|
Heat Shrink Tubing
Heat-shrink tubing (or, commonly, ''heat shrink'' or ''heatshrink'') is a shrinkable plastic tube used to insulate wires, providing abrasion resistance and environmental protection for stranded and solid wire conductors, connections, joints and terminals in electrical wiring. It can also be used to repair the insulation on wires or to bundle them together, to protect wires or small parts from minor abrasion, and to create cable entry seals, offering environmental sealing protection. Heat-shrink tubing is ordinarily made of a polyolefin, which shrinks radially (but not longitudinally) when heated, to between one-half and one-sixth of its diameter. Heat-shrink tubing is manufactured in a multitude of varieties and chemical makeups with the exact composition of each type being dependent on the intended application. From near microscopically-thin-wall tubing to rigid, heavy-wall tubing, each type has precise design and chemical additives that make it suitable for meeting any of a wid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Citizens Band Radio
Citizens band radio (CB radio) is a land mobile radio system, a system allowing short-distance one-to-many bidirectional voice communication among individuals, using two-way radios operating near 27 MHz (or the 11-m wavelength) in the high frequency or shortwave band. Citizens band is distinct from other personal radio service allocations such as FRS, GMRS, MURS, UHF CB and the Amateur Radio Service ( "ham" radio). In many countries, CB operation does not require a license and may be used for business or personal communications. Like many other land mobile radio services, multiple radios in a local area share a single frequency channel, but only one can transmit at a time. The radio is normally in receive mode to receive transmissions of other radios on the channel; when users want to communicate they press a "push to talk" button on their radio, which turns on their transmitter. Users on a channel must take turns transmitting. In the US and Canada, and in the EU and th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyolefin
A polyolefin is a type of polymer with the general formula (CH2CHR)n where R is an alkyl group. They are usually derived from a small set of simple olefins (alkenes). Dominant in a commercial sense are polyethylene and polypropylene. More specialized polyolefins include Polyisobutene, polyisobutylene and polymethylpentene. They are all colorless or white oils or solids. Many copolymers are known, such as polybutene, which derives from a mixture of different butene isomers. The name of each polyolefin indicates the olefin from which it is prepared; for example, polyethylene is derived from ethylene, and polymethylpentene is derived from 4-methyl-1-pentene. Polyolefins are not olefins themselves because the double bond of each olefin monomer is opened in order to form the polymer. Monomers having more than one double bond such as butadiene and isoprene yield polymers that contain double bonds (polybutadiene and polyisoprene) and are usually not considered polyolefins. Polyolefins are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
UV Stabilizers In Plastics
Ultraviolet radiation, also known as simply UV, is electromagnetic radiation of wavelengths of 10–400 nanometers, shorter than that of visible light, but longer than X-rays. UV radiation is present in sunlight and constitutes about 10% of the total electromagnetic radiation output from the Sun. It is also produced by electric arcs, Cherenkov radiation, and specialized lights, such as mercury-vapor lamps, tanning lamps, and black lights. The photons of ultraviolet have greater energy than those of visible light, from about 3.1 to 12 electron volts, around the minimum energy required to ionize atoms. Although long-wavelength ultraviolet is not considered an ionizing radiation because its photons lack sufficient energy, it can induce chemical reactions and cause many substances to glow or fluoresce. Many practical applications, including chemical and biological effects, are derived from the way that UV radiation can interact with organic molecules. These interacti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Peroxide
In chemistry, peroxides are a group of Chemical compound, compounds with the structure , where the R's represent a radical (a portion of a complete molecule; not necessarily a free radical) and O's are single oxygen atoms. Oxygen atoms are joined to each other and to adjacent elements through Single bond, single covalent bonds, denoted by dashes or lines. The group in a peroxide is often called the peroxide group, though some nomenclature discrepancies exist. This linkage is recognized as a common polyatomic ion, and exists in many molecules. General structure The characteristic structure of any regular peroxide is the oxygen–oxygen covalent single bond, which connects the two main atoms together. In the event that the molecule has no chemical Substituent, substituents, the peroxide group will have a [−2] Formal charge, net charge. Each oxygen atom has a charge of negative one, as 5 of its Valence electron, valence electrons remain in the outermost Atomic orbital, orbital ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron Beam
Since the mid-20th century, electron-beam technology has provided the basis for a variety of novel and specialized applications in semiconductor manufacturing, microelectromechanical systems, nanoelectromechanical systems, and microscopy. Mechanism Free electrons in a vacuum can be manipulated by Electric field, electric and magnetic fields to form a fine beam. Where the beam collides with solid-state matter, electrons are converted into heat or kinetic energy. This concentration of energy in a small volume of matter can be precisely controlled by the fields, which brings many advantages. Applications Electron beam techniques include electron probe microanalysis, transmission electron microscopy, auger spectroscopy, and scanning electron microscopy. The rapid increase of temperature at the location of impact can quickly melt a target material. In extreme working conditions, the rapid temperature increase can lead to evaporation, making an electron beam an excellent tool in heating ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
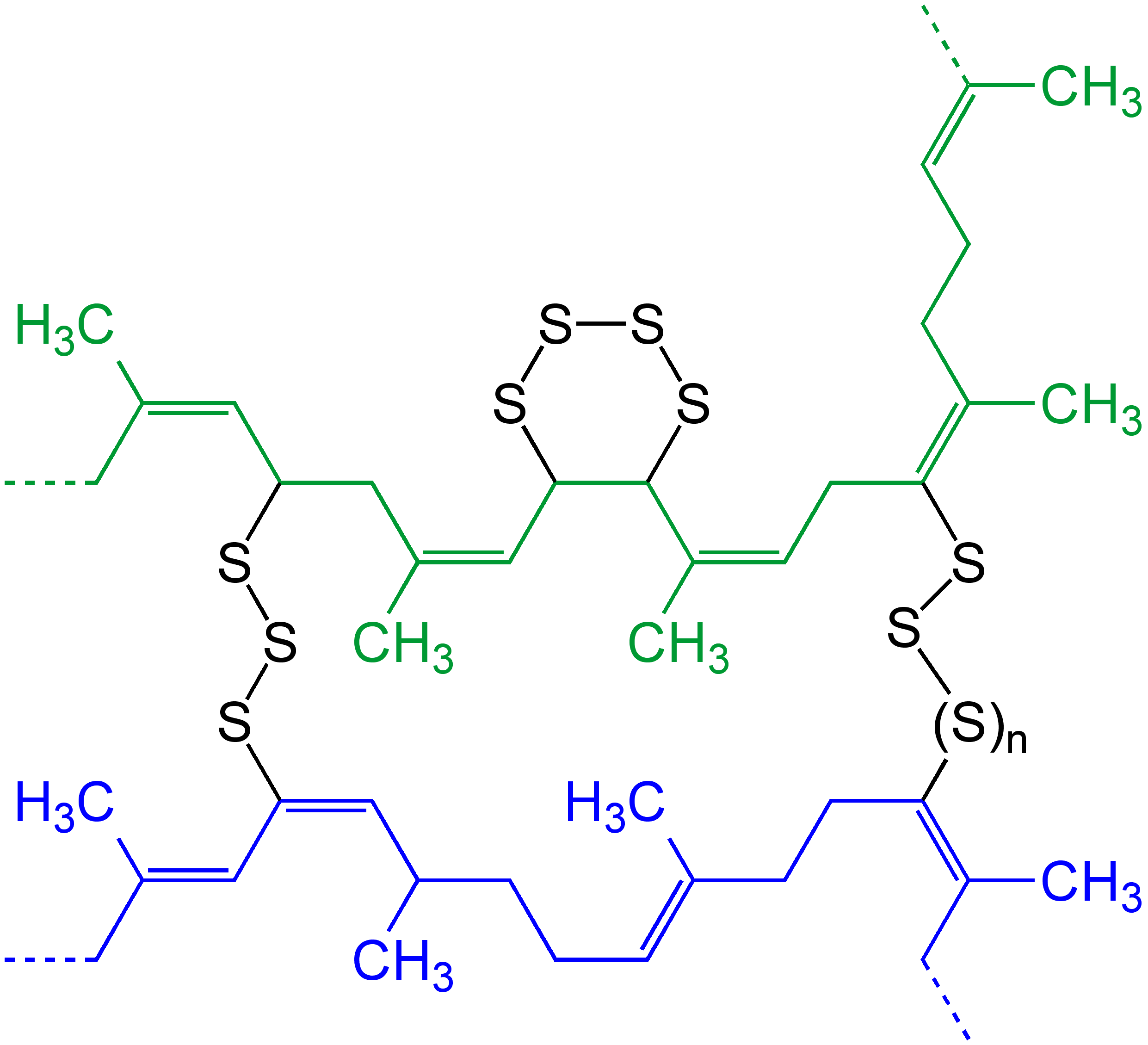
Cross-link
In chemistry and biology, a cross-link is a bond or a short sequence of bonds that links one polymer chain to another. These links may take the form of covalent bonds or ionic bonds and the polymers can be either synthetic polymers or natural polymers (such as proteins). In polymer chemistry "cross-linking" usually refers to the use of cross-links to promote a change in the polymers' physical properties. When "crosslinking" is used in the biological field, it refers to the use of a probe to link proteins together to check for protein–protein interactions, as well as other creative cross-linking methodologies. Although the term is used to refer to the "linking of polymer chains" for both sciences, the extent of crosslinking and specificities of the crosslinking agents vary greatly. Synthetic polymers : 260px, left, Chemical reactions associated with crosslinking of drying oils, the process that produces curing'' refers to the crosslinking of thermosetting">linoleum. Cros ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystallization Of Polymers
Crystallization of polymers is a process associated with partial alignment of their molecular chains. These chains fold together and form ordered regions called Lamella (materials), lamellae, which compose larger spheroidal structures named Spherulite (polymer physics), spherulites. Polymers can crystallize upon cooling from melting, mechanical stretching or solvent evaporation. Crystallization affects optical, mechanical, thermal and chemical properties of the polymer. The degree of crystallinity is estimated by different analytical methods and it typically ranges between 10 and 80%, with crystallized polymers often called "semi-crystalline". The properties of semi-crystalline polymers are determined not only by the degree of crystallinity, but also by the size and orientation of the molecular chains. Crystallization mechanisms Solidification from the melt Polymers are composed of long molecular chains which form irregular, entangled coils in the melt. Some polymers retain such ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neoprene
Neoprene (also polychloroprene) is a family of synthetic rubbers that are produced by polymerization of chloroprene.Werner Obrecht, Jean-Pierre Lambert, Michael Happ, Christiane Oppenheimer-Stix, John Dunn and Ralf Krüger "Rubber, 4. Emulsion Rubbers" in Ullmann's Encyclopedia of Industrial Chemistry, 2012, Wiley-VCH, Weinheim. Neoprene exhibits good chemical stability and maintains flexibility over a wide temperature range. Neoprene is sold either as solid rubber or in latex form and is used in a wide variety of commercial applications, such as laptop sleeves, orthopaedic braces (wrist, knee, etc.), electrical insulation, medical gloves, liquid and sheet-applied elastomeric membranes or flashings, and Motor vehicle, automotive fan belt (mechanical), belts. Production Neoprene is produced by free-radical polymerization of chloroprene. In commercial production, this polymer is prepared by Emulsion_polymerization, free radical emulsion polymerization. Polymerization is initiated us ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyvinyl Chloride
Polyvinyl chloride (alternatively: poly(vinyl chloride), colloquial: vinyl or polyvinyl; abbreviated: PVC) is the world's third-most widely produced synthetic polymer of plastic (after polyethylene and polypropylene). About 40 million tons of PVC are produced each year. PVC comes in rigid (sometimes abbreviated as RPVC) and flexible forms. Rigid PVC is used in construction for pipes, doors and windows. It is also used in making plastic bottles, packaging, and bank or membership cards. Adding plasticizers makes PVC softer and more flexible. It is used in plumbing, electrical cable insulation, flooring, signage, phonograph records, inflatable products, and in rubber substitutes. With cotton or linen, it is used in the production of canvas. Polyvinyl chloride is a white, brittle solid. It is soluble in ketones, chlorinated solvents, dimethylformamide, THF and DMAc. Discovery PVC was synthesized in 1872 by German chemist Eugen Baumann after extended investigation and experimenta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kynar
Polyvinylidene fluoride or polyvinylidene difluoride (PVDF) is a highly non-reactive thermoplastic fluoropolymer produced by the polymerization of vinylidene difluoride. Its chemical formula is (C2H2F2)''n''. PVDF is a specialty plastic used in applications requiring the highest purity, as well as resistance to solvents, acids and hydrocarbons. PVDF has low density 1.78 g/cm3 in comparison to other fluoropolymers, like polytetrafluoroethylene. It is available in the form of piping products, sheet, tubing, films, plate and an insulator for premium wire. It can be injected, molded or welded and is commonly used in the chemical, semiconductor, medical and defense industries, as well as in lithium-ion batteries. It is also available as a cross-linked closed-cell foam, used increasingly in aviation and aerospace applications, and as an exotic 3D printer filament. It can also be used in repeated contact with food products, as it is FDA-compliant and non-toxic below its degradati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |