Dithiobenzoate on:
[Wikipedia]
[Google]
[Amazon]
Dithiobenzoic acid is the
organosulfur compound
Organosulfur chemistry is the study of the properties and synthesis of organosulfur compounds, which are organic compounds that contain sulfur. They are often associated with foul odors, but many of the sweetest compounds known are organosulfur der ...
with the formula C6H5CS2H. It is a dithiocarboxylic acid Dithiocarboxylic acids are organosulfur compounds with the formula . They are the dithia analogues of carboxylic acids. A closely related and better studied family of compounds are the monothiocarboxylic acids, with the formula .
Dithiocarboxyli ...
, an analogue of benzoic acid
Benzoic acid () is a white (or colorless) solid organic compound with the formula , whose structure consists of a benzene ring () with a carboxyl () substituent. The benzoyl group is often abbreviated "Bz" (not to be confused with "Bn," which ...
, but more acidic and deeply colored.
Synthesis and reactions
It can be prepared by sulfidingbenzotrichloride
Benzotrichloride (BTC), also known as α,α,α-trichlorotoluene, phenyl chloroform or (trichloromethyl)benzene, is an organic compound with the formula C6H5CCl3. Benzotrichloride is an unstable, colorless or somewhat yellowish, viscous, chlorinat ...
:
:C6H5CCl3 + 4 KSH → C6H5CS2K + 3 KCl + 2 H2S
:C6H5CS2K + H+ → C6H5CS2H + K+
It also arises by the reaction of the Grignard reagent phenylmagnesium bromide
Phenylmagnesium bromide, with the simplified formula , is a magnesium-containing organometallic compound. It forms colorless crystals. It is commercially available as a solution in diethyl ether or tetrahydrofuran (THF). Phenylmagnesium bromide i ...
with carbon disulfide
Carbon disulfide (also spelled as carbon disulphide) is an inorganic compound with the chemical formula and structure . It is also considered as the anhydride of thiocarbonic acid. It is a colorless, flammable, neurotoxic liquid that is used as ...
, followed by acidification:
:C6H5MgBr + CS2 → C6H5CS2MgBr
:C6H5CS2MgBr + HCl → C6H5CS2H + MgBrCl
It is about 100x more acidic than benzoic acid
Benzoic acid () is a white (or colorless) solid organic compound with the formula , whose structure consists of a benzene ring () with a carboxyl () substituent. The benzoyl group is often abbreviated "Bz" (not to be confused with "Bn," which ...
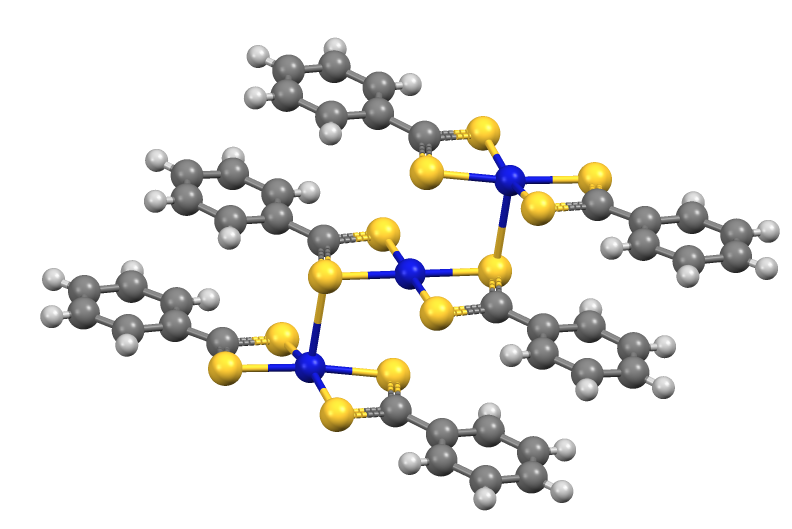
. Its conjugate base, dithiobenzoate, undergoes S-alkylation to give dithiocarboxylate esters. Similarly, dithiobenzoate reacts with "soft" metal salts to give complexes, e.g. Fe(S2CC6H5)3 and Ni(S2CC6H5)2.
Chlorination of dithiobenzoic acid gives the thioacyl chloride
In organic chemistry, a thioacyl chloride is an organic compound containing the functional group . Their formula is usually written , where R is a side chain. Thioacyl chlorides are analogous to acyl chlorides, but much rarer and less robust. Ind ...
C6H5C(S)Cl.

References