|
Tautomycetin
Tautomycetin is a natural product first isolated from ''Streptomyces griseochromogenes'', a bacterium found in the soil of the Zhejiang Province, China. It was also later found in ''Penicillium urticae''. It is a linear polyketide very similar in structure to tautomycin, both of which contain a unique dialkylmaleic anhydride moiety, which is essential for their pharmacological activity. Tautomycetin is a selective inhibitor of protein phosphatase 1. __TOC__ Biosynthesis Much of the biosynthesis of tautomycetin has been deduced. It is synthesized in ''S. griseochromogenes'' by two type I polyketide synthases, denoted by modules named TtnA and TtnB. After initial loading with acetyl-CoA, four methylmalonyl-CoAs and three malonyl-CoA Malonyl-CoA is a coenzyme A derivative of malonic acid. Functions It plays a key role in chain elongation in fatty acid biosynthesis and polyketide biosynthesis. Fatty acid biosynthesis Malonyl-CoA provides 2-carbon units to fatty acids and commi . ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tautomycin
Tautomycin is a chemical that occurs naturally in shellfish and is produced by the bacterium '' Streptomyces spiroverticillatus''. It is a polyketide-based structure characterized by a three hydroxyl groups, two ketones, a dialkylmaleic anhydride, an ester linkage (connecting anhydride unit to polyketide chain), a spiroketal and one methyl ether among others. Pharmacology It is a very potent inhibitor of the protein phosphatase In biochemistry, a phosphatase is an enzyme that uses water to cleave a phosphoric acid Ester, monoester into a phosphate ion and an Alcohol (chemistry), alcohol. Because a phosphatase enzyme catalysis, catalyzes the hydrolysis of its Substrate ...s PP1 and PP2A. Tautomycin demonstrates a slight preference for PP1 inhibition relative to PP2A inhibition. Tautomycin is closely related to another anhydride containing polyketide PP inhibitor called tautomycetin which, in addition to being useful as a lead for cancer drug discovery, also is a very pote ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
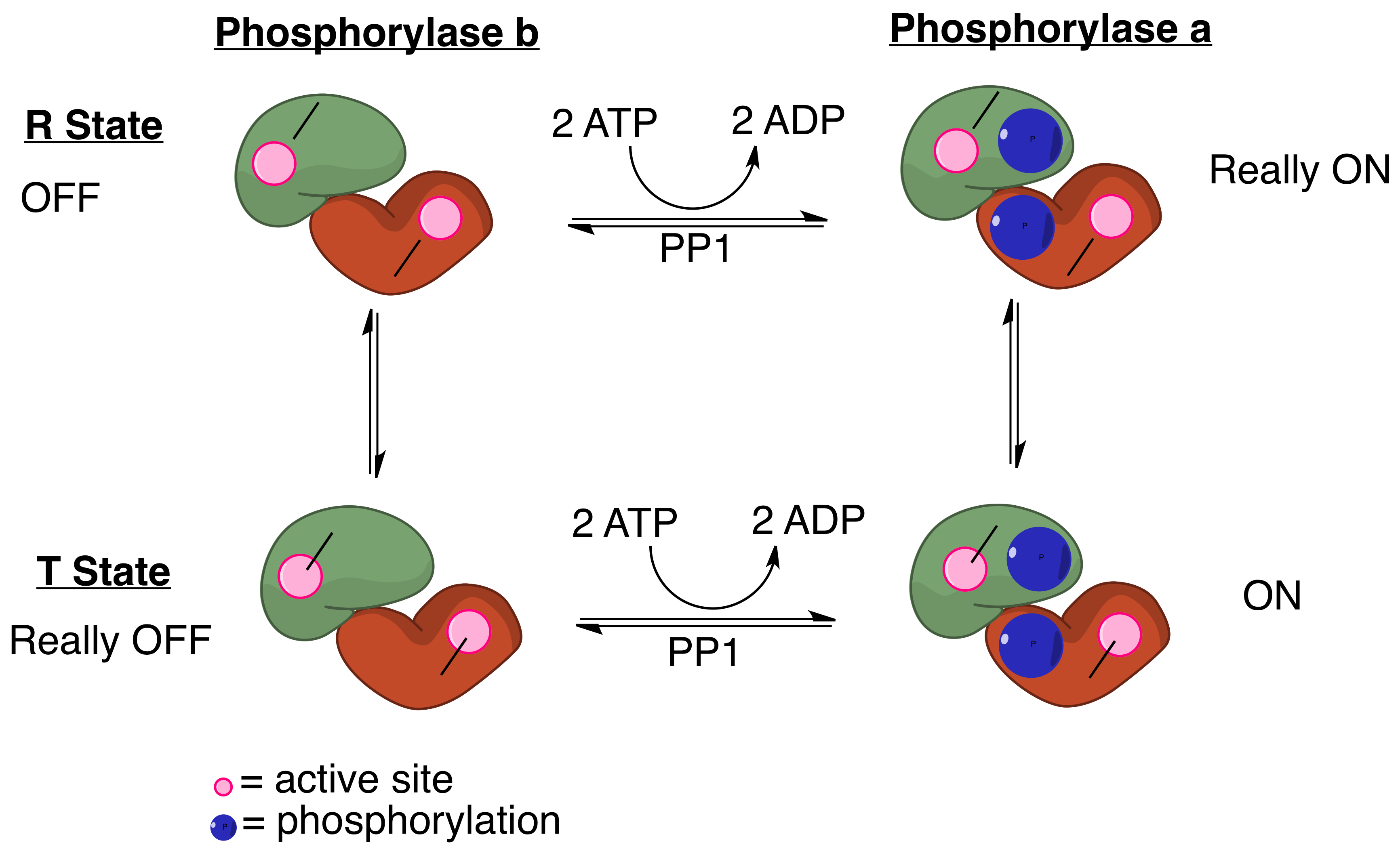
Protein Phosphatase 1
Protein phosphatase 1 (PP1) belongs to a certain class of phosphatases known as protein serine/threonine phosphatases. This type of phosphatase includes metal-dependent protein phosphatases (PPMs) and aspartate-based phosphatases. PP1 has been found to be important in the control of glycogen metabolism, muscle contraction, cell progression, neuronal activities, splicing of RNA, mitosis, cell division, apoptosis, protein synthesis, and regulation of membrane receptors and channels. Structure Each PP1 enzyme contains both a catalytic subunit and at least one regulatory subunit. The catalytic subunit consists of a 30-kD single-domain protein that can form complexes with other regulatory subunits. The catalytic subunit is highly conserved among all eukaryotes, thus suggesting a common catalytic mechanism. The catalytic subunit can form complexes with various regulatory subunits. These regulatory subunits play an important role in substrate specificity as well as compartmentaliza ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyketide Synthase
Polyketides are a class of natural products derived from a precursor molecule consisting of a chain of alternating ketone (or reduced forms of a ketone) and methylene groups: (-CO-CH2-). First studied in the early 20th century, discovery, biosynthesis, and application of polyketides has evolved. It is a large and diverse group of secondary metabolites caused by its complex biosynthesis which resembles that of fatty acid synthesis. Because of this diversity, polyketides can have various medicinal, agricultural, and industrial applications. Many polyketides are medicinal or exhibit acute toxicity. Biotechnology has enabled discovery of more naturally-occurring polyketides and evolution of new polyketides with novel or improved bioactivity. History Naturally produced polyketides by various plants and organisms have been used by humans since before studies on them began in the 19th and 20th century. In 1893, J. Norman Collie synthesized detectable amounts of orcinol by heating de ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetyl-CoA
Acetyl-CoA (acetyl coenzyme A) is a molecule that participates in many biochemical reactions in protein, carbohydrate and lipid metabolism. Its main function is to deliver the acetyl group to the citric acid cycle (Krebs cycle) to be oxidized for energy production. Coenzyme A (CoASH or CoA) consists of a β-mercaptoethylamine group linked to the vitamin pantothenic acid (B5) through an amide linkage and 3'-phosphorylated ADP. The acetyl group (indicated in blue in the structural diagram on the right) of acetyl-CoA is linked to the sulfhydryl substituent of the β-mercaptoethylamine group. This thioester linkage is a "high energy" bond, which is particularly reactive. Hydrolysis of the thioester bond is exergonic (−31.5 kJ/mol). CoA is acetylated to acetyl-CoA by the breakdown of carbohydrates through glycolysis and by the breakdown of fatty acids through β-oxidation. Acetyl-CoA then enters the citric acid cycle, where the acetyl group is oxidized to carbon dioxid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methylmalonyl-CoA
Methylmalonyl-CoA is the thioester consisting of coenzyme A linked to methylmalonic acid. It is an important intermediate in the biosynthesis of succinyl-CoA, which plays an essential role in the tricarboxylic acid cycle (aka the Citric Acid Cycle, or Krebs Cycle). The compound is sometimes referred to as "methylmalyl-CoA". Biosynthesis and metabolism Methylmalonyl-CoA results from the metabolism of fatty acid with an odd number of carbons or from cholesterol side-chains, forming Propionyl-CoA. Propionyl-CoA and bicarbonate are converted to Methylmalonyl-CoA by the enzyme propionyl-CoA Carboxylase. It then is converted into succinyl-CoA by methylmalonyl-CoA mutase (MUT). This reaction is a reversible isomerization. In this way, the compound enters the Citric Acid Cycle. The following diagram demonstrates the aforementioned reaction: Propionyl CoA + Bicarbonate → Methylmalonyl CoA → Succinyl CoA Vitamin B12 Vitamin B12 plays an integral role in this reaction. Coenzyme ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Malonyl-CoA
Malonyl-CoA is a coenzyme A derivative of malonic acid. Functions It plays a key role in chain elongation in fatty acid biosynthesis and polyketide biosynthesis. Fatty acid biosynthesis Malonyl-CoA provides 2-carbon units to fatty acids and commits them to fatty acid chain synthesis. Malonyl-CoA is formed by carboxylating acetyl-CoA using the enzyme acetyl-CoA carboxylase. One molecule of acetyl-CoA joins with a molecule of bicarbonate,Nelson D, Cox M (2008) ''Lehninger principles of biochemistry''. 5th Ed: p. 806 requiring energy rendered from ATP. Malonyl-CoA is utilised in fatty acid biosynthesis by the enzyme malonyl coenzyme A:acyl carrier protein transacylase (MCAT). MCAT serves to transfer malonate from malonyl-CoA to the terminal thiol of ''holo''- acyl carrier protein (ACP). Polyketide biosynthesis MCAT is also involved in bacterial polyketide biosynthesis. The enzyme MCAT together with an acyl carrier protein (ACP), and a polyketide synthase (PKS) and chain-length ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Claisen Condensation
The Claisen condensation is a carbon–carbon bond forming reaction that occurs between two esters or one ester and another carbonyl compound in the presence of a strong base, resulting in a β-keto ester or a β- diketone. It is named after Rainer Ludwig Claisen, who first published his work on the reaction in 1887. Requirements At least one of the reagents must be enolizable (have an α-proton and be able to undergo deprotonation to form the enolate anion). There are a number of different combinations of enolizable and nonenolizable carbonyl compounds that form a few different types of Claisen. The base used must not interfere with the reaction by undergoing nucleophilic substitution or addition with a carbonyl carbon. For this reason, the conjugate sodium alkoxide base of the alcohol formed (e.g. sodium ethoxide if ethanol is formed) is often used, since the alkoxide is regenerated. In mixed Claisen condensations, a non-nucleophilic base such as lithium diisopropylam ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dehydratase
Dehydratases are a group of lyase In biochemistry, a lyase is an enzyme that catalyzes the breaking (an elimination reaction) of various chemical bonds by means other than hydrolysis (a substitution reaction) and oxidation, often forming a new double bond or a new ring structure. ... enzymes that form double and triple bonds in a substrate through the removal of water. They can be found in many places including the mitochondria, peroxisome and cytosol. There are more than 150 different dehydratase enzymes that are classified into four groups. Dehydratases can act on hydroxyacyl-CoA with or without cofactors, and some have a metal and non-metal cluster act as their active site. A dehydratase deficiency in the body can lead to a less severe condition of hyperphenylalaninemia, which involves an over presence of phenylalanine in the blood. It is caused by a genetic recessive disorder in the autosomal DNA. Common dehydratases include: Delta-aminolevulinic acid dehydratase is found ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enoyl-acyl Carrier Protein Reductase
Enoyl-acyl carrier protein reductase (or ENR) (), is a key enzyme of the type II fatty acid synthesis (FAS) system. ENR is an attractive target for narrow-spectrum antibacterial drug discovery because of its essential role in metabolism and its sequence conservation across many bacterial species. In addition, the bacterial ENR sequence and structural organization are distinctly different from those of mammalian fatty acid biosynthesis enzymes. : At lower concentrations, Triclosan and Triclocarban provide a bacteriostatic effect by binding to ENR. Atromentin and leucomelone possess antibacterial activity, inhibiting the enzyme in the bacteria ''Streptococcus pneumoniae''. See also * Enoyl-(acyl-carrier-protein) reductase (NADPH, A-specific) * Enoyl-(acyl-carrier-protein) reductase (NADPH, B-specific) * Cis-2-enoyl-CoA reductase (NADPH) In enzymology, a cis-2-enoyl-CoA reductase (NADPH) () is an enzyme that catalyzes the chemical reaction :acyl-CoA + NADP+ \rightleftharpoons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thioesterase
Thioesterases are enzymes which belong to the esterase family. Esterases, in turn, are one type of the several hydrolases known. Thioesterases exhibit esterase activity (splitting of an ester into acid and alcohol, in the presence of water) specifically at a thiol group. Thioesterases or thiolester hydrolases are identified as members of EC 3.1.2. Family The thioesterase activity is performed by members of the acyl-CoA thioesterase (ACOT) family. The regulatory role of ACOT in fatty acid metabolism depends on their substrate specificity, tissue expression and subcellular localization. For example, deactivation of fatty acids at the ER may traffic fatty acids away from pathways associated with the ER membrane, such as glycerolipid biosynthesis. Two structurally different ACOT types lead to a similar enzymatic activity in vitro, dividing the family into type I and type II ACOTs. Type I ACOTs (ACOT1–6) contain the α/β-hydrolase domain, which is also present in many lipases a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |