 |
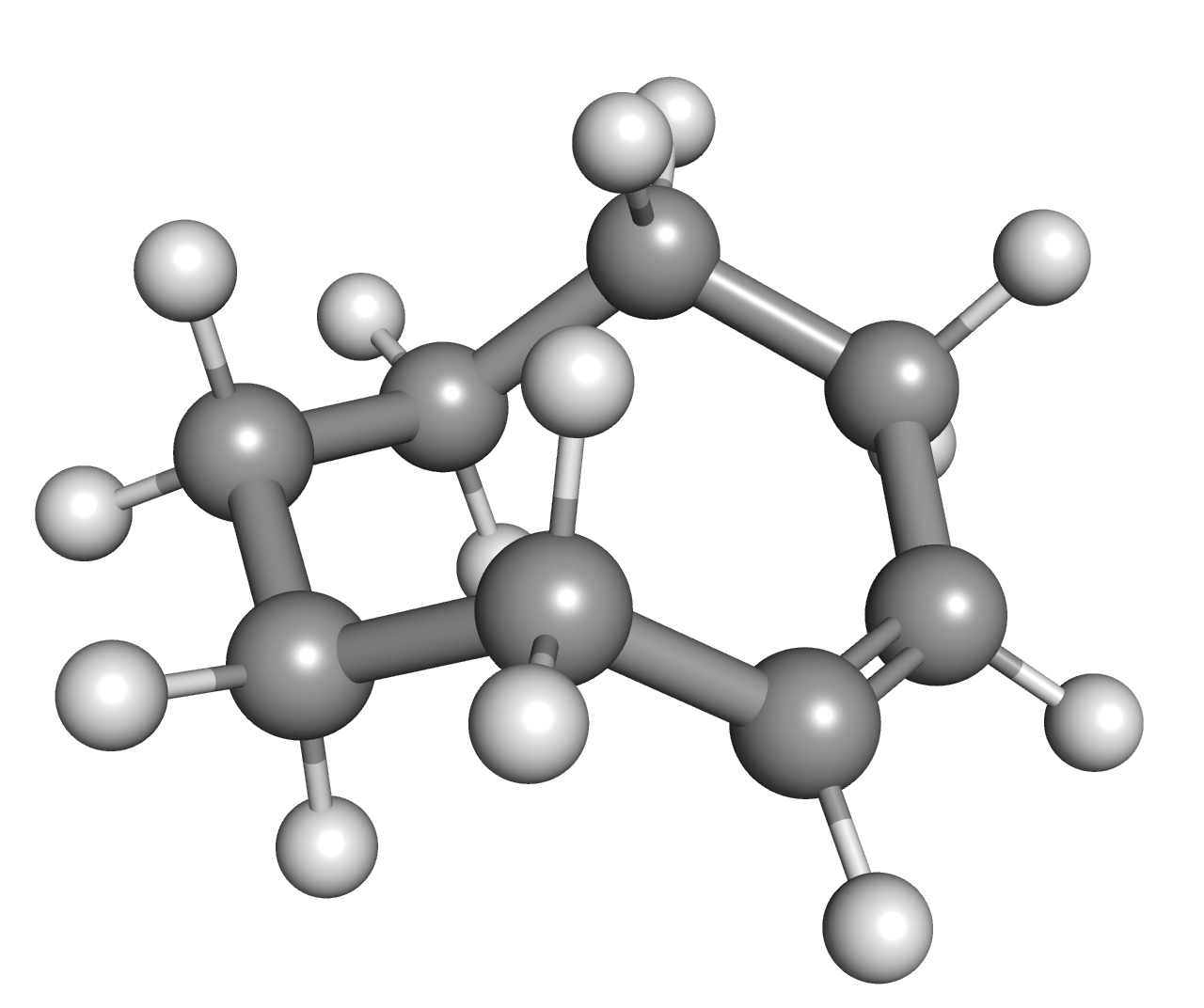
Cis-Cyclooctene
''cis''-Cyclooctene is a cycloalkene with the formula (CH2)6(CH)2. It is a colorless liquid that is used industrially to produce a polymer. It is also a ligand in organometallic chemistry. Cyclooctene is the smallest cycloalkene that can be isolated as both the ''cis''- and ''trans''-isomer. ''cis''-Cyclooctene is shaped like the 8-carbon equivalent chair conformation of cyclohexane. Uses and reactions Cyclooctene undergoes ring-opening metathesis polymerization to give polyoctenamers, which are marketed under the name Vestenamer. ''cis''-Cyclooctene (COE) is a substrate known for quite selectively forming the epoxide, as compared to other cycloalkenes, e.g. cyclohexene. Low amounts of radical by-products are found only. This behaviour is attributed to the difficulty of functionalizing allylic CH centers, which almost orthogonal allylic C-H bonds. Therefore, if radicals are around, they tend to form epoxide via an addition-elimination mechanism. It is used as an easily di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
Trans-cyclooctene Rp 3D Crown Conformation
''trans''-Cyclooctene is a cyclic compound, cyclic hydrocarbon with the formula [–(CH2)6CH=CH–], where the two C–C single bonds adjacent to the double bond are on opposite sides of the latter's plane. It is a colorless liquid with a disagreeable odor. Cyclooctene is notable as the smallest cycloalkene that is readily isolated as its ''trans''-isomer. The ''cis''-isomer is much more stable; the ring-strain energies being 16.7 and 7.4 kcal/mol, respectively. A planar arrangement of the ring carbons would be too strained, and therefore the stable conformational isomer, conformations of the ''trans'' form have a bent (non-planar) ring. Computations indicate that the most stable "crown" conformation has the carbon atoms alternately above and below the plane of the ring. A "half-chair" conformation, with about 6 kcal/mol higher energy, has carbons 2,3,5,6, and 8 on the same side of the plane of carbons 1,4, and 7. All conformations of ''trans''-cyclooctene are chirality, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
|
Sigma-Aldrich
Sigma-Aldrich (formally MilliporeSigma) is an American chemical, life science, and biotechnology company owned by the multinational chemical conglomerate Merck Group. Sigma-Aldrich was created in 1975 by the merger of Sigma Chemical Company and Aldrich Chemical Company. It grew through various acquisitions until it had over 9,600 employees and was listed on the Fortune 1000. The company has two United States headquarters, in St. Louis and Burlington, MA and has operations in approximately 40 countries. In 2015, the multinational chemical conglomerate Merck Group acquired Sigma-Aldrich for $17 billion. The company is currently a part of Merck's life science business and in combination with Merck's earlier acquired Millipore Corporation, Millipore, operates as MilliporeSigma. It is headquartered in Burlington, Massachusetts, United States. History Sigma Chemical Company of St. Louis and Aldrich Chemical Company of Milwaukee were both American specialty chemical companies when they ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
 |
Cycloalkene
In organic chemistry, a cycloalkene or cycloolefin is a type of alkene hydrocarbon which contains a closed Ring (chemistry), ring of carbon atoms and either one or more double bonds, but has no Aromaticity, aromatic character. Some cycloalkenes, such as cyclobutene and cyclopentene, can be used as monomers to produce polymer chains. Due to geometrical considerations, smaller cycloalkenes are almost always the Cis–trans isomerism, ''cis'' isomers, and the term ''cis'' tends to be omitted from the names. Cycloalkenes require considerable p-orbital overlap in the form of a bridge between the carbon-carbon double bond; however, this is not feasible in smaller molecules due to the increase of Ring strain, strain that could break the molecule apart. In greater carbon number cycloalkenes, the addition of substituents decreases strain. trans-Cycloalkenes with 7 or fewer carbons in the ring will not occur under normal conditions because of the large amount of ring strain needed. In large ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
 |
Organometallic Chemistry
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and sometimes broadened to include metalloids like boron, silicon, and selenium, as well. Aside from bonds to organyl fragments or molecules, bonds to 'inorganic' carbon, like carbon monoxide (Metal carbonyl, metal carbonyls), cyanide, or carbide, are generally considered to be organometallic as well. Some related compounds such as transition metal hydrides and metal phosphine complexes are often included in discussions of organometallic compounds, though strictly speaking, they are not necessarily organometallic. The related but distinct term "metalorganics, metalorganic compound" refers to metal-containing compounds lacking direct metal-carbon bonds but which contain organic ligands. Metal β-diketonates, alkoxides, dialkylamides, and metal p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
Cis–trans Isomerism
''Cis''–''trans'' isomerism, also known as geometric isomerism, describes certain arrangements of atoms within molecules. The prefixes "''cis''" and "''trans''" are from Latin: "this side of" and "the other side of", respectively. In the context of chemistry, ''cis'' indicates that the functional groups (substituents) are on the same side of some plane, while ''trans'' conveys that they are on opposing (transverse) sides. ''Cis''–''trans'' isomers are stereoisomers, that is, pairs of molecules which have the same formula but whose functional groups are in different orientations in three-dimensional space. ''Cis'' and ''trans'' isomers occur both in organic molecules and in inorganic coordination complexes. ''Cis'' and ''trans'' descriptors are not used for cases of conformational isomerism where the two geometric forms easily interconvert, such as most open-chain single-bonded structures; instead, the terms "''syn''" and "''anti''" are used. According to IUPAC, "geome ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
|
Isomer
In chemistry, isomers are molecules or polyatomic ions with identical molecular formula – that is, the same number of atoms of each element (chemistry), element – but distinct arrangements of atoms in space. ''Isomerism'' refers to the existence or possibility of isomers. Isomers do not necessarily share similar chemical property, chemical or physical property, physical properties. Two main forms of isomerism are structural isomerism, structural (or constitutional) isomerism, in which ''chemical bond, bonds'' between the atoms differ; and stereoisomerism (or spatial isomerism), in which the bonds are the same but the ''relative positions'' of the atoms differ. Isomeric relationships form a hierarchy. Two chemicals might be the same constitutional isomer, but upon deeper analysis be stereoisomers of each other. Two molecules that are the same stereoisomer as each other might be in different conformational forms or be different Isotopologue, isotopologues. The depth of analy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
|
Cis-cyclooctene 3D Chair Conformation
''cis''-Cyclooctene is a cycloalkene with the formula (CH2)6(CH)2. It is a colorless liquid that is used industrially to produce a polymer. It is also a ligand in organometallic chemistry. Cyclooctene is the smallest cycloalkene that can be isolated as both the ''cis''- and ''trans''-isomer. ''cis''-Cyclooctene is shaped like the 8-carbon equivalent chair conformation of cyclohexane. Uses and reactions Cyclooctene undergoes ring-opening metathesis polymerization to give polyoctenamers, which are marketed under the name Vestenamer. ''cis''-Cyclooctene (COE) is a substrate known for quite selectively forming the epoxide, as compared to other cycloalkenes, e.g. cyclohexene. Low amounts of radical by-products are found only. This behaviour is attributed to the difficulty of functionalizing allylic CH centers, which almost orthogonal allylic C-H bonds. Therefore, if radicals are around, they tend to form epoxide via an addition-elimination mechanism. It is used as an easily di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
 |
Ring-opening Metathesis Polymerization
In polymer chemistry, ring-opening metathesis polymerization (ROMP) is a type of chain-growth polymerization involving olefin metathesis. The reaction is driven by relieving ring strain in cyclic olefins. A variety of heterogeneous and homogeneous catalysts have been developed for different polymers and mechanisms. Heterogeneous catalysts are typical in large-scale commercial processes, while homogeneous catalysts are used in finer laboratory chemical syntheses. Organometallic catalysts used in ROMP usually have transition metal centres, such as tungsten, ruthenium, titanium, etc., with organic ligands. Heterogeneous catalysis : Heterogeneous catalysis consists of catalysts and substrates in different physical states. The catalyst is typically in solid phase. The mechanism of heterogeneous ring-opening metathesis polymerization is still under investigation. Ring-opening metathesis polymerization of cyclic olefins has been commercialized since the 1970s. Examples of polymers pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
Epoxide
In organic chemistry, an epoxide is a cyclic ether, where the ether forms a three-atom ring: two atoms of carbon and one atom of oxygen. This triangular structure has substantial ring strain, making epoxides highly reactive, more so than other ethers. They are produced on a large scale for many applications. In general, low molecular weight epoxides are colourless and nonpolar, and often volatile. Nomenclature A compound containing the epoxide functional group can be called an epoxy, epoxide, oxirane, and ethoxyline. Simple epoxides are often referred to as oxides. Thus, the epoxide of ethylene (C2H4) is ethylene oxide (C2H4O). Many compounds have trivial names; for instance, ethylene oxide is called "oxirane". Some names emphasize the presence of the epoxide functional group, as in the compound ''1,2-epoxyheptane'', which can also be called ''1,2-heptene oxide''. A polymer formed from epoxide precursors is called an ''epoxy''. However, few if any of the epoxy groups i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
|
Cyclohexene
Cyclohexene is a hydrocarbon with the formula . It is a cycloalkene. At room temperature, cyclohexene is a colorless liquid with a sharp odor. Among its uses, it is an chemical intermediate, intermediate in the commercial synthesis of nylon. Production and uses Cyclohexene is produced by the partial hydrogenation of benzene, a process developed by the Asahi Chemical company. The main product of the process is cyclohexane because cyclohexene is more easily hydrogenated than benzene. In the laboratory, it can be prepared by dehydration reaction, dehydration of cyclohexanol. : Reactions and uses Benzene is converted to cyclohexylbenzene by acid-catalyzed alkylation with cyclohexene. Cyclohexylbenzene is a precursor to both phenol and cyclohexanone. Hydration of cyclohexene gives cyclohexanol, which can be dehydrogenation, dehydrogenated to give cyclohexanone, a precursor to caprolactam. The oxidative cleavage of cyclohexene gives adipic acid. Hydrogen peroxide is used as the oxid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
|
Ligand
In coordination chemistry, a ligand is an ion or molecule with a functional group that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's electron pairs, often through Lewis acids and bases, Lewis bases. The nature of metal–ligand bonding can range from covalent bond, covalent to ionic bond, ionic. Furthermore, the metal–ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acids and bases, Lewis acidic "ligands". Metals and metalloids are bound to ligands in almost all circumstances, although gaseous "naked" metal ions can be generated in a high vacuum. Ligands in a complex dictate the reactivity (chemistry), reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection requires critical consideration in many practical are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |