|
Dithionite
The dithionite is the oxyanion with the formula 2O4sup>2−. It is commonly encountered as the salt sodium dithionite. For historical reasons, it is sometimes called hydrosulfite, but it contains no hydrogen and is not a sulfite. The dianion has a steric number of 4 and trigonal pyramidal geometry. Production and reactions In its main applications, dithionite is generally prepared ''in situ'' by reduction of sulfur dioxide by sodium borohydride, described by the following idealized equation: : Dithionite is a reducing agent. At pH 7, its reduction potential is −0.66 V vs SHE. Its oxidation occurs with formation of sulfite: : + 2 H2O → 2 + 2 e− + 2 H+ Dithionite undergoes acid hydrolytic disproportionation to thiosulfate and bisulfite: : 2 + H2O → + 2 It also undergoes alkaline hydrolytic disproportionation to sulfite and sulfide: : 3 Na2S2O4 + 6 NaOH → 5 Na2SO3 + Na2S + 3 H2O It is formally derived from dithionous acid (H2S2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Dithionite
Sodium dithionite (also known as sodium hydrosulfite) is a white crystalline powder with a sulfurous odor. Although it is stable in dry air, it decomposes in hot water and in acid solutions. Structure left, 220px, Packing of sodium dithionite ions in a crystal, showing the saw-horse geometry of the dianion. Color code: red = O, yellow = S. The structure has been examined by Raman spectroscopy and X-ray crystallography. The dithionite dianion has C symmetry, with almost eclipsed with a 16° O-S-S-O torsional angle. In the dihydrated form (), the dithionite anion has gauche 56° O-S-S-O torsional angle. A weak S-S bond is indicated by the S-S distance of 239 pm, which is elongated by ca. 30 pm relative to a typical S-S bond. Because this bond is fragile, the dithionite anion dissociates in solution into the O2sup>− radicals, as has been confirmed by EPR spectroscopy. It is also observed that 35S undergoes rapid exchange between S2O42− and SO2 in neutral or acidic solu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bleaching Of Wood Pulp
Bleaching of wood pulp is the chemical processing of wood pulp to lighten its color and whiten the pulp. The primary product of wood pulp is paper, for which whiteness (similar to, but distinct from brightness) is an important characteristic. These processes and chemistry are also applicable to the bleaching of non-wood pulps, such as those made from bamboo or kenaf. Paper brightness Brightness is the amount of incident light reflected from paper under specified conditions, usually reported as the percentage of light reflected, so a higher number means a brighter or whiter paper. In the US, the TAPPI T 452 or T 525 standards are used. The international community uses ISO standards. The table shows how the two systems rate high-brightness papers, but there is no simple way to convert between the two systems because the test methods are so different. The ISO rating is higher and can be over 100. This is because contemporary white paper incorporates fluorescent whiten ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
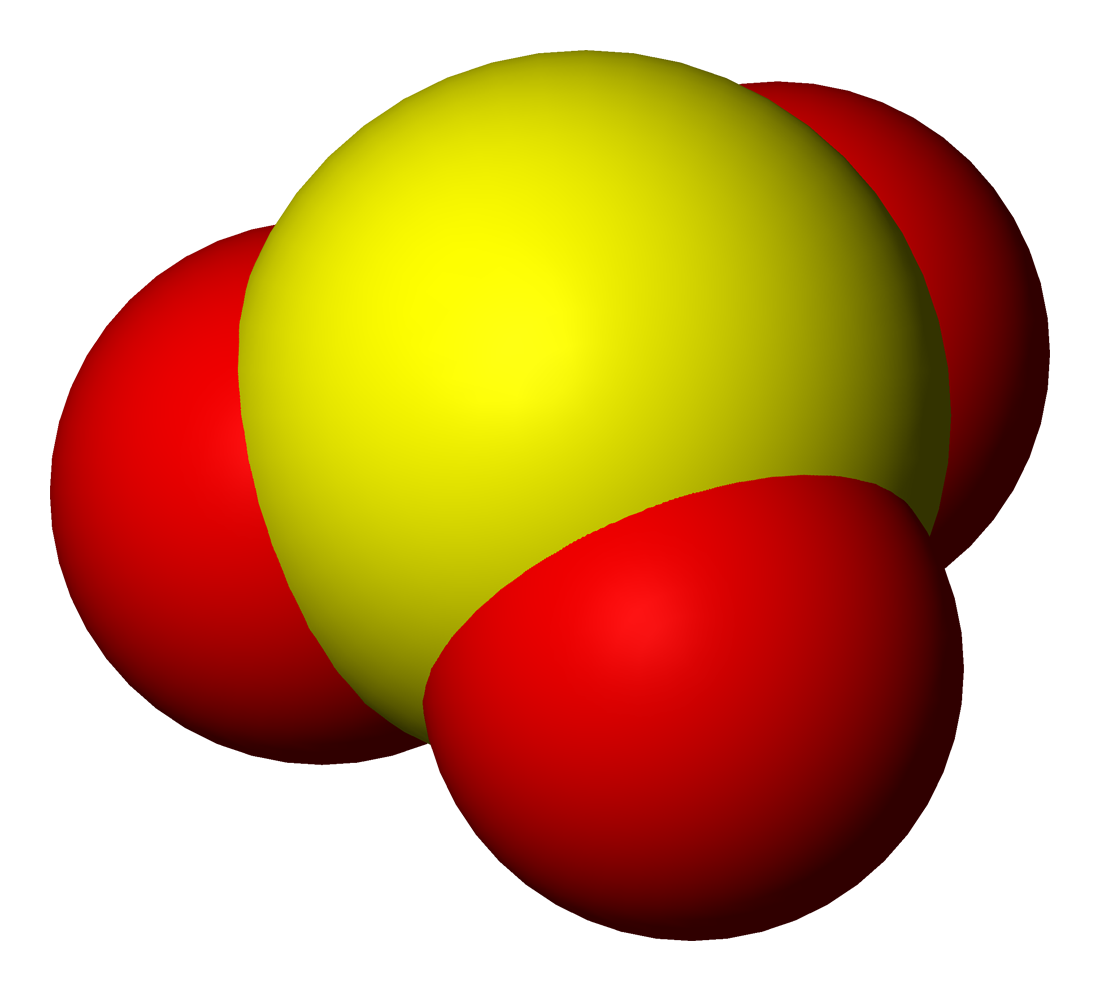
Dithionous Acid
Dithionous acid is a sulfur oxoacid with the chemical formula H2S2O4. It has not been observed experimentally. It is the conjugate acid A conjugate acid, within the Brønsted–Lowry acid–base theory, is a chemical compound formed when an acid gives a proton () to a base—in other words, it is a base with a hydrogen ion added to it, as it loses a hydrogen ion in the rever ... of the dithionite dianion, S2O42-. Dithionite is a well-known reducing agent. Related compounds * Dithionic acid (H2S2O6), another unstable protonated sulfur oxide, derived from dithionate. References Sulfur oxoacids Dithionites {{inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Borohydride
Sodium borohydride, also known as sodium tetrahydridoborate and sodium tetrahydroborate, is an inorganic compound with the formula (sometimes written as ). It is a white crystalline solid, usually encountered as an aqueous basic solution. Sodium borohydride is a reducing agent that finds application in papermaking and dye industries. It is also used as a reagent in organic synthesis. The compound was discovered in the 1940s by H. I. Schlesinger, who led a team seeking volatile uranium compounds.Hermann I Schlesinger and Herbert C Brown (1945)Preparation of alkali metal compounds. US Patent 2461661. Granted on 1949-02-15; expired on 1966-02-15. Results of this wartime research were declassified and published in 1953. Properties The compound is soluble in alcohols, certain ethers, and water, although it slowly hydrolyzes. Sodium borohydride is an odorless white to gray-white microcrystalline powder that often forms lumps. It can be purified by recrystallization from warm ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfite
Sulfites or sulphites are compounds that contain the sulfite ion (systematic name: sulfate(IV) ion), . The sulfite ion is the conjugate base of bisulfite. Although its acid (sulfurous acid) is elusive, its salts are widely used. Sulfites are substances that naturally occur in some foods and the human body. They are also used as regulated food additives. When in food or drink, sulfites are often lumped together with sulfur dioxide.SeREGULATION (EU) No 1169/2011 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL/ref> Structure The structure of the sulfite anion can be described with three equivalent resonance structures. In each resonance structure, the sulfur atom is double-bonded to one oxygen atom with a formal charge of zero (neutral), and sulfur is singly bonded to the other two oxygen atoms, which each carry a formal charge of −1, together accounting for the −2 charge on the anion. There is also a non-bonded lone pair on the sulfur, so the structure predicted by VSEPR ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solubility
In chemistry, solubility is the ability of a chemical substance, substance, the solute, to form a solution (chemistry), solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution. The extent of the solubility of a substance in a specific solvent is generally measured as the concentration of the solute in a wikt:saturated#Chemistry, saturated solution, one in which no more solute can be dissolved. At this point, the two substances are said to be at the solubility equilibrium. For some solutes and solvents, there may be no such limit, in which case the two substances are said to be "miscibility, miscible in all proportions" (or just "miscible"). The solute can be a solid, a liquid, or a gas, while the solvent is usually solid or liquid. Both may be pure substances, or may themselves be solutions. Gases are always miscible in all proportions, except in very extreme situations,J. de Swaan Arons and G. A. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chelation
Chelation () is a type of bonding of ions and their molecules to metal ions. It involves the formation or presence of two or more separate coordinate bonds between a polydentate (multiple bonded) ligand and a single central metal atom. These ligands are called chelants, chelators, chelating agents, or sequestering agents. They are usually organic compounds, but this is not a necessity. The word ''chelation'' is derived from Greek χηλή, ''chēlē'', meaning "claw"; the ligands lie around the central atom like the claws of a crab. The term ''chelate'' () was first applied in 1920 by Sir Gilbert T. Morgan and H. D. K. Drew, who stated: "The adjective chelate, derived from the great claw or ''chele'' (Greek) of the crab or other crustaceans, is suggested for the caliperlike groups which function as two associating units and fasten to the central atom so as to produce heterocyclic rings." Chelation is useful in applications such as providing nutritional supplements, in chel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Citric Acid
Citric acid is an organic compound with the formula . It is a Transparency and translucency, colorless Weak acid, weak organic acid. It occurs naturally in Citrus, citrus fruits. In biochemistry, it is an intermediate in the citric acid cycle, which occurs in the metabolism of all aerobic organisms. More than two million tons of citric acid Commodity chemicals, are manufactured every year. It is used widely as acidifier, flavoring, preservative, and chelating agent. A citrate is a derivative of citric acid; that is, the salt (chemistry), salts, esters, and the polyatomic ion, polyatomic anion found in solutions and salts of citric acid. An example of the former, a salt is trisodium citrate; an ester is triethyl citrate. When citrate anion, trianion is part of a salt, the formula of the citrate trianion is written as or . Natural occurrence and industrial production Citric acid occurs in a variety of fruits and vegetables, most notably Citrus, citrus fruits. Lemons and Lime ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron
Iron is a chemical element; it has symbol Fe () and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, forming much of Earth's outer and inner core. It is the fourth most abundant element in the Earth's crust, being mainly deposited by meteorites in its metallic state. Extracting usable metal from iron ores requires kilns or furnaces capable of reaching , about 500 °C (900 °F) higher than that required to smelt copper. Humans started to master that process in Eurasia during the 2nd millennium BC and the use of iron tools and weapons began to displace copper alloys – in some regions, only around 1200 BC. That event is considered the transition from the Bronze Age to the Iron Age. In the modern world, iron alloys, such as steel, stainless steel, cast iron and special steels, are by far the most common industrial metals, due to their mechan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Soil
Soil, also commonly referred to as earth, is a mixture of organic matter, minerals, gases, water, and organisms that together support the life of plants and soil organisms. Some scientific definitions distinguish dirt from ''soil'' by restricting the former term specifically to displaced soil. Soil consists of a solid collection of minerals and organic matter (the soil matrix), as well as a porous phase that holds gases (the soil atmosphere) and water (the soil solution). Accordingly, soil is a three- state system of solids, liquids, and gases. Soil is a product of several factors: the influence of climate, relief (elevation, orientation, and slope of terrain), organisms, and the soil's parent materials (original minerals) interacting over time. It continually undergoes development by way of numerous physical, chemical and biological processes, which include weathering with associated erosion. Given its complexity and strong internal connectedness, soil ecologists ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |