|
Marcasite
The mineral marcasite, sometimes called "white iron pyrite", is iron sulfide (FeS2) with orthorhombic crystal structure. It is physically and crystallographically distinct from pyrite, which is iron sulfide with cubic crystal structure. Both structures contain the disulfide S22− ion, having a short bonding distance between the sulfur atoms. The structures differ in how these di-anions are arranged around the Fe2+ cations. Marcasite is lighter and more brittle than pyrite. Specimens of marcasite often crumble and break up due to the unstable crystal structure. On fresh surfaces, it is pale yellow to almost white and has a bright metallic luster. It tarnishes to a yellowish or brownish color and gives a black streak. It is a brittle material that cannot be scratched with a knife. The thin, flat, tabular crystals, when joined in groups, are called "cockscombs". In the late medieval and early modern eras, the word "marcasite" meant all iron sulfides in general, including b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Marcasite Kühlungsborn Germany
The mineral marcasite, sometimes called "white iron pyrite", is iron sulfide (FeS2) with orthorhombic crystal structure. It is physically and crystallographically distinct from pyrite, which is iron sulfide with cubic crystal structure. Both structures contain the disulfide S22− ion, having a short bonding distance between the sulfur atoms. The structures differ in how these di-anions are arranged around the Fe2+ cations. Marcasite is lighter and more brittle than pyrite. Specimens of marcasite often crumble and break up due to the unstable crystal structure. On fresh surfaces, it is pale yellow to almost white and has a bright metallic luster. It tarnishes to a yellowish or brownish color and gives a black streak. It is a brittle material that cannot be scratched with a knife. The thin, flat, tabular crystals, when joined in groups, are called "cockscombs". In the late medieval and early modern eras, the word "marcasite" meant all iron sulfides in general, including both p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Marcasite Jewellery
Marcasite jewellery is jewellery made using cut and polished pieces of pyrite (fool's gold) as a gemstone, and not, as the name suggests, from marcasite. Both pyrite and marcasite are chemically iron sulfide, but differ in their crystal structures, giving them different physical properties. Pyrite is more stable and less brittle than marcasite. Marcasite can also react with moisture to form sulfuric acid. These are the reasons why pyrite is used instead of real marcasite in "marcasite" jewellery. Marcasite jewellery is frequently made by setting small pieces of faceted pyrite into silver. Cheaper costume jewellery is made by gluing pieces of pyrite rather than setting. A similar-looking type of jewellery can be made from small pieces of cut steel. The cut and polished marcasite pieces reflect light at different angles from its different facets giving it a sparkle. Thailand is one of the large producers of modern marcasite jewellery in silver. History Marcasite jewellery ha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
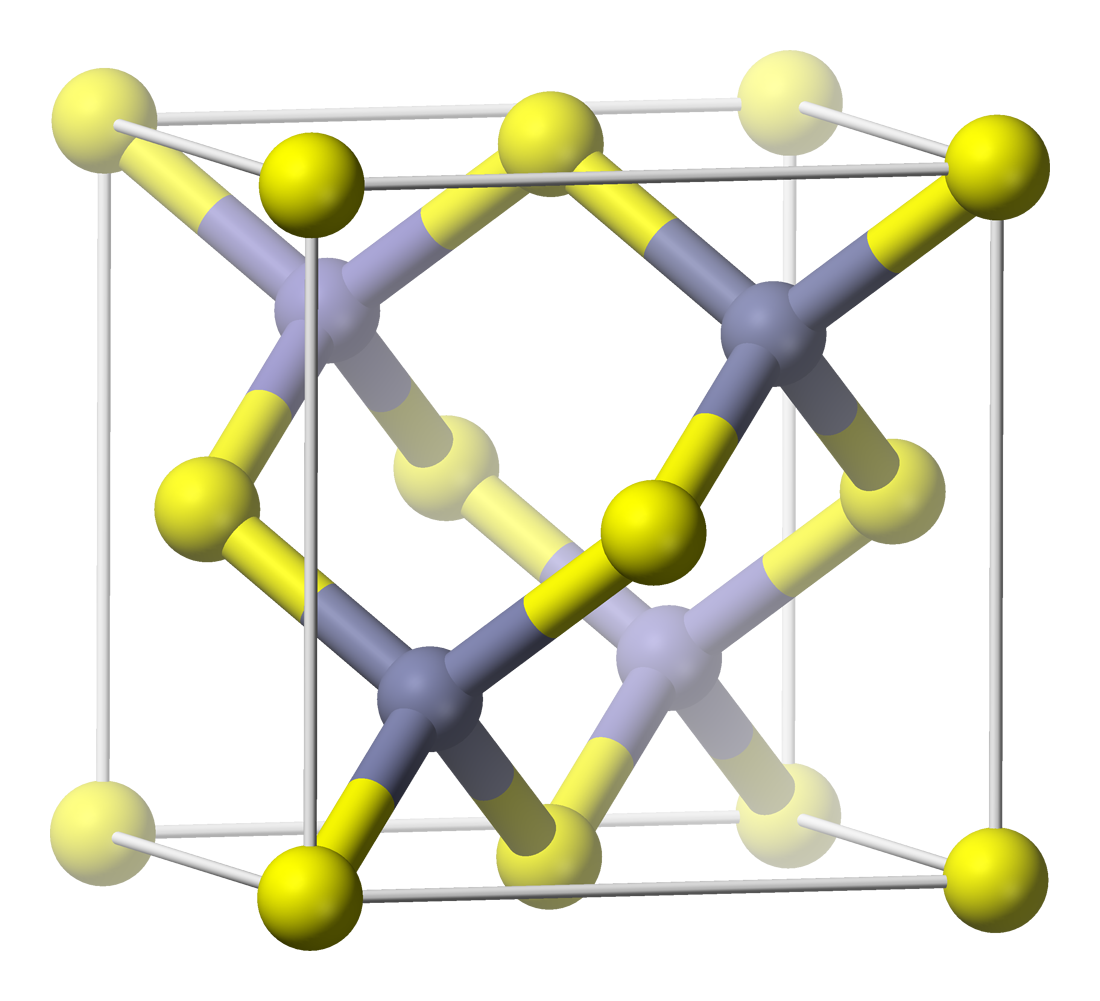
Pyrite
The mineral pyrite ( ), or iron pyrite, also known as fool's gold, is an iron sulfide with the chemical formula Fe S2 (iron (II) disulfide). Pyrite is the most abundant sulfide mineral. Pyrite's metallic luster and pale brass-yellow hue give it a superficial resemblance to gold, hence the well-known nickname of ''fool's gold''. The color has also led to the nicknames ''brass'', ''brazzle'', and ''brazil'', primarily used to refer to pyrite found in coal. The name ''pyrite'' is derived from the Greek (), 'stone or mineral which strikes fire', in turn from (), 'fire'. In ancient Roman times, this name was applied to several types of stone that would create sparks when struck against steel; Pliny the Elder described one of them as being brassy, almost certainly a reference to what is now called pyrite. By Georgius Agricola's time, , the term had become a generic term for all of the sulfide minerals. Pyrite is usually found associated with other sulfides or oxides in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfide Mineral
The sulfide minerals are a class of minerals containing sulfide (S2−) or disulfide () as the major anion. Some sulfide minerals are economically important as metal ores. The sulfide class also includes the selenide mineral, selenides, the telluride mineral, tellurides, the arsenide mineral, arsenides, the antimonide mineral, antimonides, the bismuthinides, the sulfarsenides and the sulfosalts.http://www.minerals.net/mineral/sort-met.hod/group/sulfgrp.htm Minerals.net Dana Classification, SulfidesKlein, Cornelis and Cornelius S. Hurlbut, Jr., 1986, ''Manual of Mineralogy'', Wiley, 20th ed., pp 269-293 Sulfide minerals are inorganic compounds. Minerals Common or important examples include: * Acanthite *Chalcocite *Bornite *Galena *Sphalerite *Chalcopyrite *Pyrrhotite *Millerite *Pentlandite *Covellite *Cinnabar *Realgar *Orpiment *Stibnite *Pyrite *Marcasite *Molybdenite Sulfarsenides: *Cobaltite *Arsenopyrite *Gersdorffite Sulfosalts: *Pyrargyrite *Pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron Sulfide
Iron sulfide or iron sulphide can refer to range of chemical compounds composed of iron and sulfur. Minerals By increasing order of stability: * Iron(II) sulfide, FeS * Greigite, Fe3S4 (cubic) * Pyrrhotite, Fe1−xS (where x = 0 to 0.2) (monoclinic or hexagonal) * Troilite, FeS, the endmember of pyrrhotite (hexagonal) * Mackinawite Mackinawite is an iron nickel sulfide mineral with the chemical formula (where x = 0 to 0.11). The mineral crystallizes in the tetragonal crystal system and has been described as a distorted, close packed, cubic array of S atoms with some of t ..., Fe1+xS (where x = 0 to 0.1) (tetragonal) * Marcasite, orthorhombic FeS2 * Pyrite, cubic FeS2 (fool's gold) * Arsenopyrite (''mispickel''), FeAsS, or Fe(As-S), Fe(III) mixed arseno-sulfide (monoclinic) Synthetic * Iron(III) sulfide, Fe2S3 * Iron-sulfur clusters, includes both synthetic and biological Biological * Iron–sulfur protein {{DEFAULTSORT:Iron Sulfide Iron compounds Iron minerals ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sedimentary Rock
Sedimentary rocks are types of rock (geology), rock formed by the cementation (geology), cementation of sediments—i.e. particles made of minerals (geological detritus) or organic matter (biological detritus)—that have been accumulated or deposited at Earth's surface. Sedimentation is any process that causes these particles to settle in place. Geological detritus originates from weathering and erosion of existing rocks, or from the solidification of molten lava blobs erupted by volcanoes. The geological detritus is transported to the place of deposition by water, wind, ice or Mass wasting, mass movement, which are called agents of denudation. Biological detritus is formed by bodies and parts (mainly shells) of dead aquatic organisms, as well as their fecal mass, suspended in water and slowly piling up on the floor of water bodies (marine snow). Sedimentation may also occur when dissolved minerals precipitate from aqueous solution, water solution. The sedimentary rock cover of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcite
Calcite is a Carbonate minerals, carbonate mineral and the most stable Polymorphism (materials science), polymorph of calcium carbonate (CaCO3). It is a very common mineral, particularly as a component of limestone. Calcite defines hardness 3 on the Mohs scale of mineral hardness, based on Scratch hardness, scratch hardness comparison. Large calcite crystals are used in optical equipment, and limestone composed mostly of calcite has numerous uses. Other polymorphs of calcium carbonate are the minerals aragonite and vaterite. Aragonite will change to calcite over timescales of days or less at temperatures exceeding 300 °C, and vaterite is even less stable. Etymology Calcite is derived from the German , a term from the 19th century that came from the Latin word for Lime (material), lime, (genitive ) with the suffix ''-ite'' used to name minerals. It is thus a Doublet (linguistics), doublet of the word ''wikt:chalk, chalk''. When applied by archaeology, archaeologists and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dolomite (mineral)
Dolomite () is an anhydrous carbonate mineral composed of calcium magnesium carbonate, ideally The term is also used for a sedimentary carbonate rock composed mostly of the mineral dolomite (see Dolomite (rock)). An alternative name sometimes used for the dolomitic rock type is dolostone. History As stated by Nicolas-Théodore de Saussure the mineral dolomite was probably first described by Carl Linnaeus in 1768. In 1791, it was described as a rock by the French natural history, naturalist and geologist Déodat Gratet de Dolomieu (1750–1801), first in buildings of the old city of Rome, and later as samples collected in the County_of_Tyrol, Tyrolean Alps. Nicolas-Théodore de Saussure first named the mineral (after Dolomieu) in March 1792. Properties The mineral dolomite crystallizes in the trigonal, trigonal-rhombohedral system. It forms white, tan, gray, or pink crystals. Dolomite is a double carbonate, having an alternating structural arrangement of calcium and magnesium ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
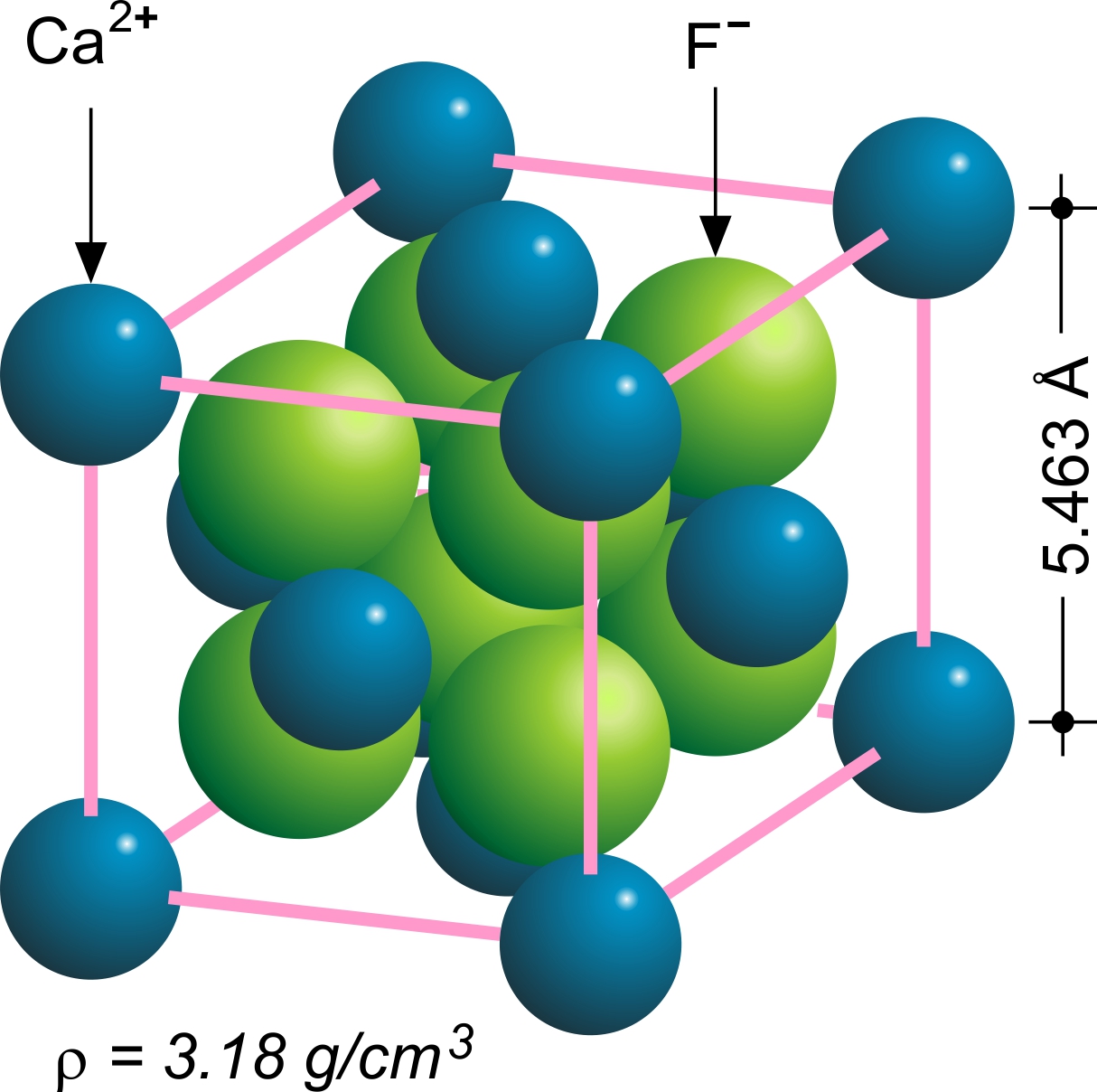
Fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon. The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite. Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite has anomalous partial dispersion, that is, its refractive index varies with the wavelength of light in a manner that differs from that of commonly used glasses, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sphalerite
Sphalerite is a sulfide mineral with the chemical formula . It is the most important ore of zinc. Sphalerite is found in a variety of deposit types, but it is primarily in Sedimentary exhalative deposits, sedimentary exhalative, Carbonate-hosted lead-zinc ore deposits, Mississippi-Valley type, and Volcanogenic massive sulfide ore deposit, volcanogenic massive sulfide deposits. It is found in association with galena, chalcopyrite, pyrite (and other sulfide mineral, sulfides), calcite, dolomite (mineral), dolomite, quartz, rhodochrosite, and fluorite. German geologist Ernst Friedrich Glocker discovered sphalerite in 1847, naming it based on the Greek word ''sphaleros'', meaning "deceiving", due to the difficulty of identifying the mineral. In addition to zinc, sphalerite is an ore of cadmium, gallium, germanium, and indium. Miners have been known to refer to sphalerite as ''zinc blende'', ''black-jack'', and ''ruby blende''. Marmatite is an opaque black variety with a high iron ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Galena
Galena, also called lead glance, is the natural mineral form of lead(II) sulfide (PbS). It is the most important ore of lead and an important source of silver. Galena is one of the most abundant and widely distributed sulfide minerals. It crystallizes in the Cubic (crystal system), cubic crystal system often showing octahedral forms. It is often associated with the minerals sphalerite, calcite and fluorite. As a pure specimen held in the hand, under standard temperature and pressure, galena is insoluble in water and so is almost non-toxic. Handling galena under these specific conditions (such as in a museum or as part of geology instruction) poses practically no risk; however, as lead(II) sulfide is reasonably reactive in a variety of environments, it can be highly toxic if swallowed or inhaled, particularly under prolonged or repeated exposure. Occurrence Galena is the main ore of lead, used since ancient times, since lead can be smelted from galena in an ordinary wood fire. G ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrrhotite
Pyrrhotite (''Pyrrhus of Epirus, pyrrhos'' in Greek language, Greek meaning "flame-coloured"'')'' is an iron sulfide mineral with the formula Fe(1−x)S (x = 0 to 0.125). It is a nonstoichiometric compound, nonstoichiometric variant of FeS, the mineral known as troilite. Pyrrhotite is also called magnetic pyrite, because the color is similar to pyrite and it is weakly magnetic. The magnetism decreases as the iron content increases, and troilite is non-magnetic. Pyrrhotite is generally tabular and brassy/bronze in color with a Lustre (mineralogy), metallic luster. The mineral occurs with Mafic, mafic igneous rocks like Norite, norites, and may form from pyrite during Metamorphism, metamorphic processes. Pyrrhotite is associated and mined with other sulfide minerals like pentlandite, pyrite, chalcopyrite, and magnetite, and has been found globally. Structure Pyrrhotite exists as a number of polytypes of Hexagonal crystal system, hexagonal or monoclinic crystal symmetry; sever ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |