 |
Calmodulin
Calmodulin (CaM) (an abbreviation for calcium-modulated protein) is a multifunctional intermediate calcium-binding messenger protein expressed in all Eukaryote, eukaryotic cells. It is an intracellular target of the Second messenger system, secondary messenger Calcium in biology, Ca2+, and the binding of Ca2+ is required for the activation of calmodulin. Once bound to Ca2+, calmodulin acts as part of a calcium Signal transduction, signal transduction pathway by modifying its interactions with various target proteins such as kinases or phosphatases. Structure Calmodulin is a small, highly conserved protein that is 148 amino acids long (16.7 kDa). The protein has two approximately symmetrical globular domains (the N- and C- domains) each containing a pair of EF hand Sequence motif, motifs separated by a flexible linker region for a total of four Ca2+ binding sites, two in each globular domain. In the Ca2+-free state, the helices that form the four EF-hands are collapsed in a co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
 |
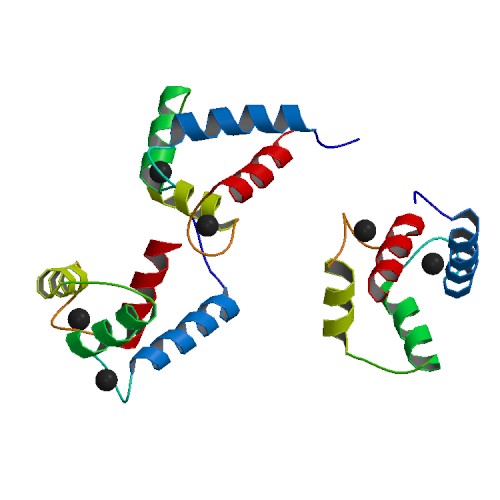
Calmodulin Binding Sites
Calmodulin (CaM) (an abbreviation for calcium-modulated protein) is a multifunctional intermediate calcium-binding messenger protein expressed in all eukaryotic cells. It is an intracellular target of the secondary messenger Ca2+, and the binding of Ca2+ is required for the activation of calmodulin. Once bound to Ca2+, calmodulin acts as part of a calcium signal transduction pathway by modifying its interactions with various target proteins such as kinases or phosphatases. Structure Calmodulin is a small, highly conserved protein that is 148 amino acids long (16.7 kDa). The protein has two approximately symmetrical globular domains (the N- and C- domains) each containing a pair of EF hand motifs separated by a flexible linker region for a total of four Ca2+ binding sites, two in each globular domain. In the Ca2+-free state, the helices that form the four EF-hands are collapsed in a compact orientation, and the central linker is disordered; in the Ca2+-saturated state, the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
 |
Calmodulin C-terminal
Calmodulin (CaM) (an abbreviation for calcium-modulated protein) is a multifunctional intermediate calcium-binding messenger protein expressed in all Eukaryote, eukaryotic cells. It is an intracellular target of the Second messenger system, secondary messenger Calcium in biology, Ca2+, and the binding of Ca2+ is required for the activation of calmodulin. Once bound to Ca2+, calmodulin acts as part of a calcium Signal transduction, signal transduction pathway by modifying its interactions with various target proteins such as kinases or phosphatases. Structure Calmodulin is a small, highly conserved protein that is 148 amino acids long (16.7 kDa). The protein has two approximately symmetrical globular domains (the N- and C- domains) each containing a pair of EF hand Sequence motif, motifs separated by a flexible linker region for a total of four Ca2+ binding sites, two in each globular domain. In the Ca2+-free state, the helices that form the four EF-hands are collapsed in a co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
 |
CaMKII
/calmodulin-dependent protein kinase II (CaM kinase II or CaMKII) is a serine/threonine-specific protein kinase that is regulated by the /calmodulin complex. CaMKII is involved in many signaling cascades and is thought to be an important mediator of learning and memory. CaMKII is also necessary for homeostasis and reuptake in cardiomyocytes, chloride transport in epithelia, positive T-cell selection, and CD8 T-cell activation. Misregulation of CaMKII is linked to Alzheimer's disease, Angelman syndrome, and heart arrhythmia. Types There are two types of CaM kinases: * Specialized CaM kinases, such as the myosin light chain kinase that phosphorylates myosin, causing smooth muscles to contract. * Multifunctional CaM kinases, also collectively called ''CaM kinase II'', which play a role in neurotransmitter secretion, transcription factor regulation, and glycogen metabolism. Structure, function, and autoregulation CaMKII accounts for 1–2% of all proteins in the brai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
|
EF Hand
The EF hand is a helix–loop–helix structural domain or ''motif'' found in a large family of calcium-binding proteins. The EF-hand motif contains a helix–loop–helix topology, much like the spread thumb and forefinger of the human hand, in which the Ca2+ ions are coordinated by ligands within the loop. The motif takes its name from traditional nomenclature used in describing the protein parvalbumin, which contains three such motifs and is probably involved in muscle relaxation via its calcium-binding activity. The EF-hand consists of two alpha helices linked by a short loop region (usually about 12 amino acids) that usually binds calcium ions. EF-hands also appear in each structural domain of the signaling protein calmodulin and in the muscle protein troponin-C. Calcium ion binding site The calcium ion is coordinated in a pentagonal bipyramidal configuration. The six residues involved in the binding are in positions 1, 3, 5, 7, 9 and 12; these residues are denoted ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
|
 |
Cooperative Binding
Cooperative binding occurs in molecular binding systems containing more than one type, or species, of molecule and in which one of the partners is not mono-valent and can bind more than one molecule of the other species. In general, molecular binding is an interaction between molecules that results in a stable physical association between those molecules. Cooperative binding occurs in a molecular binding system where two or more ''ligand'' molecules can bind to a ''receptor'' molecule. Binding can be considered "cooperative" if the actual binding of the first molecule of the ligand to the receptor changes the binding affinity of the second ligand molecule. The binding of ligand molecules to the different sites on the receptor molecule do not constitute mutually independent events. Cooperativity can be positive or negative, meaning that it becomes more or less likely that successive ligand molecules will bind to the receptor molecule. Cooperative binding is observed in many biopolym ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
 |
Kinase
In biochemistry, a kinase () is an enzyme that catalyzes the transfer of phosphate groups from high-energy, phosphate-donating molecules to specific substrates. This process is known as phosphorylation, where the high-energy ATP molecule donates a phosphate group to the substrate molecule. As a result, kinase produces a phosphorylated substrate and ADP. Conversely, it is referred to as dephosphorylation when the phosphorylated substrate donates a phosphate group and ADP gains a phosphate group (producing a dephosphorylated substrate and the high energy molecule of ATP). These two processes, phosphorylation and dephosphorylation, occur four times during glycolysis. Kinases are part of the larger family of phosphotransferases. Kinases should not be confused with phosphorylases, which catalyze the addition of inorganic phosphate groups to an acceptor, nor with phosphatases, which remove phosphate groups (dephosphorylation). The phosphorylation state of a molecule, whether it ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
|
IQ-motif
The IQ calmodulin-binding motif is an amino acid sequence motif containing the following sequence: * ILVxxx Kxxx Kx ILVWY The term "IQ" refers to the first two amino acids of the motif: isoleucine (commonly) and glutamine (invariably). Function Calmodulin (CaM) is recognized as a major calcium (Ca2+) sensor and orchestrator of regulatory events through its interaction with a diverse group of cellular proteins. Three classes of recognition motifs exist for many of the known CaM binding proteins; the IQ motif as a consensus for Ca2+-independent binding and two related motifs for Ca2+-dependent binding, termed 1-14 and 1-5-10 based on the position of conserved hydrophobic residues. Example The regulatory domain of scallop myosin is a three-chain protein complex that switches on this motor in response to Ca2+ binding. Side-chain interactions link the two light chains in tandem to adjacent segments of the heavy chain bearing the IQ-sequence motif. The Ca2+-binding site is a nov ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
|
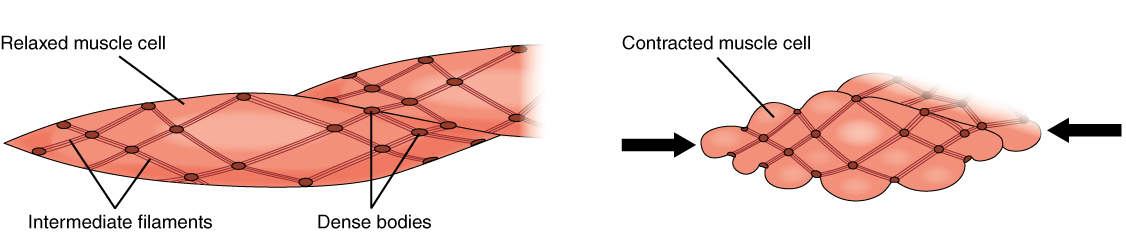 |
Smooth Muscle
Smooth muscle is one of the three major types of vertebrate muscle tissue, the others being skeletal and cardiac muscle. It can also be found in invertebrates and is controlled by the autonomic nervous system. It is non- striated, so-called because it has no sarcomeres and therefore no striations (''bands'' or ''stripes''). It can be divided into two subgroups, ''single-unit'' and ''multi-unit'' smooth muscle. Within single-unit muscle, the whole bundle or sheet of smooth muscle cells contracts as a syncytium. Smooth muscle is found in the walls of hollow organs, including the stomach, intestines, bladder and uterus. In the walls of blood vessels, and lymph vessels, (excluding blood and lymph capillaries) it is known as vascular smooth muscle. There is smooth muscle in the tracts of the respiratory, urinary, and reproductive systems. In the eyes, the ciliary muscles, iris dilator muscle, and iris sphincter muscle are types of smooth muscles. The iris dilator and s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
|
Signal Transduction
Signal transduction is the process by which a chemical or physical signal is transmitted through a cell as a biochemical cascade, series of molecular events. Proteins responsible for detecting stimuli are generally termed receptor (biology), receptors, although in some cases the term sensor is used. The changes elicited by ligand (biochemistry), ligand binding (or signal sensing) in a receptor give rise to a biochemical cascade, which is a chain of biochemical events known as a Cell signaling#Signaling pathways, signaling pathway. When signaling pathways interact with one another they form networks, which allow cellular responses to be coordinated, often by combinatorial signaling events. At the molecular level, such responses include changes in the transcription (biology), transcription or translation (biology), translation of genes, and post-translational modification, post-translational and conformational changes in proteins, as well as changes in their location. These molecula ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
|
|
Calcium In Biology
Calcium ions (Ca2+) contribute to the physiology and biochemistry of organisms' cells. They play an important role in signal transduction pathways, where they act as a second messenger, in neurotransmitter release from neurons, in contraction of all muscle cell types, and in fertilization. Many enzymes require calcium ions as a cofactor, including several of the coagulation factors. Extracellular calcium is also important for maintaining the potential difference across excitable cell membranes, as well as proper bone formation. Plasma calcium levels in mammals are tightly regulated, electronic-book electronic- with bone acting as the major mineral storage site. Calcium ions, Ca2+, are released from bone into the bloodstream under controlled conditions. Calcium is transported through the bloodstream as dissolved ions or bound to proteins such as serum albumin. Parathyroid hormone secreted by the parathyroid gland regulates the resorption of Ca2+ from bone, reabsorptio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
|
|
Sodium Channel
Sodium channels are integral membrane proteins that form ion channels, conducting sodium ions (Na+) through a cell (biology), cell's cell membrane, membrane. They belong to the Cation channel superfamily, superfamily of cation channels. Classification They are classified into 2 types: Function In excitable cells such as neurons, muscle, myocytes, and certain types of glia, sodium channels are responsible for the Action potential#Stimulation and rising phase, rising phase of action potentials. These channels go through three different states called resting, active and inactive states. Even though the resting and inactive states would not allow the ions to flow through the channels the difference exists with respect to their structural conformation. Selectivity Sodium channels are highly selective for the transport of ions across cell membranes. The high selectivity with respect to the sodium ion is achieved in many different ways. All involve encapsulation of the sodium ion in a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |