|
Transition Metal Thiosulfate Complex
A transition metal thiosulfate complex is a coordination complex containing one or more thiosulfate ligands. Thiosulfate occurs in nature and is used industrially, so its interactions with metal ions are of some practical interest. Three binding modes are common: monodentate (κ1-), ''O'',''S''-bidentate (κ2-),and bridging (μ-). Typically, thiosulfate complexes are prepared from thiosulfate salts. In some cases, they arise by oxidation of polysulfido complexes, or by binding of sulfur trioxide to sulfido ligands. Applications Silver-thiosulfate complexes are produced by common photographic fixers. By dissolving silver halides the fixer stabilises the image. The dissolution process entails reactions involving the formation of 1:2 and 1:3 complexes (X = halide): Fixation involves these chemical reactions (X = halide, typically ):Karlheinz Keller et al. "Photography" in Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. : : Sodium aurothiosulfate dihydrat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coordination Complex
A coordination complex consists of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of bound molecules or ions, that are in turn known as ''ligands'' or complexing agents. Many metal-containing compounds, especially those that include transition metals (elements like titanium that belong to the Periodic Table's d-block), are coordination complexes. Nomenclature and terminology Coordination complexes are so pervasive that their structures and reactions are described in many ways, sometimes confusingly. The atom within a ligand that is bonded to the central metal atom or ion is called the donor atom. In a typical complex, a metal ion is bonded to several donor atoms, which can be the same or different. A polydentate (multiple bonded) ligand is a molecule or ion that bonds to the central atom through several of the ligand's atoms; ligands with 2, 3, 4 or even 6 bonds to the central atom are common. These compl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiosulfate
Thiosulfate ( IUPAC-recommended spelling; sometimes thiosulphate in British English) is an oxyanion of sulfur with the chemical formula . Thiosulfate also refers to the compounds containing this anion, which are the salts of thiosulfuric acid, e.g. sodium thiosulfate . Thiosulfate also refers to the esters of thiosulfuric acid, e.g. ''O'',''S''-dimethyl thiosulfate . The prefix thio- indicates that the thiosulfate is a sulfate with one oxygen replaced by sulfur. Thiosulfate is tetrahedral at the central S atom. Thiosulfate salts occur naturally. Thiosulfate ion has C3v symmetry, and is produced by certain biochemical processes. It rapidly dechlorinates water and is notable for its use to halt bleaching in the paper-making industry. Thiosulfate salts are mainly used in dying in textiles and the bleaching of natural substances. Sodium thiosulfate, commonly called ''hypo'' (from "hyposulfite"), was widely used in photography to fix black and white negatives and prints after the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
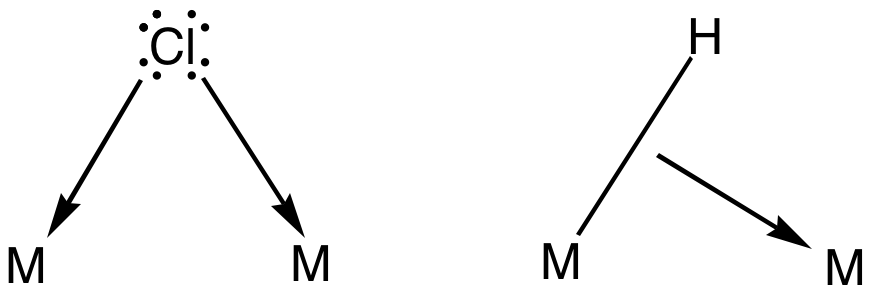
Bridging Ligand
In coordination chemistry, a bridging ligand is a ligand that connects two or more atoms, usually metal ions. The ligand may be atomic or polyatomic. Virtually all complex organic compounds can serve as bridging ligands, so the term is usually restricted to small ligands such as pseudohalides or to ligands that are specifically designed to link two metals. In naming a complex wherein a single atom bridges two metals, the bridging ligand is preceded by the Greek letter mu, μ, with a subscript number denoting the number of metals bound to the bridging ligand. μ2 is often denoted simply as μ. When describing coordination complexes care should be taken not to confuse μ with η ('eta'), which relates to hapticity. Ligands that are not bridging are called terminal ligands. List of bridging ligands Virtually all ligands are known to bridge, with the exception of amines and ammonia. Common bridging ligands include most of the common anions. Many simple organic ligands form ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
MS2O3
MS, ms, Ms, M.S., etc. may refer to: Arts and entertainment * ''Ms.'' (magazine), an American feminist magazine * Metal Storm (webzine), a heavy metal website based in Estonia Businesses and organizations * MS-13, criminal gang * Missionaries of La Salette, a Catholic male religious order * Młodzi Socjaliści (Young Socialists), a former Polish socialist youth organization * Morgan Stanley, a US investment bank (NYSE stock symbol: MS) * Mjólkursamsalan, an Icelandic dairy company Educational qualifications * Master of Science, a master's degree in the field of science * Master of Surgery, an advanced medical degree * Master Sommelier, a terminal degree in the field of wine * '' Mastère spécialisé'', a French postgraduate ''grande école'' master's degree Medicine * Mitral stenosis, narrowing of the mitral valve of the heart * Morphine sulfate, an opiate pain-relieving drug * Multiple sclerosis, a disease of the nervous system Military * Master seaman, a non-commissioned ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photographic Fixer
Photographic fixer is a mix of chemicals used in the final step in the photographic processing of film or paper. The fixer stabilises the image, removing the unexposed silver halide remaining on the photographic film or photographic paper, leaving behind the reduced metallic silver that forms the image. By fixation, the film or paper is insensitive to further action by light. Without fixing, the remaining silver halide would darken and cause fogging of the image. Fixation is commonly achieved by treating the film or paper with a solution of thiosulfate salt. Popular salts are sodium thiosulfate—commonly called hypo—and ammonium thiosulfate—commonly used in modern rapid fixer formulae. Fixation involves these chemical reactions (X = halide, typically Br−):Karlheinz Keller et al. "Photography" in Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. :AgX + 2 S2O32− → g(S2O3)2sup>3− + X− :AgX + 3 S2O32− → g(S2O3)3sup ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silver Halide
A silver halide (or silver salt) is one of the chemical compounds that can form between the Chemical element, element silver (Ag) and one of the halogens. In particular, bromine (Br), chlorine (Cl), iodine (I) and fluorine (F) may each combine with silver to produce silver bromide (AgBr), silver chloride (AgCl), silver iodide (AgI), and three forms of silver fluoride, respectively. As a group, they are often referred to as the silver halides, and are often given the pseudo-chemical notation AgX. Although most silver halides involve silver atoms with oxidation states of +1 (Ag+), silver halides in which the silver atoms have oxidation states of +2 (Ag2+) are known, of which silver(II) fluoride is the only known stable one. Silver halides are light-sensitive chemicals, and are commonly used in photographic film and paper. Applications Light sensitivity Silver halides are used in photographic film and photographic paper, including graphic art film and paper, where silver halide ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halide
In chemistry, a halide (rarely halogenide) is a binary chemical compound, of which one part is a halogen atom and the other part is an element or radical that is less electronegative (or more electropositive) than the halogen, to make a fluoride, chloride, bromide, iodide, astatide, or theoretically tennesside compound. The alkali metals combine directly with halogens under appropriate conditions forming halides of the general formula, MX (X = F, Cl, Br or I). Many salts are halides; the ''hal-'' syllable in ''halide'' and ''halite'' reflects this correlation. All Group 1 metals form halides that are white solids at room temperature. A halide ion is a halogen atom bearing a negative charge. The halide anions are fluoride (), chloride (), bromide (), iodide () and astatide (). Such ions are present in all ionic halide salts. Halide minerals contain halides. All these halides are colourless, high melting crystalline solids having high negative enthalpies of form ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Aurothiosulfate
Sodium aurothiosulfate, or sanocrysin, is the inorganic compound with the formula . This salt contains an anionic coordination complex of gold(I) bound to two thiosulfate ligands. It is colorless. History The compound was first synthesized in 1845 by Mathurin-Joseph Fordos and A. Gélis who were researching chemicals used in the Daguerrotype photographic process. It then came to be called Fordos and Gélis salt. It went out of interest until 1924 when it was noted as a chemotherapeutic agent for tuberculosis by Holger Møllgaard in Copenhagen. Other methods of synthesis were then identified. Potential applications Like several other gold compounds, this species is used as an antirheumatic. The first placebo-controlled trial was probably conducted in 1931, when sanocrysin was compared with distilled water for the treatment of tuberculosis. Aurothiosulfate complexes have been discussed in the context of the extraction of gold Gold is a chemical element with the symbol Au ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gold
Gold is a chemical element with the symbol Au (from la, aurum) and atomic number 79. This makes it one of the higher atomic number elements that occur naturally. It is a bright, slightly orange-yellow, dense, soft, malleable, and ductile metal in a pure form. Chemically, gold is a transition metal and a group 11 element. It is one of the least reactive chemical elements and is solid under standard conditions. Gold often occurs in free elemental (native state), as nuggets or grains, in rocks, veins, and alluvial deposits. It occurs in a solid solution series with the native element silver (as electrum), naturally alloyed with other metals like copper and palladium, and mineral inclusions such as within pyrite. Less commonly, it occurs in minerals as gold compounds, often with tellurium ( gold tellurides). Gold is resistant to most acids, though it does dissolve in aqua regia (a mixture of nitric acid and hydrochloric acid), forming a soluble tetrachloroau ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyanide
Cyanide is a naturally occurring, rapidly acting, toxic chemical that can exist in many different forms. In chemistry, a cyanide () is a chemical compound that contains a functional group. This group, known as the cyano group, consists of a carbon atom triple-bonded to a nitrogen atom. In inorganic cyanides, the cyanide group is present as the anion . Soluble salts such as sodium cyanide (NaCN) and potassium cyanide (KCN) are highly toxic. Hydrocyanic acid, also known as hydrogen cyanide, or HCN, is a highly volatile liquid that is produced on a large scale industrially. It is obtained by acidification of cyanide salts. Organic cyanides are usually called nitriles. In nitriles, the group is linked by a covalent bond to carbon. For example, in acetonitrile (), the cyanide group is bonded to methyl (). Although nitriles generally do not release cyanide ions, the cyanohydrins do and are thus rather toxic. Bonding The cyanide ion is isoelectronic with carbon m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IUPAC Red Book
Nomenclature of Inorganic Chemistry, IUPAC Recommendations 2005 is the 2005 version of '' Nomenclature of Inorganic Chemistry'' (which is informally called the Red Book). It is a collection of rules for naming inorganic compounds, as recommended by the International Union of Pure and Applied Chemistry (IUPAC). Summary The 2005 edition replaces their previous recommendations ''Nomenclature The Red Book of Inorganic Chemistry, IUPAC Recommendations 1990 (Red Book I)'', and "where appropriate" (sic) ''Nomenclature of Inorganic Chemistry II, IUPAC Recommendations 2000 (Red Book II)''. The recommendations take up over 300 pages''Nomenclature of Inorganic Chemistry IUPAC Recommendations'' 2005 ed. N. G. Connelly et al. RSC Publishing https://iupac.org/what-we-do/books/redbook/ and the full text can be downloaded from IUPAC. Corrections have been issued. Apart from a reorganisation of the content, there is a new section on organometallics and a formal element list to be used in place o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coordination Chemistry
A coordination complex consists of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of chemical bond, bound molecules or ions, that are in turn known as ''ligands'' or complexing agents. Many metal-containing chemical compound, compounds, especially those that include transition metals (elements like titanium that belong to the Block (periodic table), Periodic Table's d-block), are coordination complexes. Nomenclature and terminology Coordination complexes are so pervasive that their structures and reactions are described in many ways, sometimes confusingly. The atom within a ligand that is bonded to the central metal atom or ion is called the donor atom. In a typical complex, a metal ion is bonded to several donor atoms, which can be the same or different. A Ligand#Polydentate and polyhapto ligand motifs and nomenclature, polydentate (multiple bonded) ligand is a molecule or ion that bonds to the central atom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |