|
Phenylacetic Acid
Phenylacetic acid (conjugate base phenylacetate), also known by various synonyms, is an organic compound containing a phenyl functional group and a carboxylic acid functional group. It is a white solid with a strong honey-like odor. Endogenously, it is a catabolite of phenylalanine. As a commercial chemical, because it can be used in the illicit production of phenylacetone (used in the manufacture of substituted amphetamines), it is subject to controls in countries including the United States and China. Occurrence Phenylacetic acid has been found to be an active auxin (a type of plant hormone), found predominantly in fruits. However, its effect is much weaker than the effect of the basic auxin molecule indole-3-acetic acid. In addition the molecule is naturally produced by the metapleural gland of most ant species and used as an antimicrobial. It is also the oxidation product of phenethylamine in humans following metabolism by monoamine oxidase and subsequent metabolism of the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CRC Press
The CRC Press, LLC is an American publishing group that specializes in producing technical books. Many of their books relate to engineering, science and mathematics. Their scope also includes books on business, forensics and information technology. CRC Press is now a division of Taylor & Francis, itself a subsidiary of Informa. History The CRC Press was founded as the Chemical Rubber Company (CRC) in 1903 by brothers Arthur, Leo and Emanuel Friedman in Cleveland, Ohio, based on an earlier enterprise by Arthur, who had begun selling rubber laboratory aprons in 1900. The company gradually expanded to include sales of laboratory equipment to chemist A chemist (from Greek ''chēm(ía)'' alchemy; replacing ''chymist'' from Medieval Latin ''alchemist'') is a graduated scientist trained in the study of chemistry, or an officially enrolled student in the field. Chemists study the composition of ...s. In 1913 the CRC offered a short (116-page) manual called the ''Rubber Handboo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Substituted Amphetamine
Substituted amphetamines, or simply amphetamines, are a chemical class, class of compounds based upon the amphetamine structure; it includes all derivative (chemistry), derivative compounds which are formed by replacing, or substitution reaction, substituting, one or more hydrogen atoms in the amphetamine core structure with substituents. The compounds in this class span a variety of pharmacological subclasses, including stimulants, Empathogen-entactogen, empathogens, and hallucinogens, among others. Examples of substituted amphetamines are amphetamine (itself), methamphetamine, ephedrine, cathinone, phentermine, mephentermine, tranylcypromine, bupropion, methoxyphenamine, selegiline, amfepramone, amfepramone (diethylpropion), pyrovalerone, MDMA (ecstasy), and 2,5-dimethoxy-4-methylamphetamine, DOM (STP). Some of amphetamine's substituted Derivative (chemistry), derivatives occur in nature, for example in the leaves of ''Ephedra (genus), Ephedra'' and khat plants. Amphetamine w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzyl Cyanide
Benzyl cyanide (abbreviated BnCN) is an organic compound with the chemical formula C6H5CH2CN. This colorless oily aromatic liquid is an important precursor to numerous compounds in organic chemistry. It is also an important pheromone in certain species. Preparation Benzyl cyanide can be produced via Kolbe nitrile synthesis between benzyl chloride and sodium cyanide and by oxidative decarboxylation of phenylalanine. Benzyl cyanides can also be prepared by arylation of silyl-substituted acetonitrile. Reactions Benzyl cyanide undergoes many reactions characteristic of nitriles. It can be hydrolyzed to give phenylacetic acid or it can be used in the Pinner reaction to yield phenylacetic acid esters. Hydrogenation gives β-phenethylamine. The compound contains an "active methylene unit". Bromination occurs gives PhCHBrCN. A variety of base-induced reactions result in the formation of new carbon-carbon bonds. Uses Benzyl cyanide is used as a solvent and as a starting material ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of Biomolecule, biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldehyde Dehydrogenase
Aldehyde dehydrogenases () are a group of enzymes that catalyse the oxidation of aldehydes. They convert aldehydes (R–C(=O)) to carboxylic acids (R–C(=O)). The oxygen comes from a water molecule. To date, nineteen ALDH genes have been identified within the human genome. These genes participate in a wide variety of biological processes including the detoxification of exogenously and endogenously generated aldehydes. Function Aldehyde dehydrogenase is a polymorphic enzyme responsible for the oxidation of aldehydes to carboxylic acids. There are three different classes of these enzymes in mammals: class 1 (low ''K''m, cytosolic), class 2 (low ''K''m, mitochondrial), and class 3 (high ''K''m, such as those expressed in tumors, stomach, and cornea). In all three classes, constitutive and inducible forms exist. ALDH1 and ALDH2 are the most important enzymes for aldehyde oxidation, and both are tetrameric enzymes composed of 54 kDa subunits. These enzymes are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenylacetaldehyde
Phenylacetaldehyde is an organic compound used in the synthesis of fragrances and polymers. Phenylacetaldehyde is an aldehyde that consists of acetaldehyde bearing a phenyl substituent; the parent member of the phenylacetaldehyde class of compounds. It has a role as a human metabolite, a ''Saccharomyces cerevisiae'' metabolite, an ''Escherichia coli'' metabolite and a mouse metabolite. It is an alpha-CH2-containing aldehyde and a member of phenylacetaldehydes. Phenylacetaldehyde is one important oxidation-related aldehyde. Exposure to styrene gives phenylacetaldehyde as a secondary metabolite. Styrene has been implicated as reproductive toxicant, neurotoxicant, or carcinogen in vivo or in vitro. Phenylacetaldehyde could be formed by diverse thermal reactions during the cooking process together with C8 compounds is identified as a major aroma–active compound in cooked pine mushroom. Phenylacetaldehyde is readily oxidized to phenylacetic acid. Therefore will eventually be hydrolyz ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monoamine Oxidase
Monoamine oxidases (MAO) () are a family of enzymes that catalyze the oxidation of monoamines, employing oxygen to clip off their amine group. They are found bound to the outer membrane of mitochondria in most cell types of the body. The first such enzyme was discovered in 1928 by Mary Bernheim in the liver and was named tyramine oxidase. The MAOs belong to the protein family of flavin-containing amine oxidoreductases. MAOs are important in the breakdown of monoamines ingested in food, and also serve to inactivate monoamine neurotransmitters. Because of the latter, they are involved in a number of psychiatric and neurological diseases, some of which can be treated with monoamine oxidase inhibitors (MAOIs) which block the action of MAOs. Subtypes and tissue distribution In humans there are two types of MAO: MAO-A and MAO-B. * Both are found in neurons and astroglia. * Outside the central nervous system: ** MAO-A is also found in the liver, pulmonary vascular end ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenethylamine
Phenethylamine (PEA) is an organic compound, natural monoamine alkaloid, and trace amine, which acts as a central nervous system stimulant in humans. In the brain, phenethylamine regulates monoamine neurotransmission by binding to trace amine-associated receptor 1 (TAAR1) and inhibiting vesicular monoamine transporter 2 (VMAT2) in monoamine neurons. To a lesser extent, it also acts as a neurotransmitter in the human central nervous system. In mammals, phenethylamine is produced from the amino acid L-phenylalanine by the enzyme aromatic L-amino acid decarboxylase via enzymatic decarboxylation. In addition to its presence in mammals, phenethylamine is found in many other organisms and foods, such as chocolate, especially after microbial fermentation. Phenethylamine is sold as a dietary supplement for purported mood and weight loss-related therapeutic benefits; however, in orally ingested phenethylamine, a significant amount is metabolized in the small intestine by mon ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Indole-3-acetic Acid
Indole-3-acetic acid (IAA, 3-IAA) is the most common naturally occurring plant hormone of the auxin class. It is the best known of the auxins, and has been the subject of extensive studies by plant physiologists. IAA is a derivative of indole, containing a carboxymethyl substituent. It is a colorless solid that is soluble in polar organic solvents. Biosynthesis IAA is predominantly produced in cells of the apex ( bud) and very young leaves of a plant. Plants can synthesize IAA by several independent biosynthetic pathways. Four of them start from tryptophan, but there is also a biosynthetic pathway independent of tryptophan. Plants mainly produce IAA from tryptophan through indole-3-pyruvic acid. IAA is also produced from tryptophan through indole-3-acetaldoxime in ''Arabidopsis thaliana''. In rats, IAA is a product of both endogenous and colonic microbial metabolism from dietary tryptophan along with tryptophol. This was first observed in rats infected by '' Trypanosoma br ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fruit
In botany, a fruit is the seed-bearing structure in flowering plants (angiosperms) that is formed from the ovary after flowering. Fruits are the means by which angiosperms disseminate their seeds. Edible fruits in particular have long propagated using the movements of humans and other animals in a symbiotic relationship that is the means for seed dispersal for the one group and nutrition for the other; humans, and many other animals, have become dependent on fruits as a source of food. Consequently, fruits account for a substantial fraction of the world's agricultural output, and some (such as the apple and the pomegranate) have acquired extensive cultural and symbolic meanings. In common language and culinary usage, ''fruit'' normally means the seed-associated fleshy structures (or produce) of plants that typically are sweet (or sour) and edible in the raw state, such as apples, bananas, grapes, lemons, oranges, and strawberries. In botanical usage, the term ''fruit'' als ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Plant Hormone
Plant hormones (or phytohormones) are signal molecules, produced within plants, that occur in extremely low concentrations. Plant hormones control all aspects of plant growth and development, including embryogenesis, the regulation of Organ (anatomy), organ size, pathogen defense, Stress (biology), stress tolerance and Reproduction, reproductive development. Unlike in animals (in which hormone production is restricted to specialized glands) each plant cell is capable of producing hormones. Frits Warmolt Went, Went and Thimann coined the term "phytohormone" and used it in the title of their 1937 book. Phytohormones occur across the plant kingdom, and even in algae, where they have similar functions to those seen in vascular plant, vascular plants ("higher plants"). Some phytohormones also occur in microorganisms, such as unicellular fungus, fungi and bacteria, however in these cases they do not play a hormonal role and can better be regarded as secondary metabolites. Characteristi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
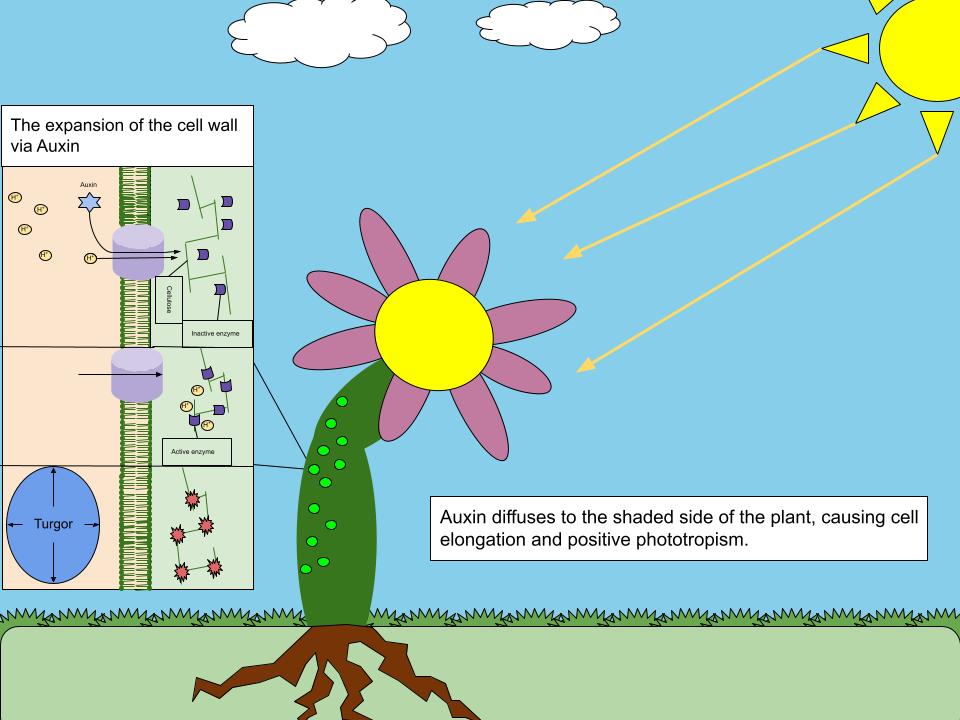
Auxin
Auxins (plural of auxin ) are a class of plant hormones (or plant-growth regulators) with some morphogen-like characteristics. Auxins play a cardinal role in coordination of many growth and behavioral processes in plant life cycles and are essential for plant body development. The Dutch biologist Frits Warmolt Went first described auxins and their role in plant growth in the 1920s. Kenneth V. Thimann became the first to isolate one of these phytohormones and to determine its chemical structure as indole-3-acetic acid (IAA). Went and Thimann co-authored a book on plant hormones, ''Phytohormones'', in 1937. Overview Auxins were the first of the major plant hormones to be discovered. They derive their name from the Greek word ( – 'to grow/increase'). Auxin is present in all parts of a plant, although in very different concentrations. The concentration in each position is crucial developmental information, so it is subject to tight regulation through both metabolism and transp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |