|
Glutamate–cysteine Ligase
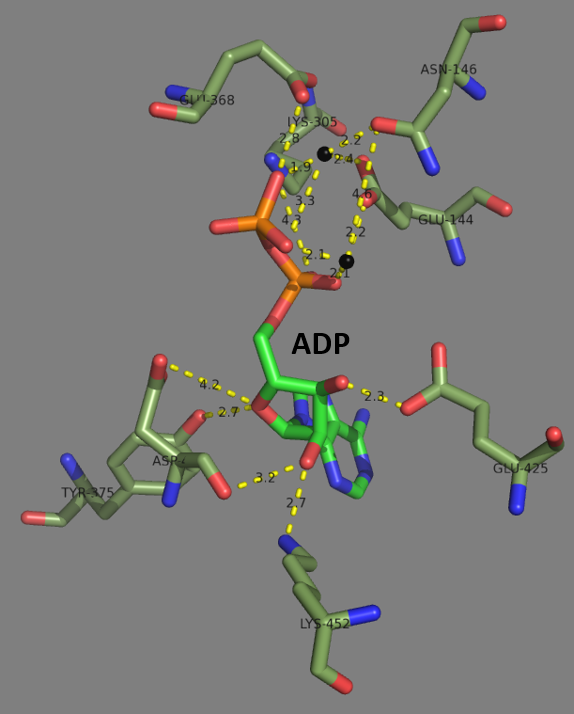
Glutamate–cysteine ligase (GCL) ), previously known as γ-glutamylcysteine synthetase (GCS), is the first enzyme of the cellular glutathione (GSH) biosynthetic pathway that catalyzes the chemical reaction: L-glutamate + L-cysteine + ATP \rightleftharpoons γ-glutamyl cysteine + ADP + Pi GSH, and by extension GCL, is critical to cell survival. Nearly every eukaryotic cell, from plants to yeast to humans, expresses a form of the GCL protein for the purpose of synthesizing GSH. To further highlight the critical nature of this enzyme, genetic knockout of GCL results in embryonic lethality. Furthermore, dysregulation of GCL enzymatic function and activity is known to be involved in the vast majority of human diseases, such as diabetes, Parkinson's disease, Alzheimer's disease, COPD, HIV/AIDS, and cancer. This typically involves impaired function leading to decreased GSH biosynthesis, reduced cellular antioxidant capacity, and the induction of oxidative stress. However, in cancer, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glutathione
Glutathione (GSH, ) is an organic compound with the chemical formula . It is an antioxidant in plants, animals, fungi, and some bacteria and archaea. Glutathione is capable of preventing damage to important cellular components caused by sources such as reactive oxygen species, free radicals, peroxides, lipid peroxides, and heavy metals. It is a tripeptide with a gamma peptide linkage between the carboxyl group of the glutamate side chain and cysteine. The carboxyl group of the cysteine residue is attached by normal peptide linkage to glycine. Biosynthesis and occurrence Glutathione biosynthesis involves two adenosine triphosphate-dependent steps: *First, γ-glutamylcysteine is synthesized from L-glutamate and L-cysteine. This conversion requires the enzyme glutamate–cysteine ligase (GCL, glutamate cysteine synthase). This reaction is the rate-limiting step in glutathione synthesis. *Second, glycine is added to the C-terminal of γ-glutamylcysteine. This condensation is ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glutathione Disulfide
Glutathione disulfide (GSSG) is a disulfide derived from two glutathione molecules. In living cells, glutathione disulfide is reduced into two molecules of glutathione with reducing equivalents from the coenzyme NADPH. This reaction is catalyzed by the enzyme glutathione reductase. Antioxidant enzymes, such as glutathione peroxidases and peroxiredoxins, generate glutathione disulfide during the reduction of peroxides such as hydrogen peroxide (H2O2) and organic hydroperoxides (ROOH): :2 GSH + ROOH → GSSG + ROH + H2O Other enzymes, such as glutaredoxins, generate glutathione disulfide through thiol-disulfide exchange with protein disulfide bonds or other low molecular mass compounds, such as coenzyme A disulfide or dehydroascorbic acid. :2 GSH + R-S-S-R → GSSG + 2 RSH The GSH:GSSG ratio is therefore an important bioindicator of cellular health, with a higher ratio signifying less oxidative stress in the organism. A lower ratio may even be indicative of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Data Bank
The Protein Data Bank (PDB) is a database for the three-dimensional structural data of large biological molecules such as proteins and nucleic acids, which is overseen by the Worldwide Protein Data Bank (wwPDB). This structural data is obtained and deposited by biologists and biochemists worldwide through the use of experimental methodologies such as X-ray crystallography, Nuclear magnetic resonance spectroscopy of proteins, NMR spectroscopy, and, increasingly, cryo-electron microscopy. All submitted data are reviewed by expert Biocuration, biocurators and, once approved, are made freely available on the Internet under the CC0 Public Domain Dedication. Global access to the data is provided by the websites of the wwPDB member organizations (PDBe, PDBj, RCSB PDB, and BMRB). The PDB is a key in areas of structural biology, such as structural genomics. Most major scientific journals and some funding agencies now require scientists to submit their structure data to the PDB. Many other ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tertiary Structure
Protein tertiary structure is the three-dimensional shape of a protein. The tertiary structure will have a single polypeptide chain "backbone" with one or more protein secondary structures, the protein domains. Amino acid side chains and the backbone may interact and bond in a number of ways. The interactions and bonds of side chains within a particular protein determine its tertiary structure. The protein tertiary structure is defined by its atomic coordinates. These coordinates may refer either to a protein domain or to the entire tertiary structure. A number of these structures may bind to each other, forming a quaternary structure. History The science of the tertiary structure of proteins has progressed from one of hypothesis to one of detailed definition. Although Emil Fischer had suggested proteins were made of polypeptide chains and amino acid side chains, it was Dorothy Maud Wrinch who incorporated geometry into the prediction of protein structures. Wrinch demon ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glutathione Synthetase
Glutathione synthetase (GSS) () is the second enzyme in the glutathione (GSH) biosynthesis pathway. It catalyses the condensation of gamma-glutamylcysteine and glycine, to form glutathione. Glutathione synthetase is also a potent antioxidant. It is found in many species including bacteria, yeast, mammals, and plants. In humans, defects in GSS are inherited in an autosomal recessive way and are the cause of severe metabolic acidosis, 5-oxoprolinuria, increased rate of haemolysis, and defective function of the central nervous system. Deficiencies in GSS can cause a spectrum of deleterious symptoms in plants and human beings alike. In eukaryotes, this is a homodimeric enzyme. The substrate-binding domain has a three-layer alpha/beta/alpha structure. This enzyme utilizes and stabilizes an acylphosphate intermediate to later perform a favorable nucleophilic attack of glycine. Structure Human and yeast glutathione synthetases are homodimers, meaning they are composed of two ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Plastid
A plastid is a membrane-bound organelle found in the Cell (biology), cells of plants, algae, and some other eukaryotic organisms. Plastids are considered to be intracellular endosymbiotic cyanobacteria. Examples of plastids include chloroplasts (used for photosynthesis); chromoplasts (used for synthesis and storage of pigments); leucoplasts (non-pigmented plastids, some of which can cellular differentiation, differentiate); and apicoplasts (non-photosynthetic plastids of apicomplexa derived from secondary endosymbiosis). A permanent primary endosymbiosis event occurred about 1.5 billion years ago in the Archaeplastida cladeEmbryophyte, land plants, red algae, green algae and glaucophytesprobably with a cyanobiont, a symbiotic cyanobacteria related to the genus ''Gloeomargarita lithophora, Gloeomargarita''. Another primary endosymbiosis event occurred later, between 140 and 90 million years ago, in the photosynthetic plastids ''Paulinella'' amoeboids of the cyanobacteria genera '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homodimer
In biochemistry, a protein dimer is a macromolecular complex or protein multimer, multimer formed by two protein monomers, or single proteins, which are usually Non-covalent interaction, non-covalently bound. Many macromolecules, such as proteins or nucleic acids, form dimers. The word ''dimer'' has roots meaning "two parts", ''wikt:di-#Prefix, di-'' + ''wikt:-mer#Suffix, -mer''. A protein dimer is a type of protein quaternary structure. A protein homodimer is formed by two identical proteins while a protein heterodimer is formed by two different proteins. Most protein dimers in biochemistry are not connected by covalent bonds. An example of a non-covalent heterodimer is the enzyme reverse transcriptase, which is composed of two different amino acid chains. An exception is dimers that are linked by disulfide bridges such as the homodimeric protein IKBKG, NEMO. Some proteins contain specialized domains to ensure dimerization (dimerization domains) and specificity. The G protein- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
GCLM
Glutamate-cysteine ligase regulatory subunit is an enzyme that in humans is encoded by the ''GCLM'' gene. Glutamate-cysteine ligase, also known as gamma-glutamylcysteine synthetase, is the first rate limiting enzyme of glutathione synthesis. The enzyme consists of two subunits, a heavy catalytic subunit and a light regulatory subunit. Gamma glutamylcysteine synthetase deficiency has been implicated in some forms of hemolytic anemia Hemolytic anemia or haemolytic anaemia is a form of anemia due to hemolysis, the abnormal breakdown of red blood cells (RBCs), either in the blood vessels (intravascular hemolysis) or elsewhere in the human body (extravascular). This most commonl .... References Further reading * * * * * * * * * * * * * * * * * * {{gene-1-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
GCLC
Glutamate–cysteine ligase catalytic subunit is an enzyme that in humans is encoded by the ''GCLC'' gene In biology, the word gene has two meanings. The Mendelian gene is a basic unit of heredity. The molecular gene is a sequence of nucleotides in DNA that is transcribed to produce a functional RNA. There are two types of molecular genes: protei .... Function Glutamate–cysteine ligase, also known as gamma-glutamylcysteine synthetase is the first rate limiting enzyme of glutathione synthesis. The enzyme consists of two subunits, a heavy catalytic subunit and a light regulatory subunit. The gene encoding the catalytic subunit encodes a protein of 367 amino acids with a calculated molecular weight of 72.773 kDa and maps to chromosome 6. The regulatory subunit is derived from a different gene located on chromosome 1p22-p21. Deficiency of gamma-glutamylcysteine synthetase in human is associated with enzymopathic hemolytic anemia. References Further reading * * * ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residue (biochemistry), residues. Proteins perform a vast array of functions within organisms, including Enzyme catalysis, catalysing metabolic reactions, DNA replication, Cell signaling, responding to stimuli, providing Cytoskeleton, structure to cells and Fibrous protein, organisms, and Intracellular transport, transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the Nucleic acid sequence, nucleotide sequence of their genes, and which usually results in protein folding into a specific Protein structure, 3D structure that determines its activity. A linear chain of amino acid residues is called a polypeptide. A protein contains at least one long polypeptide. Short polypeptides, containing less than 20–30 residues, are rarely considered to be proteins and are commonly called pep ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heterodimer
In biochemistry, a protein dimer is a macromolecular complex or multimer formed by two protein monomers, or single proteins, which are usually non-covalently bound. Many macromolecules, such as proteins or nucleic acids, form dimers. The word ''dimer'' has roots meaning "two parts", '' di-'' + '' -mer''. A protein dimer is a type of protein quaternary structure. A protein homodimer is formed by two identical proteins while a protein heterodimer is formed by two different proteins. Most protein dimers in biochemistry are not connected by covalent bonds. An example of a non-covalent heterodimer is the enzyme reverse transcriptase, which is composed of two different amino acid chains. An exception is dimers that are linked by disulfide bridges such as the homodimeric protein NEMO. Some proteins contain specialized domains to ensure dimerization (dimerization domains) and specificity. The G protein-coupled cannabinoid receptors have the ability to form both homo- and hetero ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Toxicological Sciences
''Toxicological Sciences'' is a monthly peer-reviewed scientific journal which covers all aspects of research on toxicology. It is published by Oxford University Press on behalf of the Society of Toxicology. It was established in 1981 as ''Fundamental and Applied Toxicology'' and obtained its current name in 1998. The current editor-in-chief is Jeffrey M. Peters, a professor of molecular toxicology and carcinogenesis at The Pennsylvania State University, and the Managing Editor is Virginia F. Hawkins. The editorial staff also includes Associate Editors in subject areas and an editorial board of topic experts. While its ISO 4 abbreviation is ''Toxicol. Sci.'' it is commonly referred to as ''ToxSci''. Abstracting and indexing The journal is abstracted and indexed in Biological Abstracts, BIOSIS, CAB International, Chemical Abstracts Service, Current Contents, EMBASE, Health & Safety Science Abstracts, Science Citation Index, and Toxicology Abstracts. According to the ''Jou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |