|
Trispyrazolylborate
The trispyrazolylborate ligand, abbreviated Tp−, is an anionic Denticity, tridentate and tripodal ligand. Trispyrazolylborate refers specifically to the anion [HB(C3N2H3)3]−. However, the term can also be used to refer to derivatives having substituents on the pyrazolyl rings. This class of compounds belongs to the family of ligands called scorpionate ligands. Tp ligands As suggested by the resonance structures, the nitrogen centers that are not bonded to boron are basic. These centers bind to three adjacent sites of a metal such that the simple adducts have C3v symmetry group, symmetry. The facial bonding mode is reminiscent of cyclopentadienyl ligands, although the ligand field stabilization energy of Tp− is weaker as indicated by the fact that Fe(Tp)2 is a spin crossover, spin-crossover complex whereas ferrocene is low-spin. The Tp ligands are usually prepared from the reaction of pyrazole with potassium borohydride: :KBH4 + 3 C3H3N2H → K[HB(C3N2H3)3] + 3H2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tripodal Ligand
Tripodal ligands are tridentate, tri- and tetradentate ligands. They are popular in research in the areas of coordination chemistry and homogeneous catalysis. Because the ligands are polydentate, they do not readily dissociate from the metal centre. Many tripodal ligands have C3 symmetry group, symmetry. Coordination chemistry In their coordination complexes with an octahedral molecular geometry the tridentate tripod ligands occupy one face, leading to a fixed Coordination complexes#Cis–trans isomerism and facial–meridional isomerism, facial (or ''fac'') geometry. The tetradentate tripodal ligands occupy four contiguous sites, leaving two ''cis'' positions available on the octahedral metal center. When bound to four- and five-coordinate metal centres, these ligands impose C3 symmetry, which can lead to uncommon ligand field splitting patterns. Tripodal ligands are often able to coordinately saturate metal ions with lower coordination numbers. One tripodal ligand of commercial ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Scorpionate Ligand
In coordination chemistry, a scorpionate ligand is a tridentate (three-donor-site) ligand that binds to a central atom in a ''fac'' manner. The most popular class of scorpionates are the hydrotris(pyrazolyl)borates or Tp ligands. These were also the first to become popular. These ligands first appeared in journals in 1966 from the then little-known DuPont chemist of Ukrainian descent, Swiatoslaw Trofimenko. Trofimenko called this discovery "a new and fertile field of remarkable scope". The term scorpionate comes from the fact that the ligand can bind a metal with two donor sites like the pincers of a scorpion; the third and final donor site reaches over the plane formed by the metal and the other two donor atoms to bind to the metal. The binding can be thought of as being like a scorpion grabbing the metal with two pincers before stinging it. While many scorpionate ligands are of the Tp class, many other scorpionate ligands are known. For example, the Tm and tripodal phosphine ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Tris(3,5-dimethyl-1-pyrazolyl)borate
Potassium tris(3,5-dimethyl-1-pyrazolyl)borate, abbreviated KTp*, is the potassium salt of the anion HB((CH3)2C3N2H)3. Tp*− is a tripodal ligand that binds to a metal in a facial manner, more specifically a Scorpionate ligand. KTp* is a white crystalline solid that is soluble in polar solvents, including water and several alcohols. Synthesis KTp* is synthesized in a manner similar to that of KTp by the reaction of potassium borohydride and 3,5-dimethylpyrazole. Hydrogen gas is evolved as each of the pyrazole reacts at the boron. The rate of B-N bond formation becomes more difficult with each successive 3,5-dimethylpyrazolyl due to the increase in steric hindrance around the boron: :3 Me2C3N2H2 + KBH4 → KHB(Me2C3N2H)3 + 3 H2 The required dimethylpyrazole is obtained by condensation of hydrazine and acetylacetone Acetylacetone is an organic compound with the chemical formula . It is classified as a 1,3-diketone. It exists in equilibrium with a tautomer . The mixture is a col ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bioinorganic Chemistry
Bioinorganic chemistry is a field that examines the role of metals in biology. Bioinorganic chemistry includes the study of both natural phenomena such as the behavior of metalloproteins as well as artificially introduced metals, including those that are non-essential, in medicine and toxicology. Many biological processes such as respiration depend upon molecules that fall within the realm of inorganic chemistry. The discipline also includes the study of inorganic models or mimics that imitate the behaviour of metalloproteins. As a mix of biochemistry and inorganic chemistry, bioinorganic chemistry is important in elucidating the implications of electron-transfer proteins, substrate bindings and activation, atom and group transfer chemistry as well as metal properties in biological chemistry. The successful development of truly interdisciplinary work is necessary to advance bioinorganic chemistry. Composition of living organisms About 99% of mammals' mass are the elements carb ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetraethylammonium
Tetraethylammonium (TEA) is a quaternary ammonium cation with the chemical formula , consisting of four ethyl groups (, denoted Et) attached to a central nitrogen atom. It is a counterion used in the research laboratory to prepare lipophilic salts of inorganic anions. It is used similarly to tetrabutylammonium, the difference being that its salts are less lipophilic, more easily crystallized and more Toxicity, toxic. Preparation The halide salt is prepared by the reaction of triethylamine and an ethyl halide: : This method works well for the preparation of tetraethylammonium iodide (where X = I). Most tetraethylammonium salts are prepared by salt metathesis reactions. For example, the synthesis of tetraethylammonium perchlorate, a salt that has been useful as a supporting electrolyte for polarography, polarographic studies in non-aqueous solvents, is carried out by mixing the water-soluble salts tetraethylammonium bromide and sodium perchlorate in water, from which the water-in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydride
In chemistry, a hydride is formally the anion of hydrogen (H−), a hydrogen ion with two electrons. In modern usage, this is typically only used for ionic bonds, but it is sometimes (and has been more frequently in the past) applied to all chemical compound, compounds containing covalent bond, covalently bound H atoms. In this broad and potentially archaic sense, water (H2O) is a hydride of oxygen, ammonia is a hydride of nitrogen, etc. In covalent compounds, it implies hydrogen is attached to a less electronegative chemical element, element. In such cases, the H centre has nucleophilic character, which contrasts with the protic character of acids. The hydride anion is very rarely observed. Almost all of the elements form Binary compounds of hydrogen, binary compounds with hydrogen, the exceptions being helium, He, neon, Ne, argon, Ar, krypton, Kr, promethium, Pm, osmium, Os, iridium, Ir, radon, Rn, francium, Fr, and radium, Ra. exotic atom#exotic molecules, Exotic molecules ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allyl
In organic chemistry, an allyl group is a substituent with the structural formula . It consists of a methylene bridge () attached to a vinyl group (). The name is derived from the scientific name for garlic, . In 1844, Theodor Wertheim isolated an allyl derivative from garlic oil and named it "". The term allyl applies to many compounds related to , some of which are of practical or of everyday importance, for example, allyl chloride. Allylation is any chemical reaction that adds an allyl group to a Substrate (chemistry), substrate. Nomenclature A site adjacent to the unsaturated carbon atom is called the allylic position or allylic site. A group attached at this site is sometimes described as allylic. Thus, "has an allylic hydroxyl group". Allylic Carbon–hydrogen bond, C−H bonds are about 15% weaker than the C−H bonds in ordinary Orbital hybridisation, sp3 carbon centers and are thus more reactive. Benzylic and allylic are related in terms of structure, bond strength ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrosyl
In organic chemistry, nitroso refers to a functional group in which the nitric oxide () group is attached to an organic moiety. As such, various nitroso groups can be categorized as ''C''-nitroso compounds (e.g., nitrosoalkanes; ), ''S''-nitroso compounds ( nitrosothiols; ), ''N''-nitroso compounds (e.g., nitrosamines, ), and ''O''-nitroso compounds ( alkyl nitrites; ). Synthesis Nitroso compounds can be prepared by the reduction of nitro compounds or by the oxidation of hydroxylamines. Ortho-nitrosophenols may be produced by the Baudisch reaction. In the Fischer–Hepp rearrangement, aromatic 4-nitrosoanilines are prepared from the corresponding nitrosamines. Properties Nitrosoarenes typically participate in a monomer–dimer equilibrium. The azobenzene ''N'',''N'-''dioxide (Ar(–O)N+=+N(O–)Ar) dimers, which are often pale yellow, are generally favored in the solid state, whereas the deep-green monomers are favored in dilute solution or at higher temperatures. The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
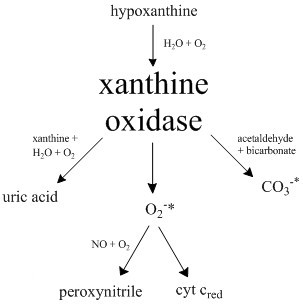
Xanthine Oxidase
Xanthine oxidase (XO or XAO) is a form of xanthine oxidoreductase, a type of enzyme that generates reactive oxygen species. These enzymes catalyze the oxidation of hypoxanthine to xanthine and can further catalyze the oxidation of xanthine to uric acid. These enzymes play an important role in the catabolism of purines in some species, including humans. Xanthine oxidase is defined as an ''enzyme activity'' (EC 1.17.3.2). The same protein, which in humans has the HGNC approved gene symbol ''XDH'', can also have xanthine dehydrogenase activity (EC 1.17.1.4). Most of the protein in the liver exists in a form with xanthine dehydrogenase activity, but it can be converted to xanthine oxidase by reversible sulfhydryl oxidation or by irreversible proteolytic modification. "XDH xanthine dehydrogenase" Reaction The following chemical reactions are catalyzed by xanthine oxidase: * hypoxanthine + H2O + O2 xanthine + H2O2 * xanthine + H2O + O2 uric acid + H2O2 * Xanthine oxidase ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hemerythrin
Hemerythrin (also spelled haemerythrin; , ) is an oligomeric protein responsible for oxygen (O2) transport in the marine invertebrate phyla of sipunculids, priapulids, brachiopods, and in a single annelid worm genus, '' Magelona''. Myohemerythrin is a monomeric O2-binding protein found in the muscles of marine invertebrates. Hemerythrin and myohemerythrin are essentially colorless when deoxygenated, but turn a violet-pink in the oxygenated state. Hemerythrin does not, as the name might suggest, contain a heme. The names of the blood oxygen transporters hemoglobin, hemocyanin, and hemerythrin do not refer to the heme group (only found in globins). Instead, these names are derived from the Greek word for blood. Hemerythrin may also contribute to innate immunity and anterior tissue regeneration in certain worms. O2 binding mechanism The mechanism of dioxygen binding is unusual. Most O2 carriers operate via formation of dioxygen complexes, but hemerythrin holds the O2 as a hydro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetonitrile
Acetonitrile, often abbreviated MeCN (methyl cyanide), is the chemical compound with the formula and structure . This colourless liquid is the simplest organic nitrile (hydrogen cyanide is a simpler nitrile, but the cyanide anion is not classed as organic). It is produced mainly as a byproduct of acrylonitrile manufacture. It is used as a polar aprotic solvent in organic synthesis and in the purification of butadiene. The skeleton is linear with a short distance of 1.16 Å. Acetonitrile was first prepared in 1847 by the French chemist Jean-Baptiste Dumas. Applications Acetonitrile is used mainly as a solvent in the purification of butadiene in refineries. Specifically, acetonitrile is fed into the top of a distillation column filled with hydrocarbons including butadiene, and as the acetonitrile falls down through the column, it absorbs the butadiene which is then sent from the bottom of the tower to a second separating tower. Heat is then employed in the separa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |