|
Inorganic Carbon
Total inorganic carbon (''C''T or TIC) is the sum of the inorganic carbon species. Carbon compounds can be distinguished as either organic or inorganic, and dissolved or particulate, depending on their composition. Organic carbon forms the backbone of key components of organic compounds such as proteins, lipids, carbohydrates, and nucleic acids. Inorganic carbon is found primarily in simple compounds such as carbon dioxide (), carbonic acid (), bicarbonate (), and carbonate (). Overview The aquatic inorganic carbon system is composed of the various ionic, dissolved, solid, and/or gaseous forms of carbon dioxide in water. These species include dissolved carbon dioxide, carbonic acid, bicarbonate anion, carbonate anion, calcium carbonate, magnesium carbonate, and others. The relative amounts of each species in a body of water depends on physical variables including temperature and salinity, as well as chemical variables like pH and gas partial pressure. Variables like alkali ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon
Carbon () is a chemical element; it has chemical symbol, symbol C and atomic number 6. It is nonmetallic and tetravalence, tetravalent—meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 electrons. It belongs to group 14 of the periodic table. Carbon makes up about 0.025 percent of Earth's crust. Three Isotopes of carbon, isotopes occur naturally, carbon-12, C and carbon-13, C being stable, while carbon-14, C is a radionuclide, decaying with a half-life of 5,700 years. Carbon is one of the timeline of chemical element discoveries#Pre-modern and early modern discoveries, few elements known since antiquity. Carbon is the 15th abundance of elements in Earth's crust, most abundant element in the Earth's crust, and the abundance of the chemical elements, fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. Carbon's abundance, its unique diversity of organic compounds, and its unusual abi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium Carbonate
Calcium carbonate is a chemical compound with the chemical formula . It is a common substance found in Rock (geology), rocks as the minerals calcite and aragonite, most notably in chalk and limestone, eggshells, gastropod shells, shellfish skeletons and pearls. Materials containing much calcium carbonate or resembling it are described as calcareous. Calcium carbonate is the active ingredient in agricultural lime and is produced when calcium ions in hard water react with carbonate ions to form limescale. It has medical use as a calcium supplement or as an antacid, but excessive consumption can be hazardous and cause hypercalcemia and digestive issues. Chemistry Calcium carbonate shares the typical properties of other carbonates. Notably, it: *reacts with acids, releasing carbonic acid which quickly disintegrates into carbon dioxide and water: : *releases carbon dioxide upon heating, called a thermal decomposition reaction, or calcination (to above 840 °C in the case of ), t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
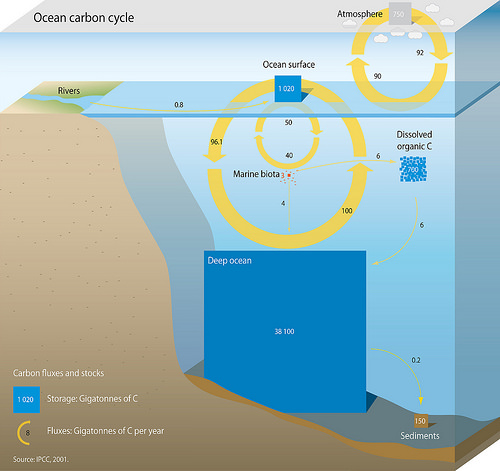
Marine Carbon Cycle
The oceanic carbon cycle (or marine carbon cycle) is composed of processes that exchange carbon between various pools within the ocean as well as between the atmosphere, Earth interior, and the Seabed, seafloor. The carbon cycle is a result of many interacting forces across multiple time and space scales that circulates carbon around the planet, ensuring that carbon is available globally. The Oceanic carbon cycle is a central process to the global carbon cycle and contains both Total inorganic carbon, inorganic carbon (carbon not associated with a living thing, such as carbon dioxide) and Organic matter, organic carbon (carbon that is, or has been, incorporated into a living thing). Part of the marine carbon cycle transforms carbon between non-living and living matter. Three main processes (or pumps) that make up the marine carbon cycle bring atmospheric carbon dioxide (CO2) into the ocean interior and distribute it through the oceans. These three pumps are: (1) the solubility pum ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Limnology
Limnology ( ; ) is the study of inland aquatic ecosystems. It includes aspects of the biological, chemical, physical, and geological characteristics of fresh and saline, natural and man-made bodies of water. This includes the study of lakes, reservoirs, ponds, rivers, springs, streams, wetlands, and groundwater.) Water systems are often categorized as either running ( lotic) or standing ( lentic). Limnology includes the study of the drainage basin, movement of water through the basin and biogeochemical changes that occur en route. A more recent sub-discipline of limnology, termed landscape limnology, studies, manages, and seeks to conserve these ecosystems using a landscape perspective, by explicitly examining connections between an aquatic ecosystem and its drainage basin. Recently, the need to understand global inland waters as part of the Earth system created a sub-discipline called global limnology. This approach considers processes in inland waters on a global scale, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oceanography
Oceanography (), also known as oceanology, sea science, ocean science, and marine science, is the scientific study of the ocean, including its physics, chemistry, biology, and geology. It is an Earth science, which covers a wide range of topics, including ocean currents, waves, and geophysical fluid dynamics; fluxes of various chemical substances and physical properties within the ocean and across its boundaries; ecosystem dynamics; and plate tectonics and seabed geology. Oceanographers draw upon a wide range of disciplines to deepen their understanding of the world’s oceans, incorporating insights from astronomy, biology, chemistry, geography, geology, hydrology, meteorology and physics. History Early history Humans first acquired knowledge of the waves and currents of the seas and oceans in pre-historic times. Observations on tides were recorded by Aristotle and Strabo in 384–322 BC. Early exploration of the oceans was primarily for cartography and mainly ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
PCO2
''p''CO2, pCO2, or P_\ce is the partial pressure of carbon dioxide (CO2), often used in reference to blood but also used in meteorology, climate science, oceanography, and limnology to describe the fractional pressure of CO2 as a function of its concentration in gas or dissolved phases. The units of ''p''CO2 are mmHg, atm, torr, Pa, or any other standard unit of atmospheric pressure. The ''p''CO2 of Earth's atmosphere has risen from approximately 280 ppm ( parts-per-million) to a mean 2019 value of 409.8 ppm as a result of anthropogenic release of carbon dioxide from fossil fuel burning. This is the highest atmospheric concentration to have existed on Earth for at least the last 800,000 years. Medicine In medicine, the partial pressure of carbon dioxide in arterial blood is called P_ or PaCO2. Measurement of P_ in the systemic circulation indicates the effectiveness of ventilation at the lungs' alveoli, given the diffusing capacity of the gas. It is a good indicator of r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dissolved Inorganic Carbon
Dissolved inorganic carbon (DIC) is the sum of the aqueous species of inorganic carbon in a solution. Carbon compounds can be distinguished as either organic or inorganic, and as dissolved or particulate, depending on their composition. Organic carbon forms the backbone of key component of organic compounds such as – proteins, lipids, carbohydrates, and nucleic acids. Inorganic carbon is found primarily in simple compounds such as carbon dioxide, carbonic acid, bicarbonate, and carbonate (CO2, H2CO3, , respectively). Dissolved inorganic carbon (DIC) includes three major aqueous species, CO2, ,, and to a lesser extent their complexes in solution with metal ions. Marine ecosystems Solubility pump Aqueous carbon dioxide reacts with water to form carbonic acid which is very unstable and will dissociate rapidly into hydronium and bicarbonate. Therefore, in seawater, dissolved inorganic carbon is commonly referred to as the collection of bicarbonate, carbonate ions, and dissolv ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkalinity
Alkalinity (from ) is the capacity of water to resist Freshwater acidification, acidification. It should not be confused with base (chemistry), basicity, which is an absolute measurement on the pH scale. Alkalinity is the strength of a buffer solution composed of weak acids and their conjugate acid, conjugate bases. It is measured by Titration, titrating the Solution (chemistry), solution with an acid such as HCl until its pH changes abruptly, or it reaches a known endpoint where that happens. Alkalinity is expressed in units of concentration, such as meq/L (milliequivalents per liter), μeq/kg (microequivalents per kilogram), or mg/L CaCO3 (milligrams per liter of calcium carbonate). Each of these measurements corresponds to an amount of acid added as a titrant. In Fresh water, freshwater, particularly those on non-limestone terrains, alkalinities are low and involve a lot of ions. In the ocean, on the other hand, alkalinity is completely dominated by carbonate and bicarbonate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inorganic Carbon
Total inorganic carbon (''C''T or TIC) is the sum of the inorganic carbon species. Carbon compounds can be distinguished as either organic or inorganic, and dissolved or particulate, depending on their composition. Organic carbon forms the backbone of key components of organic compounds such as proteins, lipids, carbohydrates, and nucleic acids. Inorganic carbon is found primarily in simple compounds such as carbon dioxide (), carbonic acid (), bicarbonate (), and carbonate (). Overview The aquatic inorganic carbon system is composed of the various ionic, dissolved, solid, and/or gaseous forms of carbon dioxide in water. These species include dissolved carbon dioxide, carbonic acid, bicarbonate anion, carbonate anion, calcium carbonate, magnesium carbonate, and others. The relative amounts of each species in a body of water depends on physical variables including temperature and salinity, as well as chemical variables like pH and gas partial pressure. Variables like alkali ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Water
Water is an inorganic compound with the chemical formula . It is a transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance. It is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as a solvent). It is vital for all known forms of life, despite not providing food energy or organic micronutrients. Its chemical formula, , indicates that each of its molecules contains one oxygen and two hydrogen atoms, connected by covalent bonds. The hydrogen atoms are attached to the oxygen atom at an angle of 104.45°. In liquid form, is also called "water" at standard temperature and pressure. Because Earth's environment is relatively close to water's triple point, water exists on Earth as a solid, a liquid, and a gas. It forms precipitation in the form of rain and aerosols in the form of fog. Clouds consist of suspended droplets of water and ice, its solid state. When finely divided, crystalline ice ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Equilibrium
In a chemical reaction, chemical equilibrium is the state in which both the Reagent, reactants and Product (chemistry), products are present in concentrations which have no further tendency to change with time, so that there is no observable change in the properties of the Thermodynamic system, system. This state results when the forward reaction proceeds at the same rate as the Reversible reaction, reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but they are equal. Thus, there are no net changes in the concentrations of the reactants and products. Such a state is known as dynamic equilibrium. It is the subject of study of ''equilibrium chemistry''. Historical introduction The Concept learning, concept of chemical equilibrium was developed in 1803, after Claude Louis Berthollet, Berthollet found that some chemical reactions are Reversible reaction, reversible. For any reaction mixture to exist at equilibrium, the reaction rate, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CO2SYS
CO2SYS is a family of software programs that calculate chemical equilibria for aquatic inorganic carbon species and parameters. Their core function is to use any two of the four central inorganic carbon system parameters ( pH, alkalinity, dissolved inorganic carbon, and partial pressure of carbon dioxide) to calculate various chemical properties of the system. These programs are widely used by oceanographers and limnologists to understand and predict chemical equilibria in natural waters. History Chemical equilibria in marine and freshwater systems were calculated according to various conventions for most of the 20th century, which led to discrepancies among laboratories' calculations and limited scientific reproducibility. CO2SYS was first published by Ernie Lewis and Doug Wallace in 1998 as a DOS-interface program written in QBasic. Subsequent developments have included several MATLAB implementations, two Microsoft Excel templates, a Python package "PyCO2SYS", and an R ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |