2+, Ni2+
Zn2+, Cd2+, Hg2+, Pb2+ , , single hydroxide bridge between two cations , - , M2(OH) , , Cu2+, Sn2+
Al3+, Sc3+, Ln3+, Ti3+, Cr3+
Th4+
VO2+, , , , , double hydroxide bridge between two cations , - , , , Be2+, Hg2+ , , six-membered ring with alternate Mn+ and OH− groups , - , (OH) , , Sn2+, Pb2+
Al3+, Cr3+, Fe3+, In3+ , , Cube with alternate vertices of Mn+ and OH− groups, one vertex missing , - , , , Mg2+, Co2+, Ni2+, Cd2+, Pb2+ , , Cube with alternate vertices of Mn+ and OH− groups , - , , , Zr4+, Th4+ , , Square of Mn+ ions with double hydroxide bridges on each side of the square The hydrolysis product of
cation
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convent ...
, dissolved in water
Water is an inorganic compound with the chemical formula . It is a transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance. It is the main constituent of Earth's hydrosphere and the fluids of all known liv ...
, of chemical formula
A chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbols, such as pare ...
(H2O)nsup>z+. The solvation number, ''n'', determined by a variety of experimental methods is 4 for Li+ and Be2+ and 6 for most elements in periods 3 and 4 of the periodic table
The periodic table, also known as the periodic table of the elements, is an ordered arrangement of the chemical elements into rows (" periods") and columns (" groups"). It is an icon of chemistry and is widely used in physics and other s ...
. Lanthanide
The lanthanide () or lanthanoid () series of chemical elements comprises at least the 14 metallic chemical elements with atomic numbers 57–70, from lanthanum through ytterbium. In the periodic table, they fill the 4f orbitals. Lutetium (el ...
and actinide
The actinide () or actinoid () series encompasses at least the 14 metallic chemical elements in the 5f series, with atomic numbers from 89 to 102, actinium through nobelium. Number 103, lawrencium, is also generally included despite being part ...
aqua ions have higher solvation numbers (often 8 to 9), with the highest known being 11 for Ac3+. The strength of the bonds between the metal ion and water molecules in the primary solvation shell
A solvation shell or solvation sheath is the solvent interface of any chemical compound or biomolecule that constitutes the solute in a solution. When the solvent is water it is called a hydration shell or hydration sphere. The number of solvent ...
increases with the electrical charge, ''z'', on the metal ion and decreases as its ionic radius
Ionic radius, ''r''ion, is the radius of a monatomic ion in an ionic crystal structure. Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cati ...
, ''r'', increases. Aqua ions are subject to hydrolysis. The logarithm of the first hydrolysis constant is proportional to ''z''2/''r'' for most aqua ions.
The aqua ion is associated, through hydrogen bond
In chemistry, a hydrogen bond (H-bond) is a specific type of molecular interaction that exhibits partial covalent character and cannot be described as a purely electrostatic force. It occurs when a hydrogen (H) atom, Covalent bond, covalently b ...
ing with other water molecules in a secondary solvation shell. Water molecules in the first hydration shell exchange with molecules in the second solvation shell and molecules in the bulk liquid. The residence time of a molecule in the first shell varies among the chemical element
A chemical element is a chemical substance whose atoms all have the same number of protons. The number of protons is called the atomic number of that element. For example, oxygen has an atomic number of 8: each oxygen atom has 8 protons in its ...
s from about 100 picoseconds to more than 200 years. Aqua ions are prominent in electrochemistry
Electrochemistry is the branch of physical chemistry concerned with the relationship between Electric potential, electrical potential difference and identifiable chemical change. These reactions involve Electron, electrons moving via an electronic ...
.
Introduction to metal aqua ions
: :chemical element
A chemical element is a chemical substance whose atoms all have the same number of protons. The number of protons is called the atomic number of that element. For example, oxygen has an atomic number of 8: each oxygen atom has 8 protons in its ...
s are metal
A metal () is a material that, when polished or fractured, shows a lustrous appearance, and conducts electrical resistivity and conductivity, electricity and thermal conductivity, heat relatively well. These properties are all associated wit ...
lic. Compounds of the metallic elements usually form simple aqua ions with the formula (H2O)nsup>z+ in low oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical Electrical charge, charge of an atom if all of its Chemical bond, bonds to other atoms are fully Ionic bond, ionic. It describes the degree of oxidation (loss of electrons ...
s. With the higher oxidation states the simple aqua ions dissociate
Dissociation in chemistry is a general process in which molecules (or ionic compounds such as salts, or complexes) separate or split into other things such as atoms, ions, or radicals, usually in a reversible manner. For instance, when an aci ...
losing hydrogen ion
A hydrogen ion is created when a hydrogen atom loses or gains an electron. A positively charged hydrogen ion (or proton) can readily combine with other particles and therefore is only seen isolated when it is in a gaseous state or a nearly particl ...
s to yield complexes that contain both water molecules and hydroxide or oxide ions, such as the vanadium
Vanadium is a chemical element; it has Symbol (chemistry), symbol V and atomic number 23. It is a hard, silvery-grey, malleable transition metal. The elemental metal is rarely found in nature, but once isolated artificially, the formation of an ...
(IV) species O(H2O)5sup>2+. In the highest oxidation states only oxyanion An oxyanion, or oxoanion, is an ion with the generic formula (where A represents a chemical element and O represents an oxygen atom). Oxyanions are formed by a large majority of the chemical elements. The formulae of simple oxyanions are determine ...
s, such as the permanganate
A permanganate () is a chemical compound with the manganate(VII) ion, , the conjugate base of permanganic acid. Because the manganese atom has a +7 oxidation state, the permanganate(VII) ion is a strong oxidising agent. The ion is a transition ...
(VII) ion, , are known. A few metallic elements that are commonly found only in high oxidation states, such as niobium
Niobium is a chemical element; it has chemical symbol, symbol Nb (formerly columbium, Cb) and atomic number 41. It is a light grey, crystalline, and Ductility, ductile transition metal. Pure niobium has a Mohs scale of mineral hardness, Mohs h ...
and tantalum
Tantalum is a chemical element; it has Symbol (chemistry), symbol Ta and atomic number 73. It is named after Tantalus, a figure in Greek mythology. Tantalum is a very hard, ductility, ductile, lustre (mineralogy), lustrous, blue-gray transition ...
, are not known to form aqua cations; near the metal–nonmetal boundary, arsenic
Arsenic is a chemical element; it has Symbol (chemistry), symbol As and atomic number 33. It is a metalloid and one of the pnictogens, and therefore shares many properties with its group 15 neighbors phosphorus and antimony. Arsenic is not ...
and tellurium
Tellurium is a chemical element; it has symbol Te and atomic number 52. It is a brittle, mildly toxic, rare, silver-white metalloid. Tellurium is chemically related to selenium and sulfur, all three of which are chalcogens. It is occasionally fou ...
are only known as hydrolysed species. Some elements, such as tin
Tin is a chemical element; it has symbol Sn () and atomic number 50. A silvery-colored metal, tin is soft enough to be cut with little force, and a bar of tin can be bent by hand with little effort. When bent, a bar of tin makes a sound, the ...
and antimony
Antimony is a chemical element; it has chemical symbol, symbol Sb () and atomic number 51. A lustrous grey metal or metalloid, it is found in nature mainly as the sulfide mineral stibnite (). Antimony compounds have been known since ancient t ...
, are clearly metals, but form only covalent compounds in the highest oxidation states: their aqua cations are restricted to their lower oxidation states. Germanium
Germanium is a chemical element; it has Symbol (chemistry), symbol Ge and atomic number 32. It is lustrous, hard-brittle, grayish-white and similar in appearance to silicon. It is a metalloid or a nonmetal in the carbon group that is chemically ...
is a semiconductor rather than a metal, but appears to form an aqua cation; similarly, hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter ...
forms an aqua cation like metals, despite being a gas. The transactinides have been greyed out due to a lack of experimental data. For some highly radioactive elements, experimental chemistry has been done, and aqua cations may have been formed, but no experimental information is available regarding the structure of those putative aqua ions.
In aqueous solution the water molecules directly attached to the metal ion are said to belong to the first coordination sphere, also known as the first, or primary, solvation shell. The bond between a water molecule and the metal ion is a dative covalent bond, with the oxygen atom donating both electrons to the bond. Each coordinated water molecule may be attached by hydrogen bond
In chemistry, a hydrogen bond (H-bond) is a specific type of molecular interaction that exhibits partial covalent character and cannot be described as a purely electrostatic force. It occurs when a hydrogen (H) atom, Covalent bond, covalently b ...
s to other water molecules. The latter are said to reside in the second coordination sphere. The second coordination sphere is not a well defined entity for ions with charge 1 or 2. In dilute solutions it merges into the water structure in which there is an irregular network of hydrogen bonds between water molecules. With tripositive ions the high charge on the cation polarizes the water molecules in the first solvation shell to such an extent that they form strong enough hydrogen bonds with molecules in the second shell to form a more stable entity.
The strength of the metal-oxygen bond can be estimated in various ways. The hydration enthalpy, though based indirectly on experimental measurements, is the most reliable measure. The scale of values is based on an arbitrarily chosen zero, but this does not affect differences between the values for two metals. Other measures include the M–O vibration frequency and the M–O bond length. The strength of the M-O bond tends to increase with the charge and decrease as the size of the metal ion increases. In fact there is a very good linear correlation between hydration enthalpy and the ratio of charge squared to ionic radius, z2/r. For ions in solution Shannon's "effective ionic radius" is the measure most often used.
Water molecules in the first and second solvation shells can exchange places. The rate of exchange varies enormously, depending on the metal and its oxidation state. Metal aqua ions are always accompanied in solution by solvated anions, but much less is known about anion solvation than about cation solvation.
Understanding of the nature of aqua ions is helped by having information on the nature of solvated cations in mixed solvents and non-aqueous solvent
A solvent (from the Latin language, Latin ''wikt:solvo#Latin, solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a Solution (chemistry), solution. A solvent is usually a liquid but can also be a solid, a gas ...
s, such as liquid ammonia, methanol
Methanol (also called methyl alcohol and wood spirit, amongst other names) is an organic chemical compound and the simplest aliphatic Alcohol (chemistry), alcohol, with the chemical formula (a methyl group linked to a hydroxyl group, often ab ...
, dimethyl formamide
Dimethylformamide, DMF is an organic compound with the chemical formula . Its structure is . Commonly abbreviated as DMF (although this initialism is sometimes used for dimethylfuran, or dimethyl fumarate), this colourless liquid is miscible wit ...
and dimethyl sulfoxide
Dimethyl sulfoxide (DMSO) is an organosulfur compound with the formula . This colorless liquid is the sulfoxide most widely used commercially. It is an important polar aprotic solvent that dissolves both polar and nonpolar compounds and is ...
to mention a few.
Occurrence in nature
Aqua ions are present in most natural waters.Stumm&Morgan Na+, K+, Mg2+ and Ca2+ are major constituents ofseawater
Seawater, or sea water, is water from a sea or ocean. On average, seawater in the world's oceans has a salinity of about 3.5% (35 g/L, 35 ppt, 600 mM). This means that every kilogram (roughly one liter by volume) of seawater has approximat ...
.
:
Many other aqua ions are present in seawater in concentrations ranging from ppm to ppt. The concentrations of sodium, potassium, magnesium and calcium in blood
Blood is a body fluid in the circulatory system of humans and other vertebrates that delivers necessary substances such as nutrients and oxygen to the cells, and transports metabolic waste products away from those same cells.
Blood is com ...
are similar to those of seawater. Blood also has lower concentrations of essential elements such as iron and zinc. Sports drink
Sports drinks, also known as electrolyte drinks, are non-caffeinated functional beverages whose stated purpose is to help athletes replace water, electrolytes, and energy before, during and (especially) after training or competition.
The eviden ...
is designed to be isotonic and also contains the minerals which are lost in perspiration
Perspiration, also known as sweat, is the fluid secreted by sweat glands in the skin of mammals.
Two types of sweat glands can be found in humans: eccrine glands and Apocrine sweat gland, apocrine glands. The eccrine sweat glands are distribu ...
.
Magnesium and calcium ions are common constituents of domestic water and are responsible for permanent and temporary hardness
In materials science, hardness (antonym: softness) is a measure of the resistance to plastic deformation, such as an indentation (over an area) or a scratch (linear), induced mechanically either by Pressing (metalworking), pressing or abrasion ...
, respectively. They are often found in mineral water
Mineral water is water from a mineral spring that contains various minerals, such as salts and sulfur compounds. It is usually still, but may be sparkling ( carbonated/ effervescent).
Traditionally, mineral waters were used or consumed at t ...
.
Experimental methods
Information obtained on the nature of ions in solution varies with the nature of the experimental method used. Some methods reveal properties of the cation directly, others reveal properties that depend on both cation and anion. Some methods supply information of a static nature, a kind of snapshot of average properties, others give information about the dynamics of the solution.Nuclear magnetic resonance (NMR)
Ions for which the water-exchange rate is slow on the NMR time-scale give separate peaks for molecules in the first solvation shell and for other water molecules. The solvation number is obtained as a ratio of peak areas. Here it refers to the number of water molecules in the first solvation shell. Molecules in the second solvation shell exchange rapidly with solvent molecules, giving rise to a small change in the chemical shift value of un-coordinated water molecules from that of water itself. The main disadvantage of this method is that it requires fairly concentrated solutions, with the associated risk of ion-pair formation with the anion.X-ray diffraction (XRD)
A solution containing an aqua ion does not have the long-range order that would be present in a crystal containing the same ion, but there is short-range order. X-ray diffraction on solutions yields aradial distribution function
In statistical mechanics, the radial distribution function, (or pair correlation function) g(r) in a system of particles (atoms, molecules, colloids, etc.), describes how density varies as a function of distance from a reference particle.
If ...
from which the coordination number of the metal ion and metal-oxygen distance may be derived. With aqua ions of high charge some information is obtained about the second solvation shell.
This technique requires the use of relatively concentrated solutions. X-rays are scattered by electrons, so scattering power increases with atomic number. This makes hydrogen atoms all but invisible to X-ray scattering.
Large angle X-ray scattering has been used to characterize the second solvation shell with trivalent ions such as Cr3+ and Rh3+. The second hydration shell of Cr3+ was found to have molecules at an average distance of . This implies that every molecule in the first hydration shell is hydrogen bonded to two molecules in the second shell.
Neutron diffraction
Diffraction by neutrons also give aradial distribution function
In statistical mechanics, the radial distribution function, (or pair correlation function) g(r) in a system of particles (atoms, molecules, colloids, etc.), describes how density varies as a function of distance from a reference particle.
If ...
. In contrast to X-ray diffraction, neutrons are scattered by nuclei and there is no relationship with atomic number. Indeed, use can be made of the fact that different isotopes of the same element can have widely different scattering powers. In a classic experiment, measurements were made on four nickel chloride solutions using the combinations of 58Ni, 60Ni, 35Cl and 37Cl isotopes to yield a very detailed picture of cation and anion solvation. Data for a number of metal salts show some dependence on the salt concentration.
: †Figures in brackets are standard deviation
In statistics, the standard deviation is a measure of the amount of variation of the values of a variable about its Expected value, mean. A low standard Deviation (statistics), deviation indicates that the values tend to be close to the mean ( ...
s on the last significant figure of the value.‡ angle between a M-OH2 bond and the plane of the water molecule.
Most of these data refer to concentrated solutions in which there are very few water molecules that are not in the primary hydration spheres of the cation or anion, which may account for some of the variation of solvation number with concentration even if there is no contact ion pairing. The angle θ gives the angle of tilt of the water molecules relative to a plane in the aqua ion. This angle is affected by the hydrogen bonds formed between water molecules in the primary and secondary solvation shells.
The measured solvation number is a time-averaged value for the solution as a whole. When a measured primary solvation number is fractional there are two or more species with integral solvation numbers present in equilibrium with each other. This also applies to solvation numbers that are integral numbers, within experimental error. For example, the solvation number of 5.5 for a lithium chloride
Lithium chloride is a chemical compound with the formula Li Cl. The salt is a typical ionic compound (with certain covalent characteristics), although the small size of the Li+ ion gives rise to properties not seen for other alkali metal chlorid ...
solution could be interpreted as being due to presence of two different aqua ions with equal concentrations.
: i(H2O)6sup>+ i(H2O)5sup>+ + H2O
Another possibility is that there is interaction between a solvated cation and an anion, forming an ion pair
In chemistry, ion association is a chemical reaction whereby ions of opposite electric charge come together in solution to form a distinct chemical entity. Ion associates are classified, according to the number of ions that associate with each ...
. This is particularly relevant when measurements are made on concentrated salt solutions. For example, a solvation number of 3 for a lithium chloride solution could be interpreted as being due to the equilibrium
: i(H2O)4sup>+ + Cl− i(H2O)3Cl+ H2O
lying wholly in favour of the ion pair.
Vibrational spectra
Infrared spectra and Raman spectra can be used to measure the M-O stretching frequency in metal aqua ions. Raman spectroscopy is particularly useful because the Raman spectrum of water is weak whereas the infrared spectrum of water is intense. Interpretation of the vibration frequencies is somewhat complicated by the presence, in octahedral and tetrahedral ions, of two vibrations, a symmetric one measured in the Raman spectrum and an anti-symmetric one, measured in the infrared spectrum. Although the relationship between vibration frequency and force constant is not simple, the general conclusion that can be taken from these data is that the strength of the M-O bond increases with increasing ionic charge and decreasing ionic size. The M-O stretching frequency of an aqua ion in solution may be compared with its counterpart in a crystal of known structure. If the frequencies are very similar it can be concluded that the coordination number of the metal ion is the same in solution as it is in a compound in the solid state.Dynamic methods
Data such as conductivity,electrical mobility
Electrical mobility is the ability of charged particles (such as electrons or protons) to move through a medium in response to an electric field that is pulling them. The separation of ions according to their mobility in gas phase is called ion ...
and diffusion
Diffusion is the net movement of anything (for example, atoms, ions, molecules, energy) generally from a region of higher concentration to a region of lower concentration. Diffusion is driven by a gradient in Gibbs free energy or chemical p ...
relate to the movement of ions through a solution. When an ion moves through a solution it tends to take both first and second solvation shells with it. Hence solvation numbers measured from dynamic properties tend to be much higher that those obtained from static properties.
:
Solvation numbers and structures
Hydrogen
Hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter ...
is not a metal, but like them it tends to lose its valence electron in chemical reactions, forming a cation H+. In aqueous solution, this immediately attaches itself to a water molecule, forming a species generally symbolised as H3O+ (sometimes loosely written H+). Such hydration forms cations that can in essence be considered as (OH2)''n''sup>+.
The solvation of H+ in water is not fully characterised and many different structures have been suggested. Two well-known structures are the ''Zundel cation'' and the ''Eigen cation''. The Eigen solvation structure has the hydronium ion at the center of an complex in which the hydronium is strongly hydrogen-bonded to three neighbouring water molecules. In the Zundel complex the proton is shared equally by two water molecules in a symmetric hydrogen bond.
Alkali metals
The hydratedlithium
Lithium (from , , ) is a chemical element; it has chemical symbol, symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard temperature and pressure, standard conditions, it is the least dense metal and the ...
cation in water is probably tetrahedral and four-coordinated. There are most probably six water molecules in the primary solvation sphere of the octahedral sodium
Sodium is a chemical element; it has Symbol (chemistry), symbol Na (from Neo-Latin ) and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 element, group 1 of the peri ...
ion. Potassium
Potassium is a chemical element; it has Symbol (chemistry), symbol K (from Neo-Latin ) and atomic number19. It is a silvery white metal that is soft enough to easily cut with a knife. Potassium metal reacts rapidly with atmospheric oxygen to ...
is seven-coordinate, and rubidium
Rubidium is a chemical element; it has Symbol (chemistry), symbol Rb and atomic number 37. It is a very soft, whitish-grey solid in the alkali metal group, similar to potassium and caesium. Rubidium is the first alkali metal in the group to have ...
and caesium
Caesium (IUPAC spelling; also spelled cesium in American English) is a chemical element; it has Symbol (chemistry), symbol Cs and atomic number 55. It is a soft, silvery-golden alkali metal with a melting point of , which makes it one of only f ...
are probably eight-coordinate square antiprismatic. No data is available for francium
Francium is a chemical element; it has symbol Fr and atomic number 87. It is extremely radioactive; its most stable isotope, francium-223 (originally called '' actinium K'' after the natural decay chain in which it appears), has a half-l ...
.
Alkaline earth metals
:§ Values extrapolated from data for solid-state crystal structures Theberyllium
Beryllium is a chemical element; it has Symbol (chemistry), symbol Be and atomic number 4. It is a steel-gray, hard, strong, lightweight and brittle alkaline earth metal. It is a divalent element that occurs naturally only in combination with ...
cation e(H2O)4sup>2+ has a very well-defined primary solvation shell with a tetrahedral BeO4 core.Richens, p. 129. For magnesium
Magnesium is a chemical element; it has Symbol (chemistry), symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 ...
, g(H2O)6sup>2+ is also a well-characterized species, with an octahedral MgO6 core. The situation for calcium
Calcium is a chemical element; it has symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to it ...
is more complicated. Neutron diffraction data gave a solvation number for calcium chloride, CaCl2, which is strongly dependent on concentration: at 1 mol·dm−3, decreasing to at 2.8 mol·dm−3. The enthalpy of solvation decreases with increasing ionic radius. Various solid hydrates are known with 8-coordination in square antiprism
In geometry, the square antiprism is the second in an infinite family of antiprisms formed by an even number, even-numbered sequence of triangle sides closed by two polygon caps. It is also known as an ''anticube''.
If all its faces are regular ...
and dodecahedral geometry. In water, calcium and strontium
Strontium is a chemical element; it has symbol Sr and atomic number 38. An alkaline earth metal, it is a soft silver-white yellowish metallic element that is highly chemically reactive. The metal forms a dark oxide layer when it is exposed to ...
are most probably eight-coordinate square antiprismatic (although seven-coordination for calcium cannot presently be excluded). Barium
Barium is a chemical element; it has symbol Ba and atomic number 56. It is the fifth element in group 2 and is a soft, silvery alkaline earth metal. Because of its high chemical reactivity, barium is never found in nature as a free element.
Th ...
is not as well-studied: it seems to have a coordination number of either eight or nine. Theoretical simulation of radium
Radium is a chemical element; it has chemical symbol, symbol Ra and atomic number 88. It is the sixth element in alkaline earth metal, group 2 of the periodic table, also known as the alkaline earth metals. Pure radium is silvery-white, ...
suggests that its aqua cation is ten-coordinate.
Group 3 metals, lanthanides and actinides

Scandium
Scandium is a chemical element; it has Symbol (chemistry), symbol Sc and atomic number 21. It is a silvery-white metallic d-block, d-block element. Historically, it has been classified as a rare-earth element, together with yttrium and the lantha ...
(III) and yttrium
Yttrium is a chemical element; it has Symbol (chemistry), symbol Y and atomic number 39. It is a silvery-metallic transition metal chemically similar to the lanthanides and has often been classified as a "rare-earth element". Yttrium is almost a ...
(III) are both eight-coordinate, but have different structures: scandium has an unusual dicapped triangular prismatic structure (with one cap location empty), while yttrium is square antiprismatic. Lutetium
Lutetium is a chemical element; it has symbol Lu and atomic number 71. It is a silvery white metal, which resists corrosion in dry air, but not in moist air. Lutetium is the last element in the lanthanide series, and it is traditionally counted am ...
(III) is tricapped triangular prismatic, but has a significant water deficit: one of the capping water molecules is significantly closer to the lutetium than the remaining ones and the average coordination number is only 8.2 rather than 9. Based on its ionic radius, lawrencium
Lawrencium is a synthetic chemical element; it has symbol Lr (formerly Lw) and atomic number 103. It is named after Ernest Lawrence, inventor of the cyclotron, a device that was used to discover many artificial radioactive elements. A radioactiv ...
(III) is probably nine-coordinate tricapped triangular prismatic with no water deficit.
The trivalent lanthanide ions decrease steadily in size from lanthanum
Lanthanum is a chemical element; it has symbol La and atomic number 57. It is a soft, ductile, silvery-white metal that tarnishes slowly when exposed to air. It is the eponym of the lanthanide series, a group of 15 similar elements bet ...
to lutetium
Lutetium is a chemical element; it has symbol Lu and atomic number 71. It is a silvery white metal, which resists corrosion in dry air, but not in moist air. Lutetium is the last element in the lanthanide series, and it is traditionally counted am ...
, an effect known as the lanthanide contraction
The lanthanide contraction is the greater-than-expected decrease in atomic radii and ionic radii of the elements in the lanthanide series, from left to right. It is caused by the poor shielding effect of nuclear charge by the 4f electrons alo ...
. From lanthanum to dysprosium
Dysprosium is a chemical element; it has symbol Dy and atomic number 66. It is a rare-earth element in the lanthanide series with a metallic silver luster. Dysprosium is never found in nature as a free element, though, like other lanthanides, it ...
, the coordination number is maintained at 9 with a tricapped trigonal prismatic structure, although starting from samarium the capping water molecules are no longer equally strongly bounded. A water deficit then appears for holmium
Holmium is a chemical element; it has symbol Ho and atomic number 67. It is a rare-earth element and the eleventh member of the lanthanide series. It is a relatively soft, silvery, fairly corrosion-resistant and malleable metal. Like many other ...
through lutetium with the average coordination number dropping to 8.2 at lutetium(III). The configuration is maintained despite the small size of the cations and the water deficit, probably due to strong hydrogen bonding. Europium
Europium is a chemical element; it has symbol Eu and atomic number 63. It is a silvery-white metal of the lanthanide series that reacts readily with air to form a dark oxide coating. Europium is the most chemically reactive, least dense, and soft ...
(II) is seven-coordinate, and cerium
Cerium is a chemical element; it has Chemical symbol, symbol Ce and atomic number 58. It is a hardness, soft, ductile, and silvery-white metal that tarnishes when exposed to air. Cerium is the second element in the lanthanide series, and while it ...
(IV) is hydrolysed to the oxygen-bridged dimer H2O)7Ce–O–Ce(OH2)7sup>6+.
Actinium
Actinium is a chemical element; it has chemical symbol, symbol Ac and atomic number 89. It was discovered by Friedrich Oskar Giesel in 1902, who gave it the name ''emanium''; the element got its name by being wrongly identified with a substa ...
(III) is eleven-coordinate in aqueous solution. Thorium
Thorium is a chemical element; it has symbol Th and atomic number 90. Thorium is a weakly radioactive light silver metal which tarnishes olive grey when it is exposed to air, forming thorium dioxide; it is moderately soft, malleable, and ha ...
(IV) is nine-coordinate tricapped trigonal prismatic, and it is assumed that the same is true for the other actinide(IV) cations in aqueous solutions (as that is also their solid-state configuration). Studies on coordination number and/or structure for actinides(III) to date stretch only to californium
Californium is a synthetic chemical element; it has symbol Cf and atomic number 98. It was first synthesized in 1950 at Lawrence Berkeley National Laboratory (then the University of California Radiation Laboratory) by bombarding curium with al ...
. However, since lawrencium(III) has a similar ionic radius to dysprosium(III), it is likely that uranium
Uranium is a chemical element; it has chemical symbol, symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Ura ...
(III) through lawrencium(III) are all nine-coordinate tricapped triangular prismatic with the capping positions fully occupied. No data is available for fermium(II), mendelevium
Mendelevium is a synthetic chemical element; it has symbol Md ( formerly Mv) and atomic number 101. A metallic radioactive transuranium element in the actinide series, it is the first element by atomic number that currently cannot be produced ...
(II), or nobelium
Nobelium is a synthetic element, synthetic chemical element; it has Chemical symbol, symbol No and atomic number 102. It is named after Alfred Nobel, the inventor of dynamite and benefactor of science. A radioactive metal, it is the tenth transura ...
(II).
Group 4-12 metals
The ions of these metals in the +2 and +3oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical Electrical charge, charge of an atom if all of its Chemical bond, bonds to other atoms are fully Ionic bond, ionic. It describes the degree of oxidation (loss of electrons ...
s have a solvation number of 6. All have a regular octahedral structure except the aqua ions of chromium
Chromium is a chemical element; it has Symbol (chemistry), symbol Cr and atomic number 24. It is the first element in Group 6 element, group 6. It is a steely-grey, Luster (mineralogy), lustrous, hard, and brittle transition metal.
Chromium ...
(II) and copper
Copper is a chemical element; it has symbol Cu (from Latin ) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orang ...
(II) which are subject to Jahn-Teller distortion. In the copper case the two axial Cu−O distances are 238 pm, whereas the four equatorial Cu−O distances are 195 pm in the solid state. However, it is unclear whether Cu2+ has a solvation number of 5 or 6 in aqueous solution, with conflicting experimental reports. The structure of cobalt
Cobalt is a chemical element; it has Symbol (chemistry), symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. ...
(III) in aqueous solution has not been determined. Copper(I) is estimated to be four-coordinate tetrahedral.
A solvation number of 6 with an octahedral structure is well established for zinc
Zinc is a chemical element; it has symbol Zn and atomic number 30. It is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodic tabl ...
(II) and cadmium
Cadmium is a chemical element; it has chemical symbol, symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12 element, group 12, zinc and mercury (element), mercury. Like z ...
(II) in dilute solutions. In concentrated solutions the Zn2+ ion may adopt a 4-coordinate, tetrahedral, structure, but the evidence is not conclusive because of the possibility of ion pairing and/or hydrolysis. The solvation number of mercury(II) is most likely to be 6. Zinc
Zinc is a chemical element; it has symbol Zn and atomic number 30. It is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodic tabl ...
(II) is six-coordinate octahedral, but cadmium
Cadmium is a chemical element; it has chemical symbol, symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12 element, group 12, zinc and mercury (element), mercury. Like z ...
(II) may be in equilibrium between six- and seven-coordination. Mercury(II) is a pseudo-Jahn-Teller-distorted octahedron. The bis aqua structure of the mercury(I) ion, H2O)-Hg-Hg-(OH2)sup>+, found in solid compounds, is not the same as that found in solution which involves three water molecules coordinated to each mercury completing a distorted tetrahedral arrangement. Another aqua species in which there is a metal-metal bond is the molybdenum
Molybdenum is a chemical element; it has Symbol (chemistry), symbol Mo (from Neo-Latin ''molybdaenum'') and atomic number 42. The name derived from Ancient Greek ', meaning lead, since its ores were confused with lead ores. Molybdenum minerals hav ...
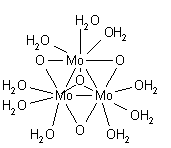
(II) species formulated as H2O)4Mo≣Mo(H2O)4sup>4+. Each molybdenum is surrounded by four water molecules in a square-planar arrangement, in a structure similar to that of the known structure of the chloro complex o2Cl8sup>4−.
There are a few divalent and trivalent aqua ions of transition metals in the second and third transition series: ruthenium
Ruthenium is a chemical element; it has symbol Ru and atomic number 44. It is a rare transition metal belonging to the platinum group of the periodic table. Like the other metals of the platinum group, ruthenium is unreactive to most chem ...
(II) and (III), rhodium
Rhodium is a chemical element; it has symbol Rh and atomic number 45. It is a very rare, silvery-white, hard, corrosion-resistant transition metal. It is a noble metal and a member of the platinum group. It has only one naturally occurring isot ...
(III), and iridium
Iridium is a chemical element; it has the symbol Ir and atomic number 77. This very hard, brittle, silvery-white transition metal of the platinum group, is considered the second-densest naturally occurring metal (after osmium) with a density ...
(III), all octahedral. (Ruthenium and iridium structures have only been examined in the solid state, but it is assumed that they are the same in aqueous solution.) Molybdenum
Molybdenum is a chemical element; it has Symbol (chemistry), symbol Mo (from Neo-Latin ''molybdaenum'') and atomic number 42. The name derived from Ancient Greek ', meaning lead, since its ores were confused with lead ores. Molybdenum minerals hav ...
(III) is questionable (and may be strongly hydrolyzed in aqueous solution), and molybdenum(II) dimerises with each molybdenum binding four water molecules. Palladium
Palladium is a chemical element; it has symbol Pd and atomic number 46. It is a rare and lustrous silvery-white metal discovered in 1802 by the English chemist William Hyde Wollaston. He named it after the asteroid Pallas (formally 2 Pallas), ...
(II) and platinum
Platinum is a chemical element; it has Symbol (chemistry), symbol Pt and atomic number 78. It is a density, dense, malleable, ductility, ductile, highly unreactive, precious metal, precious, silverish-white transition metal. Its name origina ...
(II) aqua ions were originally thought to be square planar, but are actually strongly tetragonally elongated square-pyramidal or octahedral with the extra one or two water molecules extremely loosely bound. The structure of silver
Silver is a chemical element; it has Symbol (chemistry), symbol Ag () and atomic number 47. A soft, whitish-gray, lustrous transition metal, it exhibits the highest electrical conductivity, thermal conductivity, and reflectivity of any metal. ...
(I) is disputed: it may be two-coordinate, or it may be four-coordinate with two extra very loosely bound water molecules. Gold
Gold is a chemical element; it has chemical symbol Au (from Latin ) and atomic number 79. In its pure form, it is a brightness, bright, slightly orange-yellow, dense, soft, malleable, and ductile metal. Chemically, gold is a transition metal ...
(III) is four-coordinate square planar in the solid state, and it is assumed to have the same structure in aqueous solution. Distortion occurs for low-coordinate metals with strong covalent tendencies due to the second-order Jahn-Teller effect. With oxidation state 4, however, the only unhydrolyzed species are the square antiprismatic zirconium
Zirconium is a chemical element; it has Symbol (chemistry), symbol Zr and atomic number 40. First identified in 1789, isolated in impure form in 1824, and manufactured at scale by 1925, pure zirconium is a lustrous transition metal with a greyis ...
(IV), r(H2O)8sup>4+, and hafnium
Hafnium is a chemical element; it has symbol Hf and atomic number 72. A lustrous, silvery gray, tetravalent transition metal, hafnium chemically resembles zirconium and is found in many zirconium minerals. Its existence was predicted by Dm ...
(IV), f(H2O)8sup>4+, and even they are extremely prone to hydrolysis. Such a zirconium cation is only formed in dilute solutions of ZrIV in strong acid, and in practice the cationic species encountered of zirconium and hafnium are polynuclear.
Group 13-18 elements
Boron
Boron is a chemical element; it has symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the boron group it has three ...
is not a metal, and boron(III) is too acidic for an aqua ion to exist: deprotonation proceeds as far as boric acid
Boric acid, more specifically orthoboric acid, is a compound of boron, oxygen, and hydrogen with formula . It may also be called hydrogen orthoborate, trihydroxidoboron or boracic acid. It is usually encountered as colorless crystals or a white ...
, borate
A borate is any of a range of boron oxyanions, anions containing boron and oxygen, such as orthoborate , metaborate , or tetraborate ; or any salt of such anions, such as sodium metaborate, and borax . The name also refers to esters of su ...
s, and hydroxyborates. The aluminium
Aluminium (or aluminum in North American English) is a chemical element; it has chemical symbol, symbol Al and atomic number 13. It has a density lower than that of other common metals, about one-third that of steel. Aluminium has ...
(III) aqua ion, l(H2O)6sup>3+ is very well characterized in solution and the solid state. The AlO6 core has octahedral symmetry, point group
In geometry, a point group is a group (mathematics), mathematical group of symmetry operations (isometry, isometries in a Euclidean space) that have a Fixed point (mathematics), fixed point in common. The Origin (mathematics), coordinate origin o ...
Oh. The aqua ions of gallium
Gallium is a chemical element; it has Chemical symbol, symbol Ga and atomic number 31. Discovered by the French chemist Paul-Émile Lecoq de Boisbaudran in 1875,
elemental gallium is a soft, silvery metal at standard temperature and pressure. ...
(III), indium
Indium is a chemical element; it has Symbol (chemistry), symbol In and atomic number 49. It is a silvery-white post-transition metal and one of the softest elements. Chemically, indium is similar to gallium and thallium, and its properties are la ...
(III) and thallium
Thallium is a chemical element; it has Symbol (chemistry), symbol Tl and atomic number 81. It is a silvery-white post-transition metal that is not found free in nature. When isolated, thallium resembles tin, but discolors when exposed to air. Che ...
(III) are also six-coordinate octahedral. The coordination geometry of thallium(I) is not experimentally known, but it is likely to be hemidirected with a large gap in the coordination sphere.
Silicon
Silicon is a chemical element; it has symbol Si and atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic lustre, and is a tetravalent metalloid (sometimes considered a non-metal) and semiconductor. It is a membe ...
is likewise not a metal, and silicon(IV) is a strong enough acid to deprotonate bound OH−. Thus various forms of hydrated silica
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula , commonly found in nature as quartz. In many parts of the world, silica is the major constituent of sand. Silica is one of the most complex and abundant f ...
(silicic acid
In chemistry, a silicic acid () is any chemical compound containing the element silicon attached to oxide () and hydroxyl () groups, with the general formula or, equivalently, . Orthosilicic acid is a representative example. Silicic acids are ra ...
) form.Richens, pp. 151–152 There is some evidence that germanium
Germanium is a chemical element; it has Symbol (chemistry), symbol Ge and atomic number 32. It is lustrous, hard-brittle, grayish-white and similar in appearance to silicon. It is a metalloid or a nonmetal in the carbon group that is chemically ...
(II) aqua ions can form in perchloric acid
Perchloric acid is a mineral acid with the formula H Cl O4. It is an oxoacid of chlorine. Usually found as an aqueous solution, this colorless compound is a stronger acid than sulfuric acid, nitric acid and hydrochloric acid. It is a powerful oxid ...
media. Quantum mechanical calculations suggests that the germanium(II) aqua ion shows extreme distortion of the first coordination sphere due to the high charge density and the stereochemically active lone pairs. The first shell is calculated to usually have a solvation number of 6, but numbers 4–7 are also possible and the shell splits into two with differing distances from the central Ge2+. However, germanium(II) is readily oxidised to germanium(IV),Richens, p. 152–4 for which only hydrolyzed species are expected. The important germanium(IV) species are anionic oxo-hydroxo mixed species, thus displaying intermediate behaviour between silicon and tin: the major species appear to be eO(OH)3sup>− and the octameric e8O16(OH)3sup>3−, with eO2(OH)2sup>2− occurring in smaller quantities. Tin
Tin is a chemical element; it has symbol Sn () and atomic number 50. A silvery-colored metal, tin is soft enough to be cut with little force, and a bar of tin can be bent by hand with little effort. When bent, a bar of tin makes a sound, the ...
(II) is 3-coordinate hemidirected with a very large gap in the coordination sphere of tin(II). The hydration number of lead
Lead () is a chemical element; it has Chemical symbol, symbol Pb (from Latin ) and atomic number 82. It is a Heavy metal (elements), heavy metal that is density, denser than most common materials. Lead is Mohs scale, soft and Ductility, malleabl ...
(II) is not well-established and could be anywhere from five to seven. In practice these cations tend to be polynuclear. For tin(IV) and lead(IV) there are only hydrolyzed species.
Arsenic
Arsenic is a chemical element; it has Symbol (chemistry), symbol As and atomic number 33. It is a metalloid and one of the pnictogens, and therefore shares many properties with its group 15 neighbors phosphorus and antimony. Arsenic is not ...
(III) is calculated to form hydrolyzed species only. The stable cationic arsenic(III) species in water is calculated to be s(OH)2sup>+, though hydrolysis usually proceeds further to neutral and anionic species. Antimony
Antimony is a chemical element; it has chemical symbol, symbol Sb () and atomic number 51. A lustrous grey metal or metalloid, it is found in nature mainly as the sulfide mineral stibnite (). Antimony compounds have been known since ancient t ...
(III) aqua ions may exist in dilute solutions of antimony(III) in concentrated acids.Richens, p. 155 Quantum mechanical calculations reveal a solvation number of 8, with the first coordination sphere splitting into two hydration hemispheres with 4 water molecules each. Bismuth
Bismuth is a chemical element; it has symbol Bi and atomic number 83. It is a post-transition metal and one of the pnictogens, with chemical properties resembling its lighter group 15 siblings arsenic and antimony. Elemental bismuth occurs nat ...
(III) is eight-coordinate square antiprismatic in aqueous solution, though in the solid state it is nine-coordinate tricapped triangular prismatic. Although the structures for thallium(I), germanium(II), tin(II), lead(II), and antimony(III) are affected by the lone pairs, this is not so for bismuth(III).
Selenium
Selenium is a chemical element; it has symbol (chemistry), symbol Se and atomic number 34. It has various physical appearances, including a brick-red powder, a vitreous black solid, and a grey metallic-looking form. It seldom occurs in this elem ...
(IV) is mostly present as selenous acid (H2SeO3) below pH 2; at higher pH this deprotonates to HSeO3− and then SeO32−.
:{, class="wikitable"
, +log K1,-1 = A + 11.0 z/d
!cation, , A
, -
, Mg2+, Ca2+, Sr2+, Ba2+ Al3+, Y3+, La3+, , , - , Li+, Na+, K+
Be2+, Mn2+, Fe2+, Co2+, Ni2+, Cu2+, Zn2+, Cd2+
Sc3+, Ti3+, V3+, Cr3+, Fe3+, Rh3+, Ga3+, In3+
Ce4+, Th4+, Pa4+, U4+, Np4+, Pu4+, , , , - , Ag+, Tl+
Pb2+
Ti3+, Bi3+, , , , - , Sn2+, Hg2+, Pd2+ , , ca. 12 The cations most resistant to hydrolysis for their size and charge are hard pre-transition metal ions or lanthanide ions. The slightly less resistant group includes the transition metal ions. The third group contains mostly soft ions ion of post-transition metals. The ions which show the strongest tendency to hydrolyze for their charge and size are Pd2+, Sn2+ and Hg2+. This is because of the low coordination numbers of ions in this part of the periodic table (also including Ag+ and Au+), so that fewer water molecules are present around the cation and they experience more electrostatic force than normal. A similar situation affects Be2+, the smallest aqua cation, which is also more acidic than would normally be expected. The standard enthalpy change for the first hydrolysis step is generally not very different from that of the dissociation of pure water. Consequently, the standard enthalpy change for the substitution reaction : (H2O)nsup>z+ +OH− : (H2O)n-1(OH)sup>(z-1)+ + H2O is close to zero. This is typical of reactions between a hard cation and a hard anion, such as the hydroxide ion. It means that the standard entropy charge is the major contributor to the standard free energy change and hence the equilibrium constant. : The change in ionic charge is responsible for the effect as the aqua ion has a greater ordering effect on the solution than the less highly charged hydroxo complex.
Multiple hydrolysis reactions

 The hydrolysis of beryllium shows many of the characteristics typical of multiple hydrolysis reactions. The concentrations of various species, including polynuclear species with bridging hydroxide ions, change as a function of pH up to the precipitation of an insoluble hydroxide. Beryllium hydrolysis is unusual in that the concentration of e(H2O)3(OH)sup>+ is too low to be measured. Instead a trimer ([Be3(H2O)6(OH3))3+ is formed, whose structure has been confirmed in solid salts. The formation of polynuclear species is driven by the reduction in charge density within the molecule as a whole. The local environment of the beryllium ions approximates to [Be(H2O)2(OH)2]+. The reduction in effective charge releases free energy in the form of a decrease of the entropy of ordering at the charge centers.
:{, class="wikitable"
, +Some polynuclear hydrolysis products
! Species formula!! cations!! ! scope="col" width="300" , structure
, -
, M2(OH)+, , Be2+, Mn2+, Co2+, Ni2+
The hydrolysis of beryllium shows many of the characteristics typical of multiple hydrolysis reactions. The concentrations of various species, including polynuclear species with bridging hydroxide ions, change as a function of pH up to the precipitation of an insoluble hydroxide. Beryllium hydrolysis is unusual in that the concentration of e(H2O)3(OH)sup>+ is too low to be measured. Instead a trimer ([Be3(H2O)6(OH3))3+ is formed, whose structure has been confirmed in solid salts. The formation of polynuclear species is driven by the reduction in charge density within the molecule as a whole. The local environment of the beryllium ions approximates to [Be(H2O)2(OH)2]+. The reduction in effective charge releases free energy in the form of a decrease of the entropy of ordering at the charge centers.
:{, class="wikitable"
, +Some polynuclear hydrolysis products
! Species formula!! cations!! ! scope="col" width="300" , structure
, -
, M2(OH)+, , Be2+, Mn2+, Co2+, Ni2+ Zn2+, Cd2+, Hg2+, Pb2+ , , single hydroxide bridge between two cations , - , M2(OH) , , Cu2+, Sn2+
Al3+, Sc3+, Ln3+, Ti3+, Cr3+
Th4+
VO2+, , , , , double hydroxide bridge between two cations , - , , , Be2+, Hg2+ , , six-membered ring with alternate Mn+ and OH− groups , - , (OH) , , Sn2+, Pb2+
Al3+, Cr3+, Fe3+, In3+ , , Cube with alternate vertices of Mn+ and OH− groups, one vertex missing , - , , , Mg2+, Co2+, Ni2+, Cd2+, Pb2+ , , Cube with alternate vertices of Mn+ and OH− groups , - , , , Zr4+, Th4+ , , Square of Mn+ ions with double hydroxide bridges on each side of the square The hydrolysis product of
aluminium
Aluminium (or aluminum in North American English) is a chemical element; it has chemical symbol, symbol Al and atomic number 13. It has a density lower than that of other common metals, about one-third that of steel. Aluminium has ...
formulated as l13O4(OH)24(H2O)12sup>7+ is very well characterized and may be present in nature in water at pH ca. 5.4.
The overall reaction for the loss of two protons from an aqua ion can be written as
: (H2O)nsup>z+ - 2 H+ (H2O)n-2(OH)2sup>(z-2)+
However, the equilibrium constant for the loss of two protons applies equally well to the equilibrium
: (H2O)nsup>z+ - 2 H+ O(H2O)n-2sup>(z-2)+ + H2O
because the concentration of water is assumed to be constant. This applies in general: any equilibrium constant is equally valid for a product with an oxide ion as for the product with two hydroxyl ions. The two possibilities can only be distinguished by determining the structure of a salt in the solid state. Oxo bridges tend to occur when the metal oxidation state is high. An example is provided by the molybdenum
Molybdenum is a chemical element; it has Symbol (chemistry), symbol Mo (from Neo-Latin ''molybdaenum'') and atomic number 42. The name derived from Ancient Greek ', meaning lead, since its ores were confused with lead ores. Molybdenum minerals hav ...
(IV) complex o3O4(H2O)9sup>4+ in which there is a triangle of molybdenum atoms joined by σ- bonds with an oxide bridge on each edge of the triangle and a fourth oxide which bridges to all three Mo atoms.
Oxyanions
There are very few oxo-aqua ions of metals in the oxidation state +5 or higher. Rather, the species found in aqueous solution are monomeric and polymeric oxyanions. Oxyanions can be viewed as the end products of hydrolysis, in which there are no water molecules attached to the metal, only oxide ions.Exchange kinetics
A water molecule in the first solvation shell of an aqua ion may exchange places with a water molecule in the bulk solvent. It is usually assumed that the rate-determining step is a dissociation reaction. : (H2O)nsup>z+ → (H2O)n-1sup>z+* + H2O The * symbol signifies that this is thetransition state
In chemistry, the transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest potential energy along this reaction coordinate. It is often marked w ...
in a chemical reaction. The rate of this reaction is proportional to the concentration of the aqua ion,
: