Birch reduction on:
[Wikipedia]
[Google]
[Amazon]
The Birch reduction or Metal-Ammonia reduction is an organic reaction that is used to convert  An example is the reduction of
An example is the reduction of 
 The reaction is known to be
The reaction is known to be  That step also determines the structure of the product. Although Arthur Birch originally argued that the protonation occurred at the ''meta'' position, subsequent investigation has revealed that protonation occurs at either the ''ortho'' or ''para'' position. Electron donors tend to induce ''ortho'' protonation, as shown in the reduction of
That step also determines the structure of the product. Although Arthur Birch originally argued that the protonation occurred at the ''meta'' position, subsequent investigation has revealed that protonation occurs at either the ''ortho'' or ''para'' position. Electron donors tend to induce ''ortho'' protonation, as shown in the reduction of 
 Solvated electrons will preferentially reduce sufficiently electronegative functional groups, such as
Solvated electrons will preferentially reduce sufficiently electronegative functional groups, such as
 Simple Hückel computations lead to equal electron densities at the three atoms 1, 3 and 5, but asymmetric bond orders. Modifying the exchange integrals to account for varying interatomic distances, produces maximum electron density at the central atom 1, a result confirmed by more modern RHF computations.
*
*
The result is analogous to conjugated enolates. When those anions (but not the enol
Simple Hückel computations lead to equal electron densities at the three atoms 1, 3 and 5, but asymmetric bond orders. Modifying the exchange integrals to account for varying interatomic distances, produces maximum electron density at the central atom 1, a result confirmed by more modern RHF computations.
*
*
The result is analogous to conjugated enolates. When those anions (but not the enol
 In substituted aromatics, an electron-withdrawing substituent, such as a
In substituted aromatics, an electron-withdrawing substituent, such as a 
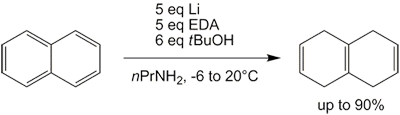 The directing effects of naphthalene substituents remain relatively unstudied theoretically. Substituents adjacent to the bridge appear to direct reduction to the unsubstituted ring; β substituents (one bond further) tend to direct reduction to the substituted ring.
The directing effects of naphthalene substituents remain relatively unstudied theoretically. Substituents adjacent to the bridge appear to direct reduction to the unsubstituted ring; β substituents (one bond further) tend to direct reduction to the substituted ring.
 For electron-donating substituents, Birch initially proposed ''meta'' attack, corresponding to the location of greatest electron density in a neutral benzene ring, a position endorsed by Krapcho and Bothner-By. These conclusions were challenged by Zimmerman in 1961, who computed electron densities of the radical and diene anions, revealing that the ''ortho'' site which was most negative and thus most likely to protonate. But the situation remained uncertain, because computations remained highly sensitive to transition geometry. Worse, Hückel orbital and unrestricted Hartree-Fock computations gave conflicting answers. Burnham, in 1969, concluded that the trustworthiest computations supported ''meta'' attack; Birch and Radom, in 1980, concluded that both ''ortho'' and ''meta'' substitutions would occur with a slight preference for ''ortho''.
*
*
In the earlier 1990s, Zimmerman and Wang developed an experiment technique to distinguish between ''ortho'' and ''meta'' protonation. The method began with the premise that carbanions are much more basic than the corresponding radical anions and thus protonate less selectively. Correspondingly, the two protonations in Birch reduction should exhibit an isotope effect: in a protium–deuterium medium, the radical anion should preferentially protonate and the carbanion deuterate. Indeed, a variety of methoxylated aromatics exhibited less ''ortho'' deuterium than ''meta'' (a 1:7 ratio). Moreover, modern electron density computations now firmly indicated ''ortho'' protonation; frontier orbital densities, most analogous to the traditional computations used in past studies, did not.
Although Birch remained reluctant to concede that ''ortho'' protonation was preferred as late as 1996,See diagrams in:
*
*
Zimmerman and Wang had won the day: modern textbooks unequivocally agree that electron-donating substituents promote ''ortho'' attack.
For electron-donating substituents, Birch initially proposed ''meta'' attack, corresponding to the location of greatest electron density in a neutral benzene ring, a position endorsed by Krapcho and Bothner-By. These conclusions were challenged by Zimmerman in 1961, who computed electron densities of the radical and diene anions, revealing that the ''ortho'' site which was most negative and thus most likely to protonate. But the situation remained uncertain, because computations remained highly sensitive to transition geometry. Worse, Hückel orbital and unrestricted Hartree-Fock computations gave conflicting answers. Burnham, in 1969, concluded that the trustworthiest computations supported ''meta'' attack; Birch and Radom, in 1980, concluded that both ''ortho'' and ''meta'' substitutions would occur with a slight preference for ''ortho''.
*
*
In the earlier 1990s, Zimmerman and Wang developed an experiment technique to distinguish between ''ortho'' and ''meta'' protonation. The method began with the premise that carbanions are much more basic than the corresponding radical anions and thus protonate less selectively. Correspondingly, the two protonations in Birch reduction should exhibit an isotope effect: in a protium–deuterium medium, the radical anion should preferentially protonate and the carbanion deuterate. Indeed, a variety of methoxylated aromatics exhibited less ''ortho'' deuterium than ''meta'' (a 1:7 ratio). Moreover, modern electron density computations now firmly indicated ''ortho'' protonation; frontier orbital densities, most analogous to the traditional computations used in past studies, did not.
Although Birch remained reluctant to concede that ''ortho'' protonation was preferred as late as 1996,See diagrams in:
*
*
Zimmerman and Wang had won the day: modern textbooks unequivocally agree that electron-donating substituents promote ''ortho'' attack.
arene
Aromatic compounds or arenes are organic compounds "with a chemistry typified by benzene" and "cyclically conjugated."
The word "aromatic" originates from the past grouping of molecules based on odor, before their general chemical properties were ...
s to 1,4-cyclohexadienes. The reaction is named after the Australian chemist Arthur Birch and involves the organic reduction
Organic reductions or organic oxidations or organic redox reactions are redox reactions that take place with organic compounds. In organic chemistry oxidations and reductions are different from ordinary redox reactions, because many reactions car ...
of aromatic ring
In organic chemistry, aromaticity is a chemical property describing the way in which a conjugated ring of unsaturated bonds, lone pairs, or empty orbitals exhibits a stabilization stronger than would be expected from conjugation alone. The e ...
s in an amine
In chemistry, amines (, ) are organic compounds that contain carbon-nitrogen bonds. Amines are formed when one or more hydrogen atoms in ammonia are replaced by alkyl or aryl groups. The nitrogen atom in an amine possesses a lone pair of elec ...
solvent
A solvent (from the Latin language, Latin ''wikt:solvo#Latin, solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a Solution (chemistry), solution. A solvent is usually a liquid but can also be a solid, a gas ...
(traditionally liquid ammonia
Ammonia is an inorganic chemical compound of nitrogen and hydrogen with the chemical formula, formula . A Binary compounds of hydrogen, stable binary hydride and the simplest pnictogen hydride, ammonia is a colourless gas with a distinctive pu ...
) with an alkali metal
The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K),The symbols Na and K for sodium and potassium are derived from their Latin names, ''natrium'' and ''kalium''; these are still the origins of the names ...
(traditionally sodium) and a proton
A proton is a stable subatomic particle, symbol , Hydron (chemistry), H+, or 1H+ with a positive electric charge of +1 ''e'' (elementary charge). Its mass is slightly less than the mass of a neutron and approximately times the mass of an e ...
source (traditionally an alcohol
Alcohol may refer to:
Common uses
* Alcohol (chemistry), a class of compounds
* Ethanol, one of several alcohols, commonly known as alcohol in everyday life
** Alcohol (drug), intoxicant found in alcoholic beverages
** Alcoholic beverage, an alco ...
). Unlike catalytic
Catalysis () is the increase in reaction rate, rate of a chemical reaction due to an added substance known as a catalyst (). Catalysts are not consumed by the reaction and remain unchanged after it. If the reaction is rapid and the catalyst ...
hydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to redox, reduce or Saturated ...
, Birch reduction does not reduce the aromatic ring all the way to a cyclohexane
Cyclohexane is a cycloalkane with the molecular formula . Cyclohexane is non-polar. Cyclohexane is a colourless, flammable liquid with a distinctive detergent-like odor, reminiscent of cleaning products (in which it is sometimes used). Cyclohexan ...
.
naphthalene
Naphthalene is an organic compound with formula . It is the simplest polycyclic aromatic hydrocarbon, and is a white Crystal, crystalline solid with a characteristic odor that is detectable at concentrations as low as 0.08 Parts-per notation ...
in ammonia and ethanol: 
Reaction mechanism and regioselectivity
A solution of sodium in liquid ammonia consists of the intensely blueelectride
An electride is an ionic compound in which an electron serves the role of the anion. Solutions
Solutions of alkali metals in ammonia are electride salts. In the case of sodium, these blue solutions consist of a(NH3)6sup>+ and solvated electron ...
salt a(NH3)xsup>+ e−. The solvated electrons add to the aromatic ring to give a radical anion
In organic chemistry, a radical anion is a free radical species that carries a negative charge. Radical anions are encountered in organic chemistry as reduced derivatives of polycyclic aromatic compounds, e.g. sodium naphthenide. An example of a ...
, which then abstracts a proton from the alcohol. The process then repeats at either the ''ortho'' or ''para'' position (depending on substituents) to give the final diene. The residual double bonds do not stabilize further radical additions.
third order
The term third order signifies, in general, lay members of Christian religious orders, who do not necessarily live in a religious community such as a monastery or a nunnery, and yet can claim to wear the religious habit and participate in the goo ...
– first order in the aromatic, first order in the alkali metal, and first order in the alcohol. This requires that the rate-limiting step be the conversion of radical anion B to the cyclohexadienyl radical C.
anisole
Anisole, or methoxybenzene, is an organic compound with the formula . It is a colorless liquid with a smell reminiscent of anise seed, and in fact many of its derivatives are found in natural and artificial fragrances. The compound is mainly ...
(1). Electron-withdrawing substituents tend to induce ''para'' protonation, as shown in the reduction of benzoic acid
Benzoic acid () is a white (or colorless) solid organic compound with the formula , whose structure consists of a benzene ring () with a carboxyl () substituent. The benzoyl group is often abbreviated "Bz" (not to be confused with "Bn," which ...
(2).
ketones
In organic chemistry, a ketone is an organic compound with the structure , where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond C=O). The simplest ketone is acetone ( ...
or nitro groups, but do not attack alcohols
In chemistry, an alcohol (), is a type of organic compound that carries at least one hydroxyl () functional group bound to a Saturated and unsaturated compounds, saturated carbon atom. Alcohols range from the simple, like methanol and ethanol ...
, carboxylic acids
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an Substituent, R-group. The general formula of a carboxylic acid is often written as or , sometimes as with R referring to an organyl ...
, or ethers.
Secondary protonation regioselectivity
The second reduction and protonation also poses mechanistic questions. Thus there are three resonance structures for the carbanion (labeled B, C and D in the picture).tautomer
In chemistry, tautomers () are structural isomers (constitutional isomers) of chemical compounds that readily interconvert.
The chemical reaction interconverting the two is called tautomerization. This conversion commonly results from the reloca ...
) kinetically protonate, they do so at the center to afford the β,γ-unsaturated carbonyl.
Modifications
Traditional Birch reduction requirescryogenic
In physics, cryogenics is the production and behaviour of materials at very low temperatures.
The 13th International Institute of Refrigeration's (IIR) International Congress of Refrigeration (held in Washington, DC in 1971) endorsed a univers ...
temperatures to liquify ammonia and pyrophoric
A substance is pyrophoric (from , , 'fire-bearing') if it ignites spontaneously in air at or below (for gases) or within 5 minutes after coming into contact with air (for liquids and solids). Examples are organolithium compounds and triethylb ...
alkali-metal electron donors. Variants have developed to reduce either inconvenience.
Many amines serve as alternative solvents: for example, bis(methoxymethyl)amine in THF or mixed ''n''-propylamine and ethylenediamine
Ethylenediamine (abbreviated as en when a ligand) is the organic compound with the formula C2H4(NH2)2. This colorless liquid with an ammonia-like odor is a basic amine. It is a widely used building block in chemical synthesis, with approximately ...
. Pure secondary and tertiary amines, however, fail to dissolve alkali metals.
To avoid direct alkali, there are chemical alternatives, such as M-SG reducing agent. The reduction can also be powered by an external potential or sacrificial anode (magnesium or aluminum), but then alkali metal salts are necessary to colocate the reactants via complexation.
Birch alkylation
In Birch alkylation theanion
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conven ...
formed in the Birch reduction is trapped by a suitable electrophile
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively Electric charge, charged, have an ...
such as a haloalkane
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents of hydrogen atom. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalka ...
, for example:
:carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an Substituent, R-group. The general formula of a carboxylic acid is often written as or , sometimes as with R referring to an organyl ...
, will stabilize the carbanion
In organic chemistry, a carbanion is an anion with a lone pair attached to a tervalent carbon atom. This gives the carbon atom a negative charge.
Formally, a carbanion is the conjugate base of a carbon acid:
:
where B stands for the base (chemist ...
to generate the least-substituted olefin
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as α-olefins.
The International Union of Pu ...
; an electron-donating substituent has the opposite effect.
:
Benkeser reduction
The Benkeser reduction is thehydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to redox, reduce or Saturated ...
of polycyclic aromatic hydrocarbon
A Polycyclic aromatic hydrocarbon (PAH) is any member of a class of organic compounds that is composed of multiple fused aromatic rings. Most are produced by the incomplete combustion of organic matter— by engine exhaust fumes, tobacco, incine ...
s, especially naphthalene
Naphthalene is an organic compound with formula . It is the simplest polycyclic aromatic hydrocarbon, and is a white Crystal, crystalline solid with a characteristic odor that is detectable at concentrations as low as 0.08 Parts-per notation ...
s using lithium
Lithium (from , , ) is a chemical element; it has chemical symbol, symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard temperature and pressure, standard conditions, it is the least dense metal and the ...
or calcium
Calcium is a chemical element; it has symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to it ...
metal
A metal () is a material that, when polished or fractured, shows a lustrous appearance, and conducts electrical resistivity and conductivity, electricity and thermal conductivity, heat relatively well. These properties are all associated wit ...
in low molecular weight alkyl amine
In chemistry, amines (, ) are organic compounds that contain carbon-nitrogen bonds. Amines are formed when one or more hydrogen atoms in ammonia are replaced by alkyl or aryl groups. The nitrogen atom in an amine possesses a lone pair of elec ...
s solvents. Unlike traditional Birch reduction, the reaction can be conducted at temperatures higher than the boiling point of ammonia (−33 °C).
For the reduction of naphthalene with lithium in a mixed ethylamine
Ethylamine, also known as ethanamine, is an organic compound with the chemical formula, formula CH3CH2NH2. This colourless gas has a strong ammonia-like odor. It condenses just below room temperature to a liquid miscibility, miscible with virtual ...
-dimethylamine
Dimethylamine is an organic compound with the formula (CH3)2NH. This secondary amine is a colorless, flammable gas with an ammonia-like odor. Dimethylamine is commonly encountered commercially as a solution in water at concentrations up to around ...
solution, the principal products are bicyclo .3.0ec-(1,9)-ene, bicyclo .3.0ec-(1,2)-ene and bicyclo .3.0ecane.
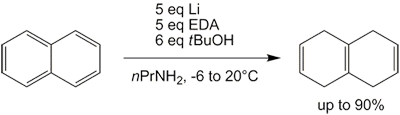 The directing effects of naphthalene substituents remain relatively unstudied theoretically. Substituents adjacent to the bridge appear to direct reduction to the unsubstituted ring; β substituents (one bond further) tend to direct reduction to the substituted ring.
The directing effects of naphthalene substituents remain relatively unstudied theoretically. Substituents adjacent to the bridge appear to direct reduction to the unsubstituted ring; β substituents (one bond further) tend to direct reduction to the substituted ring.
History
Arthur Birch, building on earlier (1937) work by Wooster and Godfrey who used water, developed the reaction in the 1940s while working in the Dyson Perrins Laboratory at theUniversity of Oxford
The University of Oxford is a collegiate university, collegiate research university in Oxford, England. There is evidence of teaching as early as 1096, making it the oldest university in the English-speaking world and the List of oldest un ...
.
*
*
*
*
*
*
Birch's original procedure used sodium
Sodium is a chemical element; it has Symbol (chemistry), symbol Na (from Neo-Latin ) and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 element, group 1 of the peri ...
and ethanol
Ethanol (also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound with the chemical formula . It is an Alcohol (chemistry), alcohol, with its formula also written as , or EtOH, where Et is the ps ...
, Alfred L. Wilds later discovered that lithium gives better yields.
The reaction was difficult to understand mechanistically, with controversy lasting into the 1990s.
The case with electron-withdrawing groups is obvious, because the Birch alkylation serves as a trap for the penultimate dianion D. This dianion appears even in alcohol-free reactions. Thus the initial protonation is ''para'' rather than ''ipso'', as seen in the B-C transformation.
Additional reading
*See also
* Solvated electron — the reducing agent * Bouveault–Blanc reduction — another reaction using solvated electrons * Synthesis of methamphetamine — an applicationReferences
{{DEFAULTSORT:Birch Reduction Organic reduction reactions Organic redox reactions Name reactions