|
Iminium
In organic chemistry, an iminium cation is a polyatomic ion with the general structure . They are common in synthetic chemistry and biology. Structure Iminium cations adopt alkene-like geometries: the central C=N unit is nearly coplanar with all four substituents. Unsymmetrical iminium cations can exist as cis and trans isomers. The C=N bonds, which are near 129 picometers in length, are shorter than C-N single bonds. Cis/trans isomers are observed. The C=N distance is slightly shorter in iminium cations than in the parent imine, and computational studies indicate that the C=N bonding is also stronger in iminium vs imine, although the C=N distance contracts only slightly. These results indicate that the barrier for rotation is higher than in the parent imines. Formation Iminium cations are obtained by protonation and alkylation of imines: : : They also are generated by the condensation of secondary amines with ketones or aldehydes: : This rapid, reversible reaction is one ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
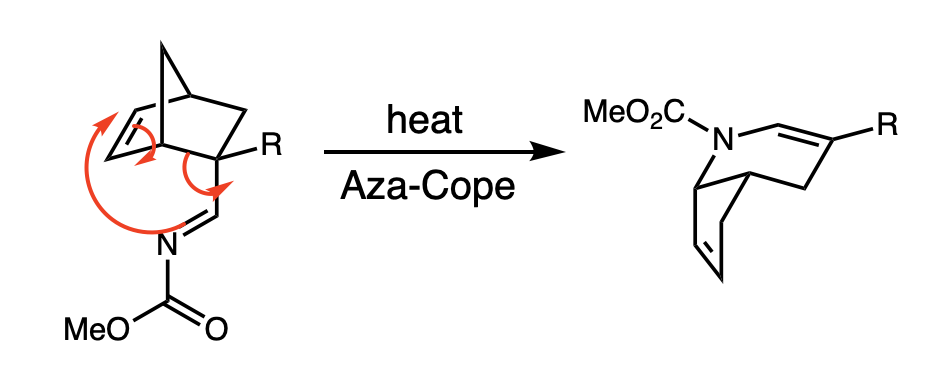
Aza-Cope Rearrangement
Rearrangements, especially those that can participate in cascade reactions, such as the aza-Cope rearrangements, are of high practical as well as conceptual importance in organic chemistry, due to their ability to quickly build structural complexity out of simple starting materials. The aza-Cope rearrangements are examples of heteroatom versions of the Cope rearrangement, which is a [3,3]-Sigmatropic reaction, sigmatropic rearrangement that shifts single and double bonds between two allylic components. #Rate acceleration due to positively charged nitrogen, In accordance with the Woodward-Hoffman rules, thermal aza-Cope rearrangements proceed suprafacially. Aza-Cope rearrangements are generally classified by the position of the nitrogen in the molecule (see figure): The first example of an aza-Cope rearrangement was the ubiquitous #cationic 2-aza-Cope Rearrangement, cationic 2-aza-Cope rearrangement, which takes place at temperatures 100-200 °C lower than the Cope rearrangemen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vilsmeier Reagent
The Vilsmeier reagent is an organic compound with the formula CH3)2NCHCll. It is a salt consisting of the N,N-dimethyliminium cation ( CH3)2N=CHClsup>+) and chloride anion. Depending on the particular reaction, the anion can vary. In typical POCl3-based reactions, the anion is PO2Cl2−. The iminium cation CH3)2N=CHClsup>+ is the reactive component of interest. This iminium species is a derivative of the imidoyl chloride CH3N=CHCl. Analogues of this particular reagent are generated when tertiary amides other than DMF are treated with POCl3. The salt is a white solid that is soluble in polar organic solvents. Vilsmeier reagent is the active intermediate in the formylation reactions, the Vilsmeier reaction or Vilsmeier-Haack reaction that use mixtures of dimethylformamide and phosphorus oxychloride Phosphoryl chloride (commonly called phosphorus oxychloride) is a colourless liquid with the formula . It hydrolyses in moist air releasing phosphoric acid and fumes of hydr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Cyanoborohydride
Sodium cyanoborohydride is a chemical compound with the formula . It is a colourless salt used in organic synthesis for chemical reduction including that of imines and carbonyls. Sodium cyanoborohydride is a milder reductant than other conventional reducing agents. Structure Sodium cyanoborohydride is a salt. The cationic sodium ion, asup>+, interacts with the anionic cyanoborohydride ion, H3(CN)sup>−. The anionic component of the salt is tetrahedral at the boron atom. The electron-withdrawing cyanide substituent draws electron density away from the negatively charged boron; thus, reducing the electrophilic capabilities of the anionic component. This electronic phenomenon causes sodium cyanoborohydride to have more mild reducing qualities than other reducing agents. For example, Na H3(CN)is less reducing than its counterpart sodium borohydride, containing H4sup>−. Uses Sodium cyanoborohydride is a mild reducing agent. It is generally used for the reduction of imines. T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Eschenmoser's Salt
In organic chemistry, Eschenmoser's salt (named for Albert Eschenmoser) is the ionic, organic compound . It is the iodide salt of the dimethylaminomethylene cation . The dimethylaminomethylene cation is a strong dimethylaminomethylating agent, used to prepare derivatives of the type .E. F. Kleinman in "Dimethylmethyleneammonium Iodide and Chloride" in Encyclopedia of Reagents for Organic Synthesis (Ed: L. Paquette) 2004, J. Wiley & Sons, New York. Enolates, silyl enol ethers, and even more acidic ketones undergo efficient dimethylaminomethylation. Once prepared, such tertiary amines can be further methylated and then subjected to base-induced elimination to afford methylidenated ketones. The salt was first prepared by the group of Albert Eschenmoser after whom the reagent is named. Structure and bonding Dimethylaminomethylene cation is described as a resonance hybrid of the carbocation and an iminium cation: : The atoms are coplanar. The cation is isoelectronic with isob ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mannich Reaction
In organic chemistry, the Mannich reaction is a three-component organic reaction that involves the amino alkylation of an acidic proton next to a carbonyl () functional group by formaldehyde () and a primary or secondary amine () or ammonia (). The final product is a β-amino-carbonyl compound also known as a Mannich base. Reactions between aldimines and α-methylene carbonyls are also considered Mannich reactions because these imines form between amines and aldehydes. The reaction is named after Carl Mannich. The Mannich reaction starts with the nucleophilic addition of an amine to a carbonyl group followed by dehydration to the Schiff base. The Schiff base is an electrophile which reacts in a second step in an electrophilic addition with an enol formed from a carbonyl compound containing an acidic alpha-proton. The Mannich reaction is a condensation reaction. In the Mannich reaction, primary or secondary amines or ammonia react with formaldehyde to form a Schiff base. T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stephen Aldehyde Synthesis
Stephen aldehyde synthesis, a named reaction in chemistry, was invented by Henry Stephen (chemist), Henry Stephen (Order of the British Empire, OBE/Member of the Order of the British Empire, MBE). This reaction involves the preparation of aldehydes (R-CHO) from nitriles (R-CN) using tin(II) chloride (SnCl2), hydrochloric acid (HCl) and quenching the resulting iminium salt ([R-CH=NH2]+Cl−) with Water (molecule), water (H2O). During the synthesis, ammonium chloride is also produced. It is a type of nucleophilic addition reaction. Mechanism The following scheme shows the reaction mechanism: By addition of hydrogen chloride the used nitrile (1) reacts to its corresponding salt (2). It is believed that this salt is reduced by a single electron transfer by the tin(II) chloride (3a and 3b). The resulting salt (4) precipitates after some time as aldimine tin chloride (5). Hydrolysis of 5 produces a hemiaminal (6) from which an aldehyde (7) is formed. Substitutes that increase the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Imine
In organic chemistry, an imine ( or ) is a functional group or organic compound containing a carbon–nitrogen double bond (). The nitrogen atom can be attached to a hydrogen or an organic group (R). The carbon atom has two additional single bonds. Imines are common in synthetic and naturally occurring compounds and they participate in many reactions. Distinction is sometimes made between aldimines and ketimines, derived from aldehydes and ketones, respectively. Structure In imines the five core atoms (C2C=NX, ketimine; and C(H)C=NX, aldimine; X = H or C) are coplanar. Planarity results from the sp2-hybridization of the mutually double-bonded carbon and the nitrogen atoms. The C=N distance is 1.29–1.31 Å for nonconjugated imines and 1.35 Å for conjugated imines. By contrast, C−N distances in amines and nitriles are 1.47 and 1.16 Å respectively. Rotation about the C=N bond is slow. Using NMR spectroscopy, both E–Z notation, ''E'' and ''Z'' isomers of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Beckmann Rearrangement
The Beckmann rearrangement, named after the German chemist Ernst Otto Beckmann (1853–1923), is a rearrangement reaction, rearrangement of an oxime functional group to substituted amides. The rearrangement has also been successfully performed on haloimines and nitrones. Cyclic oximes and haloimines yield lactams. The Beckmann rearrangement is often catalyzed by acid; however, other reagents have been known to promote the rearrangement. These include tosyl chloride, thionyl chloride, phosphorus pentachloride, phosphorus pentoxide, triethylamine, sodium hydroxide, trimethylsilyl iodide among others. The Beckmann fragmentation is another reaction that often competes with the rearrangement, though careful selection of promoting reagent and solvent conditions can favor the formation of one over the other, sometimes giving almost exclusively one product. The rearrangement occurs Stereospecificity, stereospecifically for ketoximes and N-chloro/N-fluoro imines, with the migrating group be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stork Enamine Alkylation
The Stork enamine alkylation involves the addition of an enamine to a Michael acceptor (e.g., an α,β -unsaturated carbonyl compound) or another electrophilic alkylation reagent to give an alkylated iminium product, which is hydrolyzed by dilute aqueous acid to give the alkylated ketone or aldehyde. Since enamines are generally produced from ketones or aldehydes, this overall process (known as the Stork enamine synthesis) constitutes a selective monoalkylation of a ketone or aldehyde, a process that may be difficult to achieve directly. The Stork enamine synthesis: # formation of an enamine from a ketone # addition of the enamine to an alpha, beta-unsaturated aldehyde or ketone # hydrolysis of the enamine back to a ketone The reaction also applies to acyl halides as electrophiles, which results in the formation of 1,3-diketones (Stork acylation). It is also effective for activated sp3 alkyl electrophiles, including benzylic, allylic/propargylic, α-carbonyl (e.g., bromoac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Duff Reaction
The Duff reaction or hexamine aromatic formylation is a formylation reaction used in organic chemistry for the synthesis of benzaldehydes with hexamine as the formyl carbon source. The method is generally inefficient. The reaction is named after James Cooper Duff. The reaction requires strongly polar effect, electron donating substituents on the aromatic ring such as in a phenol. Formylation occurs ''arene substitution patterns, ortho'' to the electron donating substituent preferentially, unless the ''ortho'' positions are blocked, in which case the formylation occurs at the ''para'' position. Examples The modified salicylaldehyde 3,5-di-tert-butylsalicylaldehyde, 3,5-di-''tert''-butylsalicylaldehyde is prepared by the Duff reaction: The natural product syringaldehyde can also be prepared by the Duff reaction. In this example, formylation occurs at the position para to the phenolic OH. Unlike other formylation reactions the Duff reaction is able to attach multiple aldehy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reductive Amination
Reductive amination (also known as reductive alkylation) is a form of amination that converts a carbonyl group to an amine via an intermediate imine. The carbonyl group is most commonly a ketone or an aldehyde. It is a common method to make amines and is widely used in green chemistry since it can be done catalytically in One-pot synthesis, one-pot under mild conditions. In biochemistry, Dehydrogenase, dehydrogenase enzymes use reductive amination to produce the amino acid Glutamic acid, glutamate. Additionally, there is ongoing research on alternative synthesis mechanisms with various metal catalysts which allow the reaction to be less energy taxing, and require milder reaction conditions. Investigation into biocatalysts, such as imine reductases, have allowed for higher selectivity in the reduction of chiral amines which is an important factor in pharmaceutical synthesis. Reaction process Reductive amination occurs between a carbonyl such as an aldehyde or ketone and an amine i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |