|
Curie (unit)
The curie (symbol Ci) is a non- SI unit of radioactivity originally defined in 1910. According to a notice in ''Nature'' at the time, it was to be named in honour of Pierre Curie, but was considered at least by some to be in honour of Marie Skłodowska-Curie as well, and is in later literature considered to be named for both. It was originally defined as "the quantity or mass of radium emanation in equilibrium with one gram of radium (element)", but is currently defined as 1 Ci = decays per second after more accurate measurements of the activity of Ra (which has a specific activity of ). In 1975 the General Conference on Weights and Measures gave the becquerel (Bq), defined as one nuclear decay per second, official status as the SI unit of activity. Therefore: : 1 Ci = = 37 GBq and : 1 Bq ≅ ≅ 27 pCi While its continued use is discouraged by the National Institute of Standards and Technology (NIST) and other bodies, the curie is still widely used throughout gover ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Specific Activity
Specific activity (symbol ''a'') is the activity per unit mass of a radionuclide and is a physical property of that radionuclide. It is usually given in units of becquerel per kilogram (Bq/kg), but another commonly used unit of specific activity is the curie per gram (Ci/g). In the context of radioactivity, activity or total activity (symbol ''A'') is a physical quantity defined as the number of radioactive transformations per second that occur in a particular radionuclide. The unit of activity is the ''becquerel'' (symbol Bq), which is defined equivalent to reciprocal seconds (symbol s−1). The older, non-SI unit of activity is the ''curie'' (Ci), which is radioactive decays per second. Another unit of activity is the ''rutherford'', which is defined as radioactive decays per second. The specific activity should not be confused with level of exposure to ionizing radiation and thus the exposure or absorbed dose, which is the quantity important in assessing the effects of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
General Conference On Weights And Measures
The General Conference on Weights and Measures (abbreviated CGPM from the ) is the supreme authority of the International Bureau of Weights and Measures (BIPM), the intergovernmental organization established in 1875 under the terms of the Metre Convention through which member states act together on matters related to measurement science and measurement standards. The CGPM is made up of delegates of the governments of the member states and observers from the Associates of the CGPM. It elects the International Committee for Weights and Measures (abbreviated CIPM from the ) as the supervisory board of the BIPM to direct and supervise it. Initially the work of the BIPM concerned the kilogram and the metre, but in 1921 the scope of the Metre Convention was extended to accommodate all Physical quantity, physical measurements and hence all aspects of the metric system. In 1960 the 11th CGPM approved the title International System of Units, usually known as "SI". The General Conferenc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radionuclide
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess numbers of either neutrons or protons, giving it excess nuclear energy, and making it unstable. This excess energy can be used in one of three ways: emitted from the nucleus as gamma radiation; transferred to one of its electrons to release it as a conversion electron; or used to create and emit a new particle (alpha particle or beta particle) from the nucleus. During those processes, the radionuclide is said to undergo radioactive decay. These emissions are considered ionizing radiation because they are energetic enough to liberate an electron from another atom. The radioactive decay can produce a stable nuclide or will sometimes produce a new unstable radionuclide which may undergo further decay. Radioactive decay is a random process at the level of single atoms: it is impossible to predict when one particular atom will decay. However, for a collection of atoms of a single nu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon-14
Carbon-14, C-14, C or radiocarbon, is a radioactive isotope of carbon with an atomic nucleus containing 6 protons and 8 neutrons. Its presence in organic matter is the basis of the radiocarbon dating method pioneered by Willard Libby and colleagues (1949) to date archaeological, geological and hydrogeological samples. Carbon-14 was discovered on February 27, 1940, by Martin Kamen and Sam Ruben at the University of California Radiation Laboratory in Berkeley, California. Its existence had been suggested by Franz Kurie in 1934. There are three naturally occurring isotopes of carbon on Earth: carbon-12 (C), which makes up 99% of all carbon on Earth; carbon-13 (C), which makes up 1%; and carbon-14 (C), which occurs in trace amounts, making up about 1-1.5 atoms per 10 atoms of carbon in the atmosphere. C and C are both stable; C is unstable, with half-life years. Carbon-14 has a specific activity of 62.4 mCi/mmol (2.31 GBq/mmol), or 164.9 GBq/g. Carbon-14 decay ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Composition Of The Human Body
Body composition may be analyzed in various ways. This can be done in terms of the chemical elements present, or by molecular structure e.g., water, protein, fats (or lipids), hydroxyapatite (in bones), carbohydrates (such as glycogen and glucose) and DNA. In terms of tissue type, the body may be analyzed into water, fat, connective tissue, muscle, bone, etc. In terms of cell type, the body contains hundreds of different types of cells, but notably, the largest ''number'' of cells contained in a human body (though not the largest mass of cell) are not human cells, but bacteria residing in the normal human gastrointestinal tract. Elements About 99% of the mass of the human body is made up of six elements: oxygen, carbon, hydrogen, nitrogen, calcium, and phosphorus. Only about 0.85% is composed of another five elements: potassium, sulfur, sodium, chlorine, & magnesium. All 11 are necessary for life. The remaining elements are trace elements, of which more than a dozen are t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium-40
Potassium-40 (K) is a long lived and the main naturally occurring radioactive isotope of potassium. Its half-life is 1.25 billion years. It makes up about 0.012% (120 parts-per notation, ppm) of natural potassium. Potassium-40 undergoes four different types of radioactive decay, including all three main types of beta decay: * Electron emission (β) to calcium-40, Ca with a decay energy of 1.31 electronvolt, MeV at 89.6% probability * Positron emission (β) to argon-40, Ar at 0.001% probability * Electron capture (EC) to Ar followed by a gamma decay emitting a photonAlso called a gamma ray, because it is produced by a transition in the nucleus with an energy of 1.46 MeV at 10.3% probability * Direct electron capture (EC) to the ground state of Ar at 0.1%. Both forms of the electron capture decay release further photons,Also called x-ray, as they are emitted from transitions of electrons when electrons from the outer shells fall into the inner shells to replace the electr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polonium
Polonium is a chemical element; it has symbol Po and atomic number 84. A rare and highly radioactive metal (although sometimes classified as a metalloid) with no stable isotopes, polonium is a chalcogen and chemically similar to selenium and tellurium, though its metallic character resembles that of its horizontal neighbors in the periodic table: thallium, lead, and bismuth. Due to the short half-life of all its isotopes, its natural occurrence is limited to tiny traces of the fleeting polonium-210 (with a half-life of 138 days) in uranium ores, as it is the penultimate daughter of natural uranium-238. Though two longer-lived isotopes exist (polonium-209 with a half-life of 124 years and polonium-208 with a half-life of 2.898 years), they are much more difficult to produce. Today, polonium is usually produced in milligram quantities by the neutron irradiation of bismuth. Due to its intense radioactivity, which results in the radiolysis of chemical bonds and radioactive self-he ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Median Lethal Dose
In toxicology, the median lethal dose, LD50 (abbreviation for " lethal dose, 50%"), LC50 (lethal concentration, 50%) or LCt50 is a toxic unit that measures the lethal dose of a given substance. The value of LD50 for a substance is the dose required to kill half the members of a tested population after a specified test duration. LD50 figures are frequently used as a general indicator of a substance's acute toxicity. A lower LD50 is indicative of higher toxicity. The term LD50 is generally attributed to John William Trevan. The test was created by J. W. Trevan in 1927. The term semilethal dose is occasionally used in the same sense, in particular with translations of foreign language text, but can also refer to a sublethal dose. LD50 is usually determined by tests on animals such as laboratory mice. In 2011, the U.S. Food and Drug Administration approved alternative methods to LD50 for testing the cosmetic drug botox without animal tests. Conventions The LD50 is usually expr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cobalt-60
Cobalt-60 (Co) is a synthetic radioactive isotope of cobalt with a half-life of 5.2714 years. It is produced artificially in nuclear reactors. Deliberate industrial production depends on neutron activation of bulk samples of the monoisotopic and mononuclidic cobalt isotope . (PDF also located aCanadian Nuclear FAQ Measurable quantities are also produced as a by-product of typical nuclear power plant operation and may be detected externally when leaks occur. In the latter case (in the absence of added cobalt) the incidentally produced is largely the result of multiple stages of neutron activation of iron isotopes in the reactor's steel structures via the creation of its precursor. The simplest case of the latter would result from the activation of . undergoes beta decay to an excited state of the stable isotope nickel-60 (), which then emits two gamma rays with energies of and . The overall equation of the nuclear reaction (activation and decay) is: + n → → + e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Caesium-137
Caesium-137 (), cesium-137 (US), or radiocaesium, is a radioactive isotope of caesium that is formed as one of the more common fission products by the nuclear fission of uranium-235 and other fissionable isotopes in nuclear reactors and nuclear weapons. Trace quantities also originate from spontaneous fission of uranium-238. It is among the most problematic of the short-to-medium-lifetime fission products. Caesium-137 has a relatively low boiling point of and easily becomes volatile when released suddenly at high temperature, as in the case of the Chernobyl nuclear accident and with atomic explosions, and can travel very long distances in the air. After being deposited onto the soil as radioactive fallout, it moves and spreads easily in the environment because of the high water solubility of caesium's most common chemical compounds, which are salts. Caesium-137 was discovered by Glenn T. Seaborg and Margaret Melhase. Decay Caesium-137 has a half-life of about 30.04 y ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
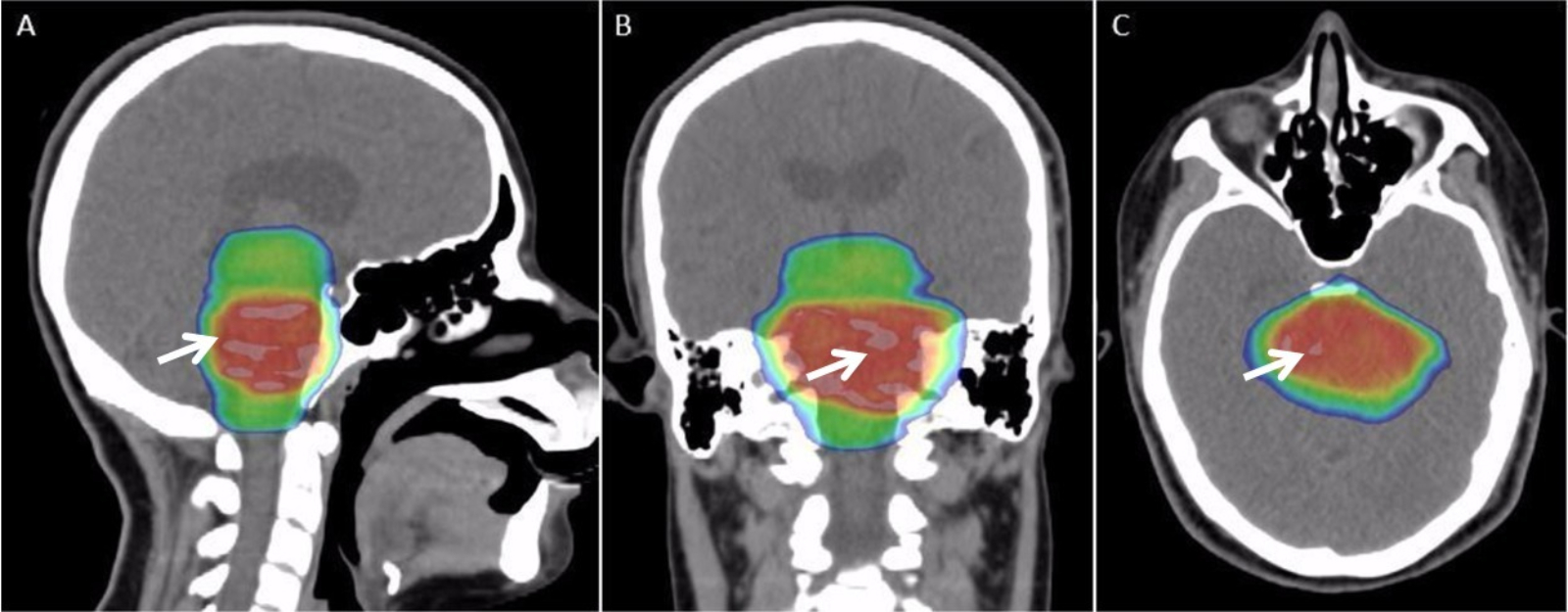
Radiotherapy
Radiation therapy or radiotherapy (RT, RTx, or XRT) is a treatment using ionizing radiation, generally provided as part of cancer therapy to either kill or control the growth of malignant cells. It is normally delivered by a linear particle accelerator. Radiation therapy may be curative in a number of types of cancer if they are localized to one area of the body, and have not spread to other parts. It may also be used as part of adjuvant therapy, to prevent tumor recurrence after surgery to remove a primary malignant tumor (for example, early stages of breast cancer). Radiation therapy is synergistic with chemotherapy, and has been used before, during, and after chemotherapy in susceptible cancers. The subspecialty of oncology concerned with radiotherapy is called radiation oncology. A physician who practices in this subspecialty is a radiation oncologist. Radiation therapy is commonly applied to the cancerous tumor because of its ability to control cell growth. Ionizin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Milliwatt
The watt (symbol: W) is the unit of power or radiant flux in the International System of Units (SI), equal to 1 joule per second or 1 kg⋅m2⋅s−3. It is used to quantify the rate of energy transfer. The watt is named in honor of James Watt (1736–1819), an 18th-century Scottish inventor, mechanical engineer, and chemist who improved the Newcomen engine with his own steam engine in 1776, which became fundamental for the Industrial Revolution. Overview When an object's velocity is held constant at one meter per second against a constant opposing force of one newton, the rate at which work is done is one watt. \mathrm. In terms of electromagnetism, one watt is the rate at which electrical work is performed when a current of one ampere (A) flows across an electrical potential difference of one volt (V), meaning the watt is equivalent to the volt-ampere (the latter unit, however, is used for a different quantity from the real power of an electrical circuit). \ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |