Bartell Mechanism on:
[Wikipedia]
[Google]
[Amazon]
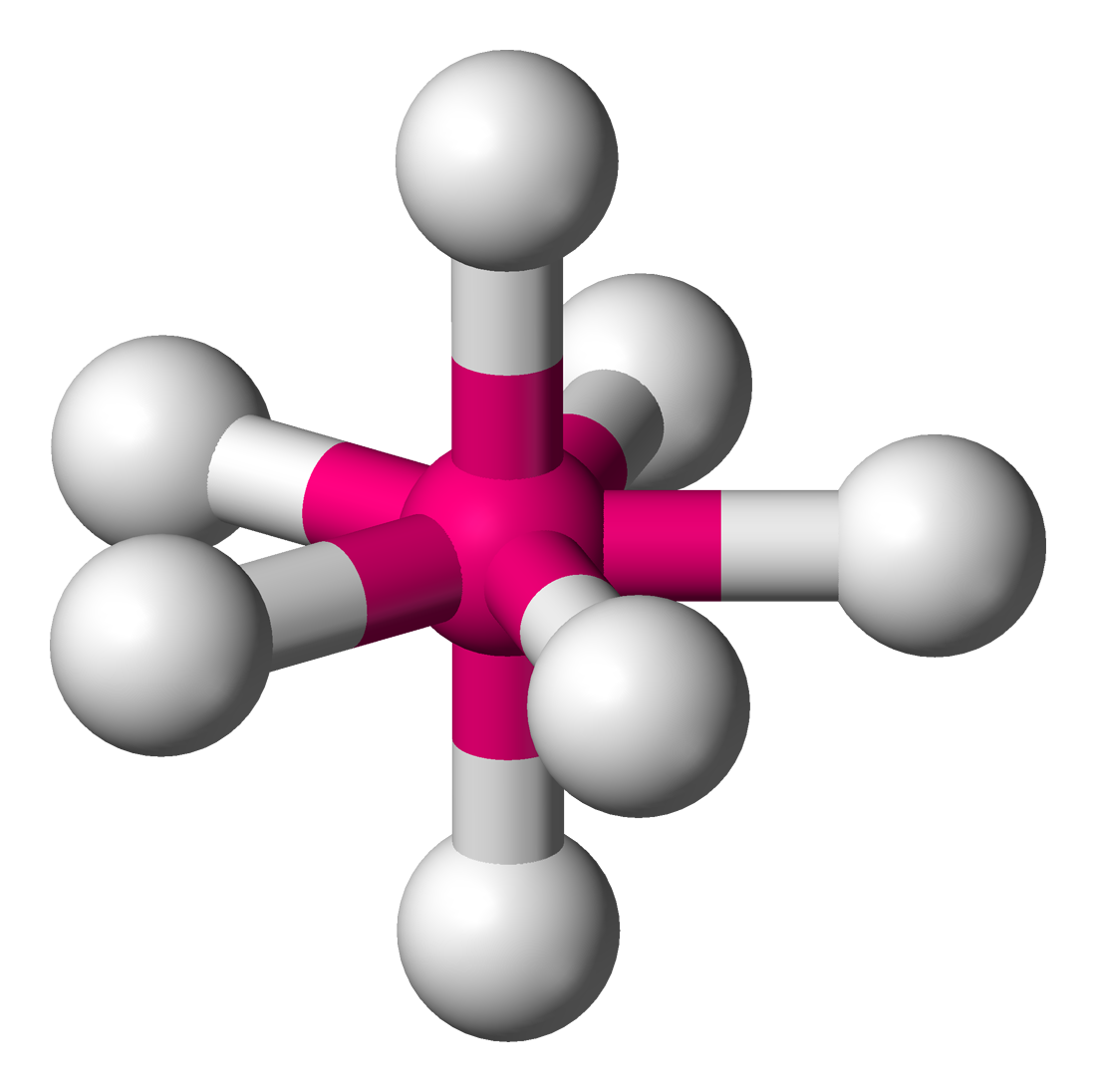 The Bartell mechanism is a
The Bartell mechanism is a
, accessed 28 May 2014 of other mechanisms; it displays characteristics of the Berry mechanism, a "lever" mechanism seen in pseudorotation of Seesaw (chemistry), disphenoidal molecules, and a "turnstile" mechanism (which can be seen in
 The Bartell mechanism is a
The Bartell mechanism is a pseudorotation
In chemistry, a pseudorotation is a set of intramolecular movements of attached groups (i.e., ligands) on a highly symmetric molecule, leading to a molecule indistinguishable from the initial one. The International Union of Pure and Applied Chem ...
al mechanism similar to the Berry mechanism
The Berry mechanism, or Berry pseudorotation mechanism, is a type of vibration causing molecules of certain geometries to isomerization, isomerize by exchanging the two axial ligands (see the figure) for two of the equatorial ones. It is the most ...
. It occurs only in molecules with a pentagonal bipyramidal molecular geometry, such as IF7. This mechanism was first predicted by H. B. Bartell. The mechanism exchanges the axial atoms with one pair of the equatorial atoms with an energy requirement of about 2.7 kcal/mol. Similarly to the Berry mechanism in square planar molecules, the symmetry of the intermediary phase of the vibrational mode is "chimeric"LS Bartell, MJ Rothman & A Gavezzotti, 1982, ''J. Chem. Phys.'' 76:4136-4413.M Cass, KK Hii & HS Rzepa, 2005, "Mechanisms that interchange axial and equatorial atoms in fluxional processes: Illustration of the Berry pseudorotation, the turnstile and the lever mechanisms via animation of transition state normal vibrational modes", ''J. Chem. Educ.'' (online), 2005; se, accessed 28 May 2014 of other mechanisms; it displays characteristics of the Berry mechanism, a "lever" mechanism seen in pseudorotation of Seesaw (chemistry), disphenoidal molecules, and a "turnstile" mechanism (which can be seen in
trigonal bipyramidal
In chemistry, a trigonal bipyramid formation is a molecular geometry with one atom at the center and 5 more atoms at the corners of a triangular bipyramid. This is one geometry for which the bond angles surrounding the central atom are not identi ...
molecules under certain conditions).
References
See also
*Pseudorotation
In chemistry, a pseudorotation is a set of intramolecular movements of attached groups (i.e., ligands) on a highly symmetric molecule, leading to a molecule indistinguishable from the initial one. The International Union of Pure and Applied Chem ...
* Bailar twist
* Berry mechanism
The Berry mechanism, or Berry pseudorotation mechanism, is a type of vibration causing molecules of certain geometries to isomerization, isomerize by exchanging the two axial ligands (see the figure) for two of the equatorial ones. It is the most ...
* Ray–Dutt twist
The Ray–Dutt twist is a mechanism proposed for the racemization of octahedral complexes containing three bidentate chelate rings. Such complexes typically adopt an octahedral molecular geometry in their ground states, in which case they possess ...
* Fluxional molecule
In chemistry and molecular physics, fluxional (or non-rigid) molecules are molecules that undergo dynamics such that some or all of their atoms interchange between symmetry-equivalent positions. Because virtually all molecules are fluxional in som ...
Molecular geometry
Chemical kinetics
{{molecular-geometry-stub