Core Charge on:
[Wikipedia]
[Google]
[Amazon]
Core electrons are the

electron
The electron (, or in nuclear reactions) is a subatomic particle with a negative one elementary charge, elementary electric charge. It is a fundamental particle that comprises the ordinary matter that makes up the universe, along with up qua ...
s in an atom
Atoms are the basic particles of the chemical elements. An atom consists of a atomic nucleus, nucleus of protons and generally neutrons, surrounded by an electromagnetically bound swarm of electrons. The chemical elements are distinguished fr ...
that are not valence electron
In chemistry and physics, valence electrons are electrons in the outermost shell of an atom, and that can participate in the formation of a chemical bond if the outermost shell is not closed. In a single covalent bond, a shared pair forms with b ...
s and do not participate as directly in chemical bond
A chemical bond is the association of atoms or ions to form molecules, crystals, and other structures. The bond may result from the electrostatic force between oppositely charged ions as in ionic bonds or through the sharing of electrons a ...
ing. The nucleus and the core electrons of an atom form the atomic core. Core electrons are tightly bound to the nucleus. Therefore, unlike valence electrons, core electrons play a secondary role in chemical bonding and reactions by screening the positive charge of the atomic nucleus from the valence electrons.
The number of valence electrons of an element can be determined by the periodic table group
In chemistry, a group (also known as a family) is a column of elements in the periodic table of the chemical elements. There are 18 numbered groups in the periodic table; the 14 f-block columns, between groups 2 and 3, are not numbered. The elem ...
of the element (see valence electron
In chemistry and physics, valence electrons are electrons in the outermost shell of an atom, and that can participate in the formation of a chemical bond if the outermost shell is not closed. In a single covalent bond, a shared pair forms with b ...
):
*For main-group element
In chemistry and atomic physics, the main group is the group (periodic table), group of chemical element, elements (sometimes called the representative elements) whose lightest members are represented by helium, lithium, beryllium, boron, carbon ...
s, the number of valence electrons ranges from 1 to 8 (''n''s and ''n''p orbitals).
*For transition metal
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. The lanthanide and actinid ...
s, the number of valence electrons ranges from 3 to 12 (''n''s and (''n''−1)d orbitals).
*For lanthanide
The lanthanide () or lanthanoid () series of chemical elements comprises at least the 14 metallic chemical elements with atomic numbers 57–70, from lanthanum through ytterbium. In the periodic table, they fill the 4f orbitals. Lutetium (el ...
s and actinide
The actinide () or actinoid () series encompasses at least the 14 metallic chemical elements in the 5f series, with atomic numbers from 89 to 102, actinium through nobelium. Number 103, lawrencium, is also generally included despite being part ...
s, the number of valence electrons ranges from 3 to 16 (''n''s, (''n''−2)f and (''n''−1)d orbitals).
All other non-valence electrons for an atom of that element are considered core electrons.
Orbital theory
A more complex explanation of the difference between core and valence electrons can be described with atomic orbital theory. In atoms with a single electron the energy of an orbital is determined exclusively by the principal quantum number ''n''. The ''n'' = 1 orbital has the lowest possible energy in the atom. For large ''n'', the energy increases so much that the electron can easily escape from the atom. In single electron atoms, all energy levels with the same principle quantum number are degenerate, and have the same energy. In atoms with more than one electron, the energy of an electron depends not only on the properties of the orbital it resides in, but also on its interactions with the other electrons in other orbitals. This requires consideration of the ''ℓ'' quantum number. Higher values of ''ℓ'' are associated with higher values of energy; for instance, the 2p state is higher than the 2s state. When ''ℓ'' = 2, the increase in energy of the orbital becomes large enough to push the energy of orbital above the energy of the s-orbital in the next higher shell; when ''ℓ'' = 3 the energy is pushed into the shell two steps higher. The filling of the 3d orbitals does not occur until the 4s orbitals have been filled. The increase in energy for subshells of increasing angular momentum in larger atoms is due to electron–electron interaction effects, and it is specifically related to the ability of low angular momentum electrons to penetrate more effectively toward the nucleus, where they are subject to less screening from the charge of intervening electrons. Thus, in atoms of higheratomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of its atomic nucleus. For ordinary nuclei composed of protons and neutrons, this is equal to the proton number (''n''p) or the number of pro ...
, the ''ℓ'' of electrons becomes more and more of a determining factor in their energy, and the principal quantum numbers ''n'' of electrons becomes less and less important in their energy placement. The energy sequence of the first 35 subshells (e.g., 1s, 2s, 2p, 3s, etc.) is given in the following table ot shown? Each cell represents a subshell with ''n'' and ''ℓ'' given by its row and column indices, respectively. The number in the cell is the subshell's position in the sequence. See the periodic table below, organized by subshells.
Atomic core
The atomic core refers to the central part of theatom
Atoms are the basic particles of the chemical elements. An atom consists of a atomic nucleus, nucleus of protons and generally neutrons, surrounded by an electromagnetically bound swarm of electrons. The chemical elements are distinguished fr ...
excluding the valence electrons. The atomic core has a positive electric charge
Electric charge (symbol ''q'', sometimes ''Q'') is a physical property of matter that causes it to experience a force when placed in an electromagnetic field. Electric charge can be ''positive'' or ''negative''. Like charges repel each other and ...
called the core charge and is the effective nuclear charge
In atomic physics, the effective nuclear charge of an electron in a multi-electron atom or ion is the number of elementary charges (e) an electron experiences by the nucleus. It is denoted by ''Z''eff. The term "effective" is used because the shi ...
experienced by an outer shell electron
The electron (, or in nuclear reactions) is a subatomic particle with a negative one elementary charge, elementary electric charge. It is a fundamental particle that comprises the ordinary matter that makes up the universe, along with up qua ...
. In other words, core charge is an expression of the attractive force experienced by the valence electrons to the core of an atom
Atoms are the basic particles of the chemical elements. An atom consists of a atomic nucleus, nucleus of protons and generally neutrons, surrounded by an electromagnetically bound swarm of electrons. The chemical elements are distinguished fr ...
which takes into account the shielding effect
In chemistry, the shielding effect sometimes referred to as atomic shielding or
electron shielding describes the attraction between an electron and the nucleus in any atom with more than one electron. The shielding effect can be defined as a r ...
of core electrons. Core charge can be calculated by taking the number of proton
A proton is a stable subatomic particle, symbol , Hydron (chemistry), H+, or 1H+ with a positive electric charge of +1 ''e'' (elementary charge). Its mass is slightly less than the mass of a neutron and approximately times the mass of an e ...
s in the nucleus minus the number of core electrons, also called inner shell electrons, and is always a positive value in neutral atoms.
The mass of the core is almost equal to the mass of the atom. The atomic core can be considered spherically symmetric with sufficient accuracy. The core radius is at least three times smaller than the radius of the corresponding atom (if we calculate the radii by the same methods). For heavy atoms, the core radius grows slightly with increasing number of electrons. The radius of the core of the heaviest naturally occurring element - uranium
Uranium is a chemical element; it has chemical symbol, symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Ura ...
- is comparable to the radius of a lithium atom, although the latter has only three electrons.
Chemical methods cannot separate the electrons of the core from the atom. When ionized by flame or ultraviolet
Ultraviolet radiation, also known as simply UV, is electromagnetic radiation of wavelengths of 10–400 nanometers, shorter than that of visible light, but longer than X-rays. UV radiation is present in sunlight and constitutes about 10% of ...
radiation, atomic cores, as a rule, also remain intact.
Core charge is a convenient way of explaining trends in the periodic table. Since the core charge increases as you move across a row of the periodic table
The periodic table, also known as the periodic table of the elements, is an ordered arrangement of the chemical elements into rows (" periods") and columns (" groups"). It is an icon of chemistry and is widely used in physics and other s ...
, the outer-shell electrons are pulled more and more strongly towards the nucleus and the atomic radius
The atomic radius of a chemical element is a measure of the size of its atom, usually the mean or typical distance from the center of the nucleus to the outermost isolated electron. Since the boundary is not a well-defined physical entity, there ...
decreases. This can be used to explain a number of periodic trends such as atomic radius, first ionization energy (IE), electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the ...
, and oxidizing
Redox ( , , reduction–oxidation or oxidation–reduction) is a type of chemical reaction in which the oxidation states of the reactants change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is ...
.
Core charge can also be calculated as 'atomic number' minus 'all electrons except those in the outer shell'. For example, chlorine
Chlorine is a chemical element; it has Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between ...
(element 17), with electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon ato ...
1s2 2s2 2p6 3s2 3p5, has 17 protons and 10 inner shell electrons (2 in the first shell, and 8 in the second) so:
:Core charge = 17 − 10 = +7
A core charge is the net charge of a nucleus, considering the completed shells of electrons to act as a 'shield.' As a core charge increases, the valence electrons are more strongly attracted to the nucleus, and the atomic radius
The atomic radius of a chemical element is a measure of the size of its atom, usually the mean or typical distance from the center of the nucleus to the outermost isolated electron. Since the boundary is not a well-defined physical entity, there ...
decreases across the period.
Relativistic effects
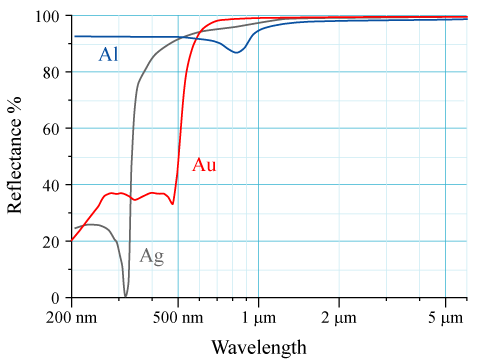
For elements with high atomic number ''Z'', relativistic effects can be observed for core electrons. The velocities of core s electrons reach relativistic momentum which leads to contraction of 6s orbitals relative to 5d orbitals. Physical properties affected by these relativistic effects include lowered melting temperature of mercury and the observed golden colour ofgold
Gold is a chemical element; it has chemical symbol Au (from Latin ) and atomic number 79. In its pure form, it is a brightness, bright, slightly orange-yellow, dense, soft, malleable, and ductile metal. Chemically, gold is a transition metal ...
and caesium
Caesium (IUPAC spelling; also spelled cesium in American English) is a chemical element; it has Symbol (chemistry), symbol Cs and atomic number 55. It is a soft, silvery-golden alkali metal with a melting point of , which makes it one of only f ...
due to narrowing of energy gap. Gold appears yellow because it absorbs blue light more than it absorbs other visible wavelengths of light and so reflects back yellow-toned light.

Electron transition
A core electron can be removed from its core-level upon absorption of electromagnetic radiation. This will either excite the electron to an empty valence shell or cause it to be emitted as a ''photoelectron'' due to thephotoelectric effect
The photoelectric effect is the emission of electrons from a material caused by electromagnetic radiation such as ultraviolet light. Electrons emitted in this manner are called photoelectrons. The phenomenon is studied in condensed matter physi ...
. The resulting atom will have an empty space in the core electron shell, often referred to as a ''core-hole''. It is in a metastable state and will decay within 10−15 s, releasing the excess energy via X-ray fluorescence
X-ray fluorescence (XRF) is the emission of characteristic "secondary" (or fluorescent) X-rays from a material that has been excited by being bombarded with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis ...
(as a characteristic X-ray) or by the Auger effect
The Auger effect (; ) or Meitner-Auger effect is a physical phenomenon in which atoms eject electrons. It occurs when an inner-shell vacancy in an atom is filled by an electron, releasing energy that causes the emission of another electron from a ...
. Detection of the energy emitted by a valence electron falling into a lower-energy orbital provides useful information on the electronic and local lattice structures of a material. Although most of the time this energy is released in the form of a ''photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless particles that can ...
'', the energy can also be transferred to another electron, which is ejected from the atom. This second ejected electron is called an Auger electron and this process of electronic transition with indirect radiation emission is known as the Auger effect.
Every atom except hydrogen has core-level electrons with well-defined binding energies. It is therefore possible to select an element to probe by tuning the X-ray energy to the appropriate absorption edge. The spectra of the radiation emitted can be used to determine the elemental composition of a material.
See also
*Atomic orbital
In quantum mechanics, an atomic orbital () is a Function (mathematics), function describing the location and Matter wave, wave-like behavior of an electron in an atom. This function describes an electron's Charge density, charge distribution a ...
*Auger effect
The Auger effect (; ) or Meitner-Auger effect is a physical phenomenon in which atoms eject electrons. It occurs when an inner-shell vacancy in an atom is filled by an electron, releasing energy that causes the emission of another electron from a ...
*Lanthanide contraction
The lanthanide contraction is the greater-than-expected decrease in atomic radii and ionic radii of the elements in the lanthanide series, from left to right. It is caused by the poor shielding effect of nuclear charge by the 4f electrons alo ...
*Relativistic quantum chemistry
Relativistic quantum chemistry combines relativistic mechanics with quantum chemistry to calculate elemental properties and structure, especially for the heavier elements of the periodic table. A prominent example is an explanation for the color of ...
*Shielding effect
In chemistry, the shielding effect sometimes referred to as atomic shielding or
electron shielding describes the attraction between an electron and the nucleus in any atom with more than one electron. The shielding effect can be defined as a r ...
* Surface core level shift
*Valence electron
In chemistry and physics, valence electrons are electrons in the outermost shell of an atom, and that can participate in the formation of a chemical bond if the outermost shell is not closed. In a single covalent bond, a shared pair forms with b ...
References
{{Electron configuration navbox Atomic physics Atomic, molecular, and optical physics Quantum chemistry