|
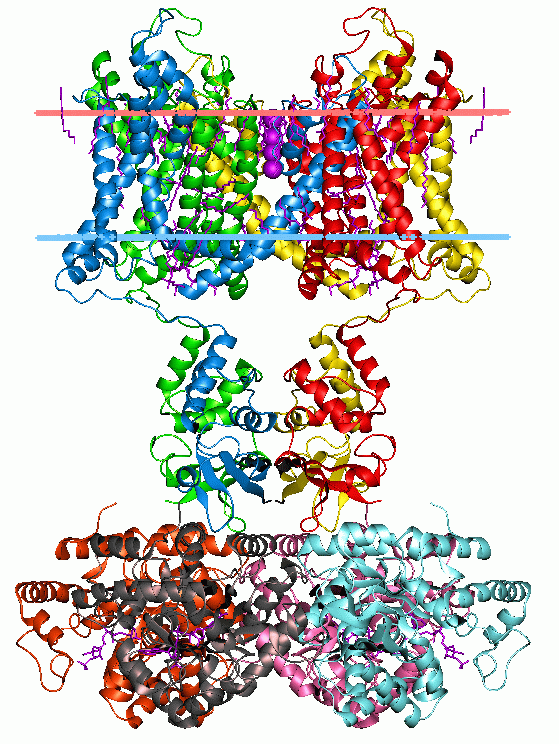
Cys-loop Receptor
The Cys-loop ligand-gated ion channel superfamily is composed of nicotinic acetylcholine, GABAA, GABAA-ρ, glycine, 5-HT3, and zinc-activated (ZAC) receptors. These receptors are composed of five protein subunits which form a pentameric arrangement around a central pore. There are usually 2 alpha subunits and 3 other beta, gamma, or delta subunits (some consist of 5 alpha subunits). The name of the family refers to a characteristic loop formed by 13 highly conserved amino acids between two cysteine (Cys) residues, which form a disulfide bond near the N-terminal extracellular domain. Cys-loop receptors are known only in eukaryotes, but are part of a larger family of pentameric ligand-gated ion channels. Only the Cys-loop clade includes the pair of bridging cysteine residues. The larger superfamily includes bacterial (e.g. GLIC) as well as non-Cys-loop eukaryotic receptors, and is referred to as "pentameric ligand-gated ion channels", or "Pro-loop receptors". All subunits consi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ligand-gated Ion Channel
Ligand-gated ion channels (LICs, LGIC), also commonly referred to as ionotropic receptors, are a group of transmembrane ion-channel proteins which open to allow ions such as sodium, Na+, potassium, K+, calcium, Ca2+, and/or chloride, Cl− to pass through the membrane in response to the binding of a chemical messenger (i.e. a ligand (biochemistry), ligand), such as a neurotransmitter. When a presynaptic neuron is excited, it releases a neurotransmitter from vesicles into the synaptic cleft. The neurotransmitter then binds to receptors located on the postsynaptic neuron. If these receptors are ligand-gated ion channels, a resulting conformational change opens the ion channels, which leads to a flow of ions across the cell membrane. This, in turn, results in either a depolarization, for an excitatory receptor response, or a hyperpolarization (biology), hyperpolarization, for an inhibitory response. These receptor proteins are typically composed of at least two different domains ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aromatic Amino Acids
An aromatic amino acid is an amino acid that includes an aromatic ring. Among the 20 standard amino acids, histidine, phenylalanine, tryptophan, tyrosine, are classified as aromatic. Properties and function Optical properties Aromatic amino acids, excepting histidine, absorb ultraviolet light above and beyond 250 nm and will fluoresce under these conditions. This characteristic is used in quantitative analysis, notably in determining the concentrations of these amino acids in solution. Most proteins absorb at 280 nm due to the presence of tyrosine and tryptophan. Of the aromatic amino acids, tryptophan has the highest extinction coefficient; its absorption maximum occurs at 280 nm. The absorption maximum of tyrosine occurs at 274 nm. Role in protein structure and function Aromatic amino acids stabilize folded structures of many proteins. Aromatic residues are found predominantly sequestered within the cores of globular proteins, although often comprise key portions of p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecular Neuroscience
Molecular neuroscience is a branch of neuroscience that observes concepts in molecular biology applied to the nervous systems of animals. The scope of this subject covers topics such as molecular neuroanatomy, mechanisms of molecular signaling in the nervous system, the effects of genetics and epigenetics on neuronal development, and the molecular basis for neuroplasticity and neurodegenerative diseases. As with molecular biology, molecular neuroscience is a relatively new field that is considerably dynamic. Locating neurotransmitters In molecular biology, communication between neurons typically occurs by chemical transmission across gaps between the cells called synapses. The transmitted chemicals, known as neurotransmitters, regulate a significant fraction of vital body functions. It is possible to anatomically locate neurotransmitters by labeling techniques. It is possible to chemically identify certain neurotransmitters such as catecholamines by fixing neural tissue se ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionotropic Receptors
Ligand-gated ion channels (LICs, LGIC), also commonly referred to as ionotropic receptors, are a group of transmembrane ion-channel proteins which open to allow ions such as Na+, K+, Ca2+, and/or Cl− to pass through the membrane in response to the binding of a chemical messenger (i.e. a ligand), such as a neurotransmitter. When a presynaptic neuron is excited, it releases a neurotransmitter from vesicles into the synaptic cleft. The neurotransmitter then binds to receptors located on the postsynaptic neuron. If these receptors are ligand-gated ion channels, a resulting conformational change opens the ion channels, which leads to a flow of ions across the cell membrane. This, in turn, results in either a depolarization, for an excitatory receptor response, or a hyperpolarization, for an inhibitory response. These receptor proteins are typically composed of at least two different domains: a transmembrane domain which includes the ion pore, and an extracellular domain w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ion Channels
Ion channels are pore-forming membrane proteins that allow ions to pass through the channel pore. Their functions include establishing a resting membrane potential, shaping action potentials and other electrical signals by gating the flow of ions across the cell membrane, controlling the flow of ions across secretory and epithelial cells, and regulating cell volume. Ion channels are present in the membranes of all cells. Ion channels are one of the two classes of ionophoric proteins, the other being ion transporters. The study of ion channels often involves biophysics, electrophysiology, and pharmacology, while using techniques including voltage clamp, patch clamp, immunohistochemistry, X-ray crystallography, fluoroscopy, and RT-PCR. Their classification as molecules is referred to as channelomics. Basic features There are two distinctive features of ion channels that differentiate them from other types of ion transporter proteins: # The rate of ion transport thr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrophysiology
Electrophysiology (from [see the Electron#Etymology, etymology of "electron"]; ; and ) is the branch of physiology that studies the electrical properties of biological cell (biology), cells and tissues. It involves measurements of voltage changes or electric current or manipulations on a wide variety of scales from single ion channel proteins to whole organs like the heart. In neuroscience, it includes measurements of the electrical activity of neurons, and, in particular, action potential activity. Recordings of large-scale electric signals from the nervous system, such as electroencephalography, may also be referred to as electrophysiological recordings. They are useful for electrodiagnostic medicine, electrodiagnosis and monitoring (medicine), monitoring. Definition and scope Classical electrophysiological techniques Principle and mechanisms Electrophysiology is the branch of physiology that pertains broadly to the flow of ions (ion current) in biological tissues and, in p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nicotinic Agonists
A nicotinic agonist is a drug that mimics the action of acetylcholine (ACh) at nicotinic acetylcholine receptors (nAChRs). The nAChR is named for its affinity for nicotine. Examples include nicotine (by definition), acetylcholine (the endogenous agonist of nAChRs), choline, epibatidine, lobeline, varenicline and cytisine. History Nicotine has been known for centuries for its intoxicating effect. It was first isolated in 1828 from the tobacco plant by German chemists Posselt and Reimann. The discovery of positive effects from nicotine on animal memory was discovered by in vivo researches in the mid 1980s. Those researches led to a new era in studies of nicotinic acetylcholine receptor (nAChR) and their stimulation but until then the focus had mainly been on nicotine addiction. The development of nAChR agonists began in the early 1990s after the discovery of nicotine's positive effects. Some research showed a possible therapy option in preclinical researches. ABT-418 was one o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ion Channel
Ion channels are pore-forming membrane proteins that allow ions to pass through the channel pore. Their functions include establishing a resting membrane potential, shaping action potentials and other electrical signals by Gating (electrophysiology), gating the flow of ions across the cell membrane, controlling the flow of ions across secretion, secretory and epithelial cells, and regulating cell (biology), cell volume. Ion channels are present in the membranes of all cells. Ion channels are one of the two classes of ionophore, ionophoric proteins, the other being ion transporters. The study of ion channels often involves biophysics, electrophysiology, and pharmacology, while using techniques including voltage clamp, patch clamp, immunohistochemistry, X-ray crystallography, fluoroscopy, and RT-PCR. Their classification as molecules is referred to as channelomics. Basic features There are two distinctive features of ion channels that differentiate them from other types of ion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron Microscope
An electron microscope is a microscope that uses a beam of electrons as a source of illumination. It uses electron optics that are analogous to the glass lenses of an optical light microscope to control the electron beam, for instance focusing it to produce magnified images or electron diffraction patterns. As the wavelength of an electron can be up to 100,000 times smaller than that of visible light, electron microscopes have a much higher Angular resolution, resolution of about 0.1 nm, which compares to about 200 nm for optical microscope, light microscopes. ''Electron microscope'' may refer to: * Transmission electron microscopy, Transmission electron microscope (TEM) where swift electrons go through a thin sample * Scanning transmission electron microscopy, Scanning transmission electron microscope (STEM) which is similar to TEM with a scanned electron probe * Scanning electron microscope (SEM) which is similar to STEM, but with thick samples * Electron microprobe sim ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Binding Domain
In molecular biology, binding domain is a protein domain which binds to a specific atom or molecule, such as calcium or DNA. A protein domain is a part of a protein sequence and a tertiary structure that can change or evolve, function, and live by itself independent of the rest of the protein chain. Upon binding, proteins may undergo a conformational change. Binding domains are essential for the function of many proteins. They are essential because they help splice, assemble, and translate proteins.Yong, J., T. J. Golembe, D. J. Battle, L. Pellizzoni, and G. Dreyfuss. "SnRNAs Contain Specific SMN-binding Domains That Are Essential for SnRNP Assembly". ''Molecular and Cellular Biology''. U.S. National Library of Medicine, April 2004. Retrieved April 2017. Examples of binding domains include the Zinc finger, which binds to DNA, and EF hand, which binds to calcium. See also *DNA-binding domain *Receptor (biochemistry) In biochemistry and pharmacology, receptors are chemical ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allosteric Regulation
In the fields of biochemistry and pharmacology an allosteric regulator (or allosteric modulator) is a substance that binds to a site on an enzyme or receptor distinct from the active site, resulting in a conformational change that alters the protein's activity, either enhancing or inhibiting its function. In contrast, substances that bind directly to an enzyme's active site or the binding site of the endogenous ligand of a receptor are called orthosteric regulators or modulators. The site to which the effector binds is termed the ''allosteric site'' or ''regulatory site''. Allosteric sites allow effectors to bind to the protein, often resulting in a conformational change and/or a change in protein dynamics. Effectors that enhance the protein's activity are referred to as ''allosteric activators'', whereas those that decrease the protein's activity are called ''allosteric inhibitors''. Allosteric regulations are a natural example of control loops, such as feedback from do ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |