|
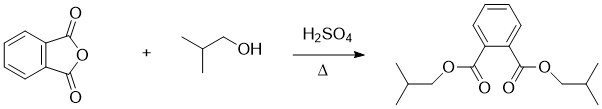
Diisobutyl Phthalate
Diisobutyl phthalate (DIBP) is a phthalate ester having the structural formula . It is formed by the esterification of isobutanol and phthalic anhydride. This and other phthalates are used as plasticizers due to their flexibility and durability. They are found in many industrial and personal products, such as lacquers, nail polish and cosmetics. DIBP can be absorbed via oral ingestion and dermal exposure. When it comes to excretion, DIBP is first converted into the hydrolytic monoester monoisobutyl phthalate (MIBP). The primary excretory route is urine, with Bile duct, biliary excretion being noted in minor amounts. DIBP has lower density and freezing point than the related compound dibutyl phthalate (DBP). Industry use It is used as a plasticizer additive in a range of plastic and rubber materials. It has low volatility, which makes it ideal for use in products that require long-lasting flexibility, e.g. automotive parts, wire and cable insulation, and flooring. It is dense and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phthalate
Phthalates ( ), or phthalate esters, are esters of phthalic acid. They are mainly used as plasticizers, i.e., substances added to plastics to increase their flexibility, transparency, durability, and longevity. They are used primarily to soften polyvinyl chloride (PVC). While phthalates are commonly used as plasticizers, not all plasticizers are phthalates. The two terms are specific, unique, and not used interchangeably. Lower-molecular-weight phthalates are typically replaced in many products in the United States, Canada, and European Union over health concerns. They are being replaced by higher molecular-weight phthalates as well as non-phthalic plasticizers. Phthalates are commonly ingested in small quantities via the diet. One of the most commonly known phthalates is bis(2-ethylhexyl) phthalate (DEHP). In many countries, DEHP is regulated as a toxin, and is banned from use in broad categories of consumer goods, such as cosmetics, children's toys, medical devices, and fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Filtration
Filtration is a physical separation process that separates solid matter and fluid from a mixture using a ''filter medium'' that has a complex structure through which only the fluid can pass. Solid particles that cannot pass through the filter medium are described as ''oversize'' and the fluid that passes through is called the ''filtrate''. Oversize particles may form a filter cake on top of the filter and may also block the filter lattice, preventing the fluid phase from crossing the filter, known as ''blinding''. The size of the largest particles that can successfully pass through a filter is called the effective ''pore size'' of that filter. The separation of solid and fluid is imperfect; solids will be contaminated with some fluid and filtrate will contain fine particles (depending on the pore size, filter thickness and biological activity). Filtration occurs both in nature and in engineered systems; there are biological, geological, and industrial forms. In everyday us ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biodegradation
Biodegradation is the breakdown of organic matter by microorganisms, such as bacteria and fungi. It is generally assumed to be a natural process, which differentiates it from composting. Composting is a human-driven process in which biodegradation occurs under a specific set of circumstances. The process of biodegradation is threefold: first an object undergoes biodeterioration, which is the mechanical weakening of its structure; then follows biofragmentation, which is the breakdown of materials by microorganisms; and finally assimilation, which is the incorporation of the old material into new cells. In practice, almost all chemical compounds and materials are subject to biodegradation, the key element being time. Things like vegetables may degrade within days, while glass and some plastics take many millennia to decompose. A standard for biodegradability used by the European Union is that greater than 90% of the original material must be converted into , water and minerals b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isobutyraldehyde
Isobutyraldehyde is the chemical compound with the formula (CH3)2CHCHO. It is an aldehyde, isomeric with ''n''-butyraldehyde (butanal). Isobutyraldehyde is made, often as a side-product, by the hydroformylation of propene. Its odour is described as that of wet cereal or straw. It undergoes the Cannizzaro reaction even though it has alpha hydrogen atom. It is a colorless volatile liquid. Synthesis Isobutyraldehyde is produced industrially by the hydroformylation of propene. Several million tons are produced annually.Boy Cornils, Richard W. Fischer, Christian Kohlpaintner "Butanals" in Ullmann's Encyclopedia of Industrial Chemistry, 2000, Wiley-VCH, Weinheim. Biological routes In the context of butanol fuel, isobutyraldehyde is of interest as a precursor to isobutanol. ''E. coli'' as well as several other organisms has been genetically modified to produce isobutanol. α- Ketoisovalerate, derived from oxidative deamination of valine, is prone to decarboxylation to give isobutyr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photodegradation
Photodegradation is the alteration of materials by light. Commonly, the term is used loosely to refer to the combined action of sunlight and air, which cause oxidation and hydrolysis. Often photodegradation is intentionally avoided, since it destroys paintings and other artifacts. It is, however, partly responsible for remineralization of biomass and is used intentionally in some disinfection technologies. Photodegradation does not apply to how materials may be aged or degraded via infrared light or heat, but does include degradation in all of the ultraviolet light wavebands. Applications Foodstuffs The protection of food from photodegradation is very important. Some nutrients, for example, are affected by degradation when exposed to sunlight. In the case of beer, UV radiation causes a process that entails the degradation of hop bitter compounds to 3-methyl-2-buten-1-thiol and therefore changes the taste. As amber-colored glass has the ability to absorb UV radiation, beer bottles ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isobutanol
Isobutanol (IUPAC nomenclature: 2-methylpropan-1-ol) is an organic compound with the formula (CH3)2CHCH2OH (sometimes represented as ''i''-BuOH). This colorless, flammable liquid with a characteristic smell is mainly used as a solvent either directly or as its esters. Its isomers are 1-butanol, 2-butanol, and ''tert''-butanol, all of which are important industrially. History and occurrence Fusel alcohols including isobutanol are grain fermentation byproducts. Therefore, trace amounts of isobutanol may be present in many alcoholic beverages. In 1852 Charles Adolphe Wurtz subjected such alcohols to fractional distillation and identified in some of them a butylic alcohol boiling at around 108°C. Its structure was initially unclear, with some chemists believing it corresponded to butyric acid, but theoretical considerations indicated that normal butanol should have a higher boiling point, and in 1867 Emil Erlenmeyer and independently Vladimir Markovnikov determined its actua ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phthalic Acid
In organic chemistry, phthalic acid is an aromatic dicarboxylic acid, with formula and structure . Although phthalic acid is of modest commercial importance, the closely related derivative phthalic anhydride is a commodity chemical produced on a large scale. Phthalic acid is one of three isomers of benzenedicarboxylic acid, the others being isophthalic acid and terephthalic acid. Production Phthalic acid is produced by the catalytic oxidation of naphthalene or ''ortho-''xylene directly to phthalic anhydride and a subsequent hydrolysis of the anhydride. Phthalic acid was first obtained by French chemist Auguste Laurent in 1836 by oxidizing naphthalene tetrachloride. Believing the resulting substance to be a naphthalene derivative, he named it "naphthalic acid". After the Swiss chemist Jean Charles Galissard de Marignac determined its correct formula, Laurent gave it its present name. Manufacturing methods in the nineteenth century included oxidation of naphthalene tetrach ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of Biomolecule, biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adhesive
Adhesive, also known as glue, cement, mucilage, or paste, is any non-metallic substance applied to one or both surfaces of two separate items that binds them together and resists their separation. The use of adhesives offers certain advantages over other binding techniques such as sewing, mechanical fastenings, and welding. These include the ability to bind different materials together, the more efficient distribution of stress across a joint, the cost-effectiveness of an easily mechanized process, and greater flexibility in design. Disadvantages of adhesive use include decreased stability at high temperatures, relative weakness in bonding large objects with a small bonding surface area, and greater difficulty in separating objects during testing. Adhesives are typically organized by the method of adhesion followed by ''reactive'' or ''non-reactive'', a term which refers to whether the adhesive chemically reacts in order to harden. Alternatively, they can be organized either ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bis(2-ethylhexyl) Phthalate
Bis(2-ethylhexyl) phthalate, di-2-ethylhexyl phthalate, diethylhexyl phthalate, diisooctyl phthalate, DEHP; incorrectly — dioctyl phthalate, DIOP) is an organic compound with the formula C6H4(CO2C8H17)2. DEHP is the most common member of the class of phthalates, which are used as plasticizers. It is the diester of phthalic acid and the branched-chain 2-ethylhexanol. This colorless viscous liquid is soluble in oil, but not in water. Production Di(2-ethylhexyl) phthalate is produced commercially by the reaction of excess 2-ethylhexanol with phthalic anhydride in the presence of an acid catalyst such as sulfuric acid or ''para''-toluenesulfonic acid. It was first produced in commercial quantities in Japan circa 1933 and in the United States in 1939. : DEHP has two stereocenters,Sheikh, I. A. (2016) Stereoselectivity and the potential endocrine disrupting activity of di-(2-ethylhexyl)phthalate (DEHP) against human progesterone receptor: a computational perspective. Journal of a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diisononyl Phthalate
Diisononyl phthalate (DINP) is a phthalate used as a plasticizer. DINP is typically a mixture of chemical compounds consisting of various isononyl esters of phthalic acid۔ Use It is commonly used in a large variety of plastic products including: * Plasticizer in polyvinyl chloride (PVC) * Construction and building material * Fuel and automotive products * In electronics materials (as a flexible vinyl additive) * As cable insulation additive * Adhesives and sealants * Paints and coatings * Rubber products. Health issues The European Union has set a maximum specific migration limit (SML) from food contact materials of 9 mg/kg food for the sum of diisononyl phthalates and diisodecyl phthalates. DINP is listed as a substance "known to the State of California to cause cancer" under Proposition 65 legislation. Studies find that exposure to environmentally relevant concentrations of DINP in zebrafish disrupt the endocannabinoid system (ECS) and affect reproduction in a ge ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |