|
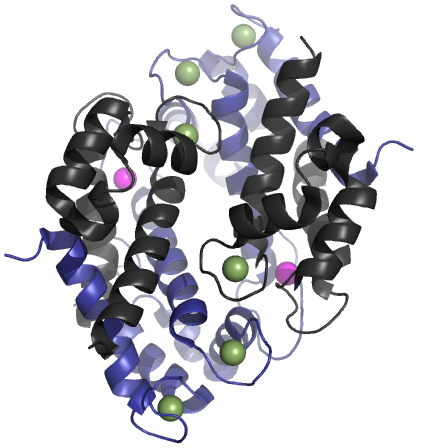
Calprotectin
Calprotectin is a complex of the mammalian proteins S100A8 and S100A9. Other names for calprotectin include MRP8-MRP14, calgranulin A and B, cystic fibrosis antigen, L1, 60BB antigen, and 27E10 antigen. The proteins exist as homodimers but preferentially exist as S100A8/A9 heterodimers or heterotetramers (calprotectin) with antimicrobial, proinflammatory and prothrombotic properties. In the presence of calcium, calprotectin is capable of sequestering the transition metals iron, manganese and zinc via chelation. This metal sequestration affords the complex antimicrobial properties. Calprotectin is the only known antimicrobial manganese sequestration protein complex. Calprotectin comprises as much as 60% of the soluble protein content of the cytosol of a neutrophil, and it is secreted by an unknown mechanism during inflammation. Faecal calprotectin has been used to detect intestinal inflammation (colitis or enteritis) and can serve as a biomarker for inflammatory bowel diseases ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
S100A8
S100 calcium-binding protein A8 (S100A8) is a protein that in humans is encoded by the ''S100A8'' gene. It is also known as calgranulin A. The proteins S100A8 and S100A9 form a heterodimer called calprotectin Calprotectin is a complex of the mammalian proteins S100A8 and S100A9. Other names for calprotectin include MRP8-MRP14, calgranulin A and B, cystic fibrosis antigen, L1, 60BB antigen, and 27E10 antigen. The proteins exist as homodimers but pre .... The protein encoded by this gene is a member of the S100 family of proteins containing 2 EF-hand calcium-binding motifs. S100 proteins are localized in the cytoplasm and/or nucleus of a wide range of cells, and involved in the regulation of a number of cellular processes such as cell cycle progression and differentiation. S100 genes include at least 13 members which are located as a cluster on chromosome 1q21. This protein may function in the inhibition of casein kinase and as a cytokine. Altered expression of this protein ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
S100A9
S100 calcium-binding protein A9 (S100A9) also known as migration inhibitory factor-related protein 14 (MRP14) or calgranulin B is a protein that in humans is encoded by the ''S100A9'' gene. The proteins S100A8 and S100A9 form a heterodimer called calprotectin. Function S100A9 is a member of the S100 family of proteins containing 2 EF hand calcium-binding motifs. S100 proteins are localized in the cytoplasm and/or nucleus of a wide range of cells, and involved in the regulation of a number of cellular processes such as cell cycle progression and differentiation. S100 genes include at least 13 members which are located as a cluster on chromosome 1q21. This protein may function in the inhibition of casein kinase. MRP14 complexes with MRP-8 ( S100A8), another member of the S100 family of calcium-modulated proteins; together, MRP8 and MRP14 regulate myeloid cell function by binding to Toll-like receptor 4 (TLR4) and the receptor for advanced glycation end products. Intracell ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Multiprotein Complex
A protein complex or multiprotein complex is a group of two or more associated polypeptide chains. Protein complexes are distinct from multienzyme complexes, in which multiple catalytic domains are found in a single polypeptide chain. Protein complexes are a form of quaternary structure. Proteins in a protein complex are linked by non-covalent protein–protein interactions. These complexes are a cornerstone of many (if not most) biological processes. The cell is seen to be composed of modular supramolecular complexes, each of which performs an independent, discrete biological function. Through proximity, the speed and selectivity of binding interactions between enzymatic complex and substrates can be vastly improved, leading to higher cellular efficiency. Many of the techniques used to enter cells and isolate proteins are inherently disruptive to such large complexes, complicating the task of determining the components of a complex. Examples of protein complexes include the p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biomarker
In biomedical contexts, a biomarker, or biological marker, is a measurable indicator of some biological state or condition. Biomarkers are often measured and evaluated using blood, urine, or soft tissues to examine normal biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention. as cited in Biomarkers are used in many scientific fields. Medicine Biomarkers used in the medical field, are a part of a relatively new clinical toolset categorized by their clinical applications. The three main classes are molecular biomarkers, cellular biomarkers or imaging biomarkers. All three types of biomarkers have a clinical role in narrowing or guiding treatment decisions and follow a sub-categorization of being either predictive, prognostic, or diagnostic. Predictive Predictive molecular, cellular, or imaging biomarkers that pass validation can serve as a method of predicting clinical outcomes. Predictive biomarkers are used to help optimize ide ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Non-covalent Interactions
In chemistry, a non-covalent interaction differs from a covalent bond in that it does not involve the sharing of electrons, but rather involves more dispersed variations of electromagnetic interactions between molecules or within a molecule. The chemical energy released in the formation of non-covalent interactions is typically on the order of 1–5 kcal/ mol (1000–5000 calories per 6.02 molecules). Non-covalent interactions can be classified into different categories, such as electrostatic, π-effects, van der Waals forces, and hydrophobic effects. Non-covalent interactions are critical in maintaining the three-dimensional structure of large molecules, such as proteins and nucleic acids. In addition, they are also involved in many biological processes in which large molecules bind specifically but transiently to one another (see the properties section of the DNA page). These interactions also heavily influence drug design, crystallinity and design of materials, particularly ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gel Filtration
Size-exclusion chromatography (SEC), also known as molecular sieve chromatography, is a chromatographic method in which molecules in solution are separated by their size, and in some cases molecular weight. It is usually applied to large molecules or macromolecular complexes such as proteins and industrial polymers. Typically, when an aqueous solution is used to transport the sample through the column, the technique is known as gel-filtration chromatography, versus the name gel permeation chromatography, which is used when an organic solvent is used as a mobile phase. The chromatography column is packed with fine, porous beads which are commonly composed of dextran, agarose, or polyacrylamide polymers. The pore sizes of these beads are used to estimate the dimensions of macromolecules. SEC is a widely used polymer characterization method because of its ability to provide good molar mass distribution (Mw) results for polymers. Applications The main application of gel-filtration chro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Primary Structure
Protein primary structure is the linear sequence of amino acids in a peptide or protein. By convention, the primary structure of a protein is reported starting from the amino-terminal (N) end to the carboxyl-terminal (C) end. Protein biosynthesis is most commonly performed by ribosomes in cells. Peptides can also be synthesized in the laboratory. Protein primary structures can be directly sequenced, or inferred from DNA sequences. Formation Biological Amino acids are polymerised via peptide bonds to form a long backbone, with the different amino acid side chains protruding along it. In biological systems, proteins are produced during translation by a cell's ribosomes. Some organisms can also make short peptides by non-ribosomal peptide synthesis, which often use amino acids other than the standard 20, and may be cyclised, modified and cross-linked. Chemical Peptides can be synthesised chemically via a range of laboratory methods. Chemical methods typically synthesis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Dimer
In biochemistry, a protein dimer is a macromolecular complex formed by two protein monomers, or single proteins, which are usually non-covalently bound. Many macromolecules, such as proteins or nucleic acids, form dimers. The word ''dimer'' has roots meaning "two parts", ''di-'' + '' -mer''. A protein dimer is a type of protein quaternary structure. A protein homodimer is formed by two identical proteins. A protein heterodimer is formed by two different proteins. Most protein dimers in biochemistry are not connected by covalent bonds. An example of a non-covalent heterodimer is the enzyme reverse transcriptase, which is composed of two different amino acid chains. An exception is dimers that are linked by disulfide bridges such as the homodimeric protein NEMO. Some proteins contain specialized domains to ensure dimerization (dimerization domains) and specificity. The G protein-coupled cannabinoid receptors have the ability to form both homo- and heterodimers with sev ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Atomic Mass Unit
The dalton or unified atomic mass unit (symbols: Da or u) is a non-SI unit of mass widely used in physics and chemistry. It is defined as of the mass of an unbound neutral atom of carbon-12 in its nuclear and electronic ground state and at rest. The atomic mass constant, denoted ''m''u, is defined identically, giving . This unit is commonly used in physics and chemistry to express the mass of atomic-scale objects, such as atoms, molecules, and elementary particles, both for discrete instances and multiple types of ensemble averages. For example, an atom of helium-4 has a mass of . This is an intrinsic property of the isotope and all helium-4 atoms have the same mass. Acetylsalicylic acid (aspirin), , has an average mass of approximately . However, there are no acetylsalicylic acid molecules with this mass. The two most common masses of individual acetylsalicylic acid molecules are , having the most common isotopes, and , in which one carbon is carbon-13. The molecular ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sequence Homology
Sequence homology is the biological homology between DNA, RNA, or protein sequences, defined in terms of shared ancestry in the evolutionary history of life. Two segments of DNA can have shared ancestry because of three phenomena: either a speciation event (orthologs), or a duplication event (paralogs), or else a horizontal (or lateral) gene transfer event (xenologs). Homology among DNA, RNA, or proteins is typically inferred from their nucleotide or amino acid sequence similarity. Significant similarity is strong evidence that two sequences are related by evolutionary changes from a common ancestral sequence. Alignments of multiple sequences are used to indicate which regions of each sequence are homologous. Identity, similarity, and conservation The term "percent homology" is often used to mean "sequence similarity”, that is the percentage of identical residues (''percent identity''), or the percentage of residues conserved with similar physicochemical properties ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sepsis
Sepsis, formerly known as septicemia (septicaemia in British English) or blood poisoning, is a life-threatening condition that arises when the body's response to infection causes injury to its own tissues and organs. This initial stage is followed by suppression of the immune system. Common signs and symptoms include fever, increased heart rate, increased breathing rate, and confusion. There may also be symptoms related to a specific infection, such as a cough with pneumonia, or painful urination with a kidney infection. The very young, old, and people with a weakened immune system may have no symptoms of a specific infection, and the body temperature may be low or normal instead of having a fever. Severe sepsis causes poor organ function or blood flow. The presence of low blood pressure, high blood lactate, or low urine output may suggest poor blood flow. Septic shock is low blood pressure due to sepsis that does not improve after fluid replacement. Sepsis is caus ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |