|
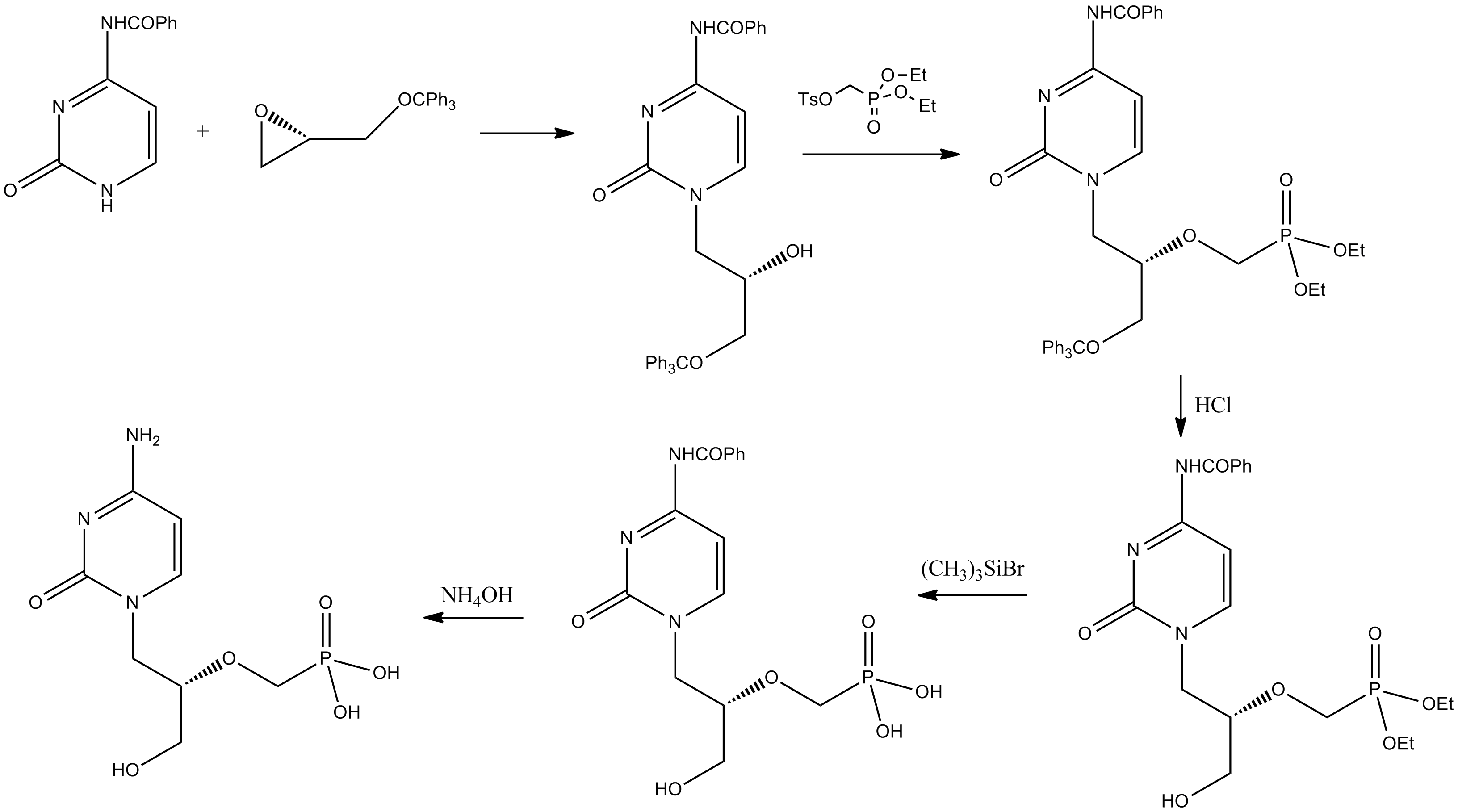
Cidofovir
Cidofovir, brand name Vistide, is a topical or injectable antiviral medication primarily used as a treatment for cytomegalovirus (CMV) retinitis (an infection of the retina of the eye) in people with AIDS. Cidofovir was approved for medical use in 1996. Medical use DNA virus Its only indication that has received regulatory approval worldwide is cytomegalovirus retinitis. Cidofovir has also shown efficacy in the treatment of aciclovir-resistant HSV infections. Cidofovir has also been investigated as a treatment for progressive multifocal leukoencephalopathy with successful case reports of its use. Despite this, the drug failed to demonstrate any efficacy in controlled studies. Cidofovir might have anti-smallpox efficacy and might be used on a limited basis in the event of a bioterror incident involving smallpox cases. Brincidofovir, a cidofovir derivative with much higher activity against smallpox that can be taken orally has been developed. It has inhibitory effects o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Brincidofovir
Brincidofovir, sold under the brand name Tembexa, is an antiviral drug used to treat smallpox. Brincidofovir is a prodrug of cidofovir. Conjugated to a lipid, the compound is designed to release cidofovir intracellularly, allowing for higher intracellular and lower plasma concentrations of cidofovir, effectively increasing its activity against dsDNA viruses, as well as oral bioavailability. The most common side effects include diarrhea, nausea, vomiting, and abdominal pain. It carries an FDA-mandated Boxed warning, black box warning of an increased risk of death with extended use. Brincidofovir was approved for medical use in the United States in June 2021. Medical uses Brincidofovir is Indication (medicine), indicated for the treatment of human smallpox disease caused by the variola virus. Mechanism of action Brincidofovir is a prodrug that is composed of cidofovir Conjugation (biochemistry), conjugated with a lipid molecule. The lipid aspect of the molecule takes on the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Progressive Multifocal Leukoencephalopathy
Progressive multifocal leukoencephalopathy (PML) is a rare and often fatal viral disease characterized by progressive damage (''-pathy'') or inflammation of the white matter (''leuko-'') of the brain (''-encephalo-'') at multiple locations (''multifocal''). It is caused by the JC virus, which is normally present and kept under control by the immune system. The JC virus is harmless except in cases of weakened immune systems. In general, PML has a mortality rate of 30–50% in the first few months, and those who survive can be left with varying degrees of neurological disabilities. PML occurs almost exclusively in patients with severe immune deficiency, most commonly among patients with acquired immune deficiency syndrome (AIDS), but people on chronic immunosuppressive medications including chemotherapy are also at increased risk of PML, such as patients with transplants, Hodgkin's lymphoma, multiple sclerosis, psoriasis, rheumatoid arthritis, and other autoimmune diseases. Signs ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Smallpox
Smallpox was an infectious disease caused by Variola virus (often called Smallpox virus), which belongs to the genus '' Orthopoxvirus''. The last naturally occurring case was diagnosed in October 1977, and the World Health Organization (WHO) certified the global eradication of the disease in 1980, making smallpox the only human disease to have been eradicated to date. The initial symptoms of the disease included fever and vomiting. This was followed by formation of ulcers in the mouth and a skin rash. Over a number of days, the skin rash turned into the characteristic fluid-filled blisters with a dent in the center. The bumps then scabbed over and fell off, leaving scars. The disease was transmitted from one person to another primarily through prolonged face-to-face contact with an infected person or rarely via contaminated objects. Prevention was achieved mainly through the smallpox vaccine. Once the disease had developed, certain antiviral medications could poten ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gilead Sciences
Gilead Sciences, Inc. () is an American biopharmaceutical company headquartered in Foster City, California, that focuses on researching and developing antiviral drugs used in the treatment of HIV/AIDS, hepatitis B, hepatitis C, influenza, and COVID-19, including ledipasvir/sofosbuvir and sofosbuvir. Gilead is a member of the Nasdaq-100 and the S&P 100. Gilead was founded in 1987 under the name Oligogen by Michael L. Riordan. The original name was a reference to oligonucleotides, small strands of DNA used to target genetic sequences. Gilead held its initial public offering in 1992, and successfully developed drugs like Tamiflu and Vistide that decade. In the 2000s, Gilead received approval for drugs including Viread and Hepsera, among others. It began evolving from a biotechnology company into a pharmaceutical company, acquiring several subsidiaries, though it still relied heavily on contracting to manufacture its drugs. The company continued its growth in the 2010s. Howev ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Probenecid
Probenecid, also sold under the brand name Probalan, is a medication that increases uric acid excretion in the urine. It is primarily used in treating gout and hyperuricemia. Probenecid was developed as an alternative to caronamide to competitively inhibit renal excretion of some drugs, thereby increasing their plasma concentration and prolonging their effects. Medical uses Probenecid is primarily used to treat gout and hyperuricemia. Probenecid is sometimes used to increase the concentration of some antibiotics and to protect the kidneys when given with cidofovir. Specifically, a small amount of evidence supports the use of intravenous cefazolin once rather than three times a day when it is combined with probenecid. It has also found use as a masking agent, potentially helping athletes using performance-enhancing substances to avoid detection by drug tests. Adverse effects Mild symptoms such as nausea, loss of appetite, dizziness, vomiting, headache, sore gums, or frequent uri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Intravenous
Intravenous therapy (abbreviated as IV therapy) is a medical technique that administers fluids, medications and nutrients directly into a person's vein. The intravenous route of administration is commonly used for rehydration or to provide nutrients for those who cannot, or will not—due to reduced mental states or otherwise—consume food or water per os, by mouth. It may also be used to administer pharmaceutical drug, medications or other medical therapy such as blood transfusion, blood products or electrolytes to correct electrolyte imbalances. Attempts at providing intravenous therapy have been recorded as early as the 1400s, but the practice did not become widespread until the 1900s after the development of techniques for safe, effective use. The intravenous route is the fastest way to deliver medications and fluid replacement throughout the body as they are introduced directly into the circulatory system and thus quickly distributed. For this reason, the intravenous route ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
FGF2
Fibroblast growth factor 2 (FGF-2), also known as basic fibroblast growth factor (bFGF) and FGF-β, is a growth factor and signaling protein encoded by the ''FGF2'' gene. It binds to and exerts effects via specific fibroblast growth factor receptor ''(FGFR) proteins'', themselves a family of closely related molecules. Fibroblast growth factor protein was first purified in 1975; soon thereafter three variants were isolated: 'basic FGF' (FGF2); Heparin-binding growth factor-2; and Endothelial cell growth factor-2. Gene sequencing revealed that this group is the same FGF2 protein and is a member of a family of FGF proteins. Function Like other FGF family members, basic fibroblast growth factor possesses broad mitogenic and cell survival activities, and is involved in a variety of biological processes, including embryonic development, cell growth, morphogenesis, tissue repair, tumor growth and invasion. In normal tissue, bFGF is present in basement membranes and in the subend ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutropenia
Neutropenia is an abnormally low concentration of neutrophils (a type of white blood cell) in the blood. Neutrophils make up the majority of circulating white blood cells and serve as the primary defense against infections by destroying bacteria, bacterial fragments and immunoglobulin-bound viruses in the blood. People with neutropenia are more susceptible to bacterial infections and, without prompt medical attention, the condition may become life-threatening (neutropenic sepsis). Neutropenia can be divided into congenital and acquired, with severe congenital neutropenia (SCN) and cyclic neutropenia (CyN) being autosomal dominant and mostly caused by heterozygous mutations in the ELANE gene ( neutrophil elastase). Neutropenia can be acute (temporary) or chronic (long lasting). The term is sometimes used interchangeably with "leukopenia" ("deficit in the number of white blood cells"). Decreased production of neutrophils is associated with deficiencies of vitamin B12 and fol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
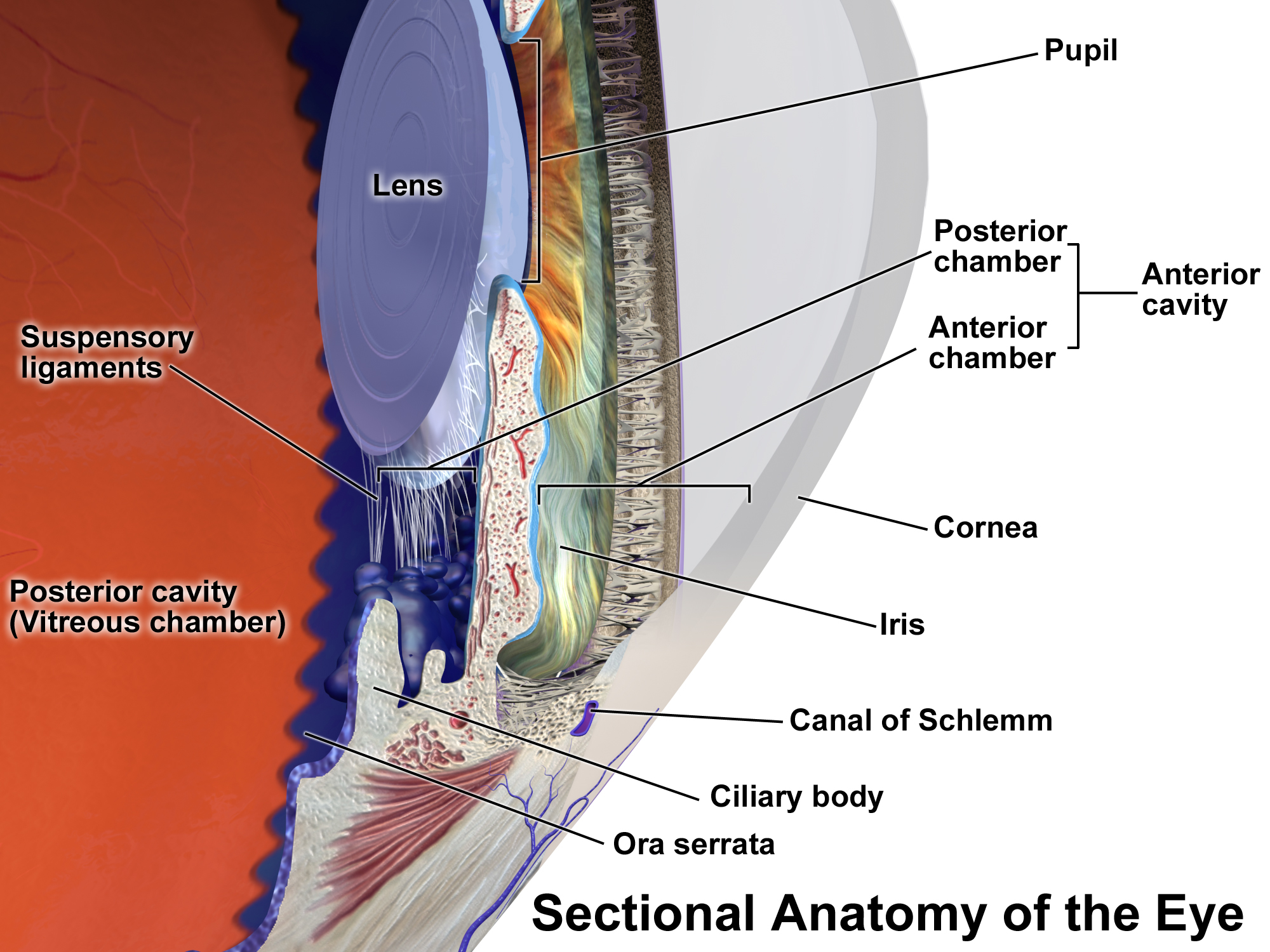
Iritis
Uveitis () is inflammation of the uvea, the pigmented layer of the eye between the inner retina and the outer fibrous layer composed of the sclera and cornea. The uvea consists of the middle layer of pigmented vascular structures of the eye and includes the iris, ciliary body, and choroid. Uveitis is described anatomically, by the part of the eye affected, as anterior, intermediate or posterior, or panuveitic if all parts are involved. Anterior uveitis (iridocyclitis) is the most common, with the incidence of uveitis overall affecting approximately 1:4500, most commonly those between the ages of 20–60. Symptoms include eye pain, eye redness, floaters and blurred vision, and ophthalmic examination may show dilated ciliary blood vessels and the presence of cells in the anterior chamber. Uveitis may arise spontaneously, have a genetic component, or be associated with an autoimmune disease or infection. While the eye is a relatively protected environment, its immune mechanisms ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uveitis
Uveitis () is inflammation of the uvea, the pigmented layer of the eye between the inner retina and the outer fibrous layer composed of the sclera and cornea. The uvea consists of the middle layer of pigmented vascular structures of the eye and includes the iris, ciliary body, and choroid. Uveitis is described anatomically, by the part of the eye affected, as anterior, intermediate or posterior, or panuveitic if all parts are involved. Anterior uveitis ( iridocyclitis) is the most common, with the incidence of uveitis overall affecting approximately 1:4500, most commonly those between the ages of 20–60. Symptoms include eye pain, eye redness, floaters and blurred vision, and ophthalmic examination may show dilated ciliary blood vessels and the presence of cells in the anterior chamber. Uveitis may arise spontaneously, have a genetic component, or be associated with an autoimmune disease or infection. While the eye is a relatively protected environment, its immune mecha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
European Medicines Agency
The European Medicines Agency (EMA) is an agency of the European Union (EU) in charge of the evaluation and supervision of pharmaceutical products. Prior to 2004, it was known as the European Agency for the Evaluation of Medicinal Products or European Medicines Evaluation Agency (EMEA).Set up by EC Regulation No. 2309/93 as the European Agency for the Evaluation of Medicinal Products, and renamed by EC Regulation No. 726/2004 to the European Medicines Agency, it had the acronym EMEA until December 2009. The European Medicines Agency does not call itself EMA either – it has no official acronym but may reconsider if EMA becomes commonly accepted (secommunication on new visual identity an). The EMA was set up in 1995, with funding from the European Union and the pharmaceutical industry, as well as indirect subsidy from member states, its stated intention to harmonise (but not replace) the work of existing national medicine regulatory bodies. The hope was that this plan would ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |