|
Paraffin Wax
Paraffin wax (or petroleum wax) is a soft colorless solid derived from petroleum, coal, or oil shale that consists of a mixture of hydrocarbon molecules containing between 20 and 40 carbon atoms. It is solid at room temperature and melting point, begins to melt above approximately , and its boiling point is above . Common applications for paraffin wax include lubrication, electrical insulation, and candles; dyed paraffin wax can be made into crayons. Un-dyed, unscented paraffin candles are odorless and bluish-white. Paraffin wax was first created by Carl Reichenbach#Scientific contributions, Carl Reichenbach in Germany in 1830 and marked a major advancement in candlemaking technology, as it burned more cleanly and reliably than tallow candles and was cheaper to produce. In chemistry, ''paraffin'' is used synonymously with ''alkane'', indicating hydrocarbons with the general formula C''n''H2''n''+2. The name is derived from Latin ''parum'' ("very little") + ''affinis'', meaning ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Candle Black
A candle is an ignitable candle wick, wick embedded in wax, or another flammable solid substance such as tallow, that provides light, and in some cases, a Aroma compound, fragrance. A candle can also provide heat or a method of keeping time. Candles have been used for over two millennia around the world, and were a significant form of indoor lighting until the invention of other types of light sources. Although electric light has largely made candle use nonessential for illumination, candles are still commonly used for functional, symbolic and aesthetic purposes and in specific cultural and religious settings. Early candles may be made of beeswax, but these candles were expensive and their use was limited to the elite and the churches. Tallow was a cheaper but a less aesthetically pleasing alternative. A variety of different materials have been developed in the modern era for making candles, including paraffin wax, which together with efficient production techniques, made can ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethyl Ether
Diethyl ether, or simply ether, is an organic compound with the chemical formula , sometimes abbreviated as . It is a colourless, highly Volatility (chemistry), volatile, sweet-smelling ("ethereal odour"), extremely flammable liquid. It belongs to the ether class of organic compounds. It is a common solvent and was formerly used as a general anesthetic. Production Most diethyl ether is produced as a byproduct of the vapor-phase Hydration reaction, hydration of ethylene to make ethanol. This process uses solid-supported phosphoric acid Catalysis, catalysts and can be adjusted to make more ether if the need arises: Vapor-phase Dehydration reaction, dehydration of ethanol over some Aluminium oxide, alumina catalysts can give diethyl ether yields of up to 95%. : Diethyl ether can be prepared both in laboratories and on an industrial scale by the acid ether synthesis. Uses The dominant use of diethyl ether is as a solvent. One particular application is in the production of cell ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phase Change Material
A phase-change material (PCM) is a substance which releases/absorbs sufficient energy at phase transition to provide useful heat or cooling. Generally the transition will be from one of the first two fundamental states of matter - solid and liquid - to the other. The phase transition may also be between non-classical states of matter, such as the conformity of crystals, where the material goes from conforming to one crystalline structure to conforming to another, which may be a higher or lower energy state. The energy released/absorbed by phase transition from solid to liquid, or vice versa, the heat of fusion is generally much higher than the sensible heat. Ice, for example, requires 333.55 J/g to melt, but then water will rise one degree further with the addition of just 4.18 J/g. Water/ice is therefore a very useful phase change material and has been used to store winter cold to cool buildings in summer since at least the time of the Achaemenid Empire. By melting and sol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
James Chadwick
Sir James Chadwick (20 October 1891 – 24 July 1974) was an English nuclear physicist who received the Nobel Prize in Physics in 1935 for his discovery of the neutron. In 1941, he wrote the final draft of the MAUD Report, which inspired the U.S. government to begin serious atomic bomb research efforts. He was the head of the British team that worked on the Manhattan Project during World War II. He was knighted in Britain in 1945 for his achievements in nuclear physics. Chadwick graduated from the Victoria University of Manchester in 1911, where he studied under Ernest Rutherford (known as the "father of nuclear physics"). At Manchester, he continued to study under Rutherford until he was awarded his MSc in 1913. The same year, Chadwick was awarded an 1851 Research Fellowship from the Royal Commission for the Exhibition of 1851. He elected to study beta radiation under Hans Geiger in Berlin. Using Geiger's recently developed Geiger counter, Chadwick was able to demons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutron Moderator
In nuclear engineering, a neutron moderator is a medium that reduces the speed of fast neutrons, ideally without capturing any, leaving them as thermal neutrons with only minimal (thermal) kinetic energy. These thermal neutrons are immensely more susceptible than fast neutrons to propagate a nuclear chain reaction of uranium-235 or other fissile isotope by colliding with their atomic nucleus. Water (sometimes called "light water" in this context) is the most commonly used moderator (roughly 75% of the world's reactors). Solid graphite (20% of reactors) and heavy water (5% of reactors) are the main alternatives. Beryllium has also been used in some experimental types, and hydrocarbons have been suggested as another possibility. Moderation Neutrons are normally bound into an atomic nucleus and do not exist free for long in nature. The unbound neutron has a half-life of 10 minutes and 11 seconds. The release of neutrons from the nucleus requires exceeding the binding ener ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polytetrafluoroethylene
Polytetrafluoroethylene (PTFE) is a synthetic fluoropolymer of tetrafluoroethylene, and has numerous applications because it is chemically inert. The commonly known brand name of PTFE-based composition is Teflon by Chemours, a corporate spin-off, spin-off from DuPont (1802–2017), DuPont, which originally invented the compound in 1938. Polytetrafluoroethylene is a fluorocarbon solid, as it is a high-molecular-weight polymer consisting wholly of carbon and fluorine. PTFE is hydrophobic: neither water nor water-containing substances Wetting, wet PTFE, as fluorocarbons exhibit only small London dispersion forces due to the low polarizability, electric polarizability of fluorine. PTFE has one of the lowest Friction#Coefficient of friction, coefficients of friction of any solid. Polytetrafluoroethylene is used as a non-stick coating for Cookware and bakeware, pans and other Cookware and bakeware, cookware. It is Chemically inert, non-reactive, partly because of the strength of car ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Plastics
Plastics are a wide range of synthetic or semisynthetic materials composed primarily of polymers. Their defining characteristic, plasticity, allows them to be molded, extruded, or pressed into a diverse range of solid forms. This adaptability, combined with a wide range of other properties such as low weight, durability, flexibility, chemical resistance, low toxicity, and low-cost production, has led to their widespread use around the world. While most plastics are produced from natural gas and petroleum, a growing minority are produced from renewable resources like polylactic acid. Between 1950 and 2017, 9.2 billion metric tons of plastic are estimated to have been made, with more than half of this amount being produced since 2004. In 2023 alone, preliminary figures indicate that over 400 million metric tons of plastic were produced worldwide. If global trends in plastic demand continue, it is projected that annual global plastic production will exceed 1.3 billion tons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
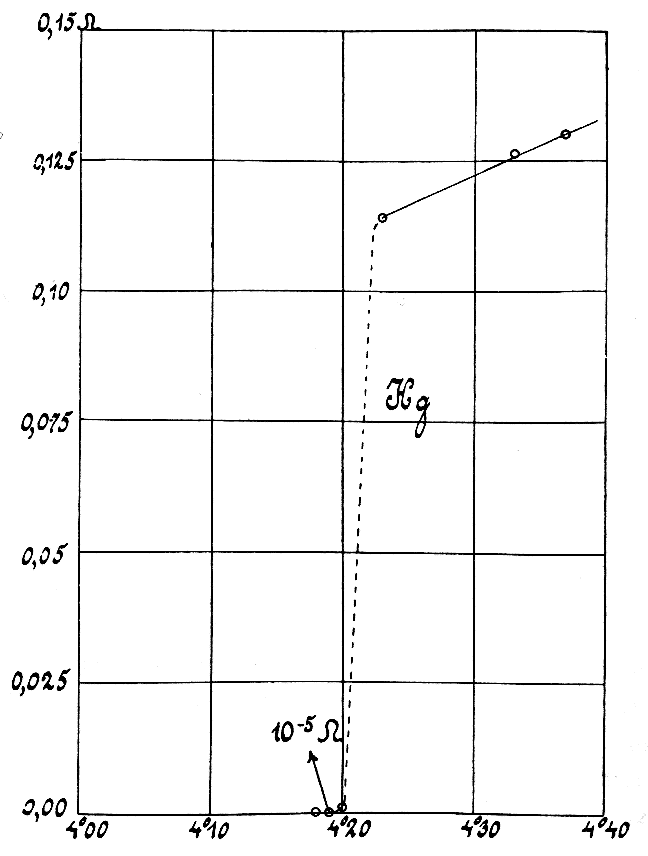
Resistivity
Electrical resistivity (also called volume resistivity or specific electrical resistance) is a fundamental specific property of a material that measures its electrical resistance or how strongly it resists electric current. A low resistivity indicates a material that readily allows electric current. Resistivity is commonly represented by the Greek letter (rho). The SI unit of electrical resistivity is the ohm-metre (Ω⋅m). For example, if a solid cube of material has sheet contacts on two opposite faces, and the resistance between these contacts is , then the resistivity of the material is . Electrical conductivity (or specific conductance) is the reciprocal of electrical resistivity. It represents a material's ability to conduct electric current. It is commonly signified by the Greek letter (sigma), but (kappa) (especially in electrical engineering) and (gamma) are sometimes used. The SI unit of electrical conductivity is siemens per metre (S/m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrical Insulator
An electrical insulator is a material in which electric current does not flow freely. The atoms of the insulator have tightly bound electrons which cannot readily move. Other materials—semiconductors and electrical conductor, conductors—conduct electric current more easily. The property that distinguishes an insulator is its resistivity; insulators have higher resistivity than semiconductors or conductors. The most common examples are Nonmetal (chemistry), non-metals. A perfect insulator does not exist because even the materials used as insulators contain small numbers of mobile charges (charge carriers) which can carry current. In addition, all insulators become electrically conductive when a sufficiently large voltage is applied that the electric field tears electrons away from the atoms. This is known as electrical breakdown, and the voltage at which it occurs is called the breakdown voltage of an insulator. Some materials such as glass, Electrical insulation paper, paper ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hentriacontane
Hentriacontane, also called ''untriacontane'', is a solid, long-chain alkane hydrocarbon with the structural formula CH3(CH2)29CH3. It is the main component of paraffin wax. It is found in a variety of plants, including peas (''Pisum sativum''), '' Acacia senegal'', ''Gymnema sylvestre'' and others, and also comprises about 8–9% of beeswax Bee hive wax complex Beeswax (also known as cera alba) is a natural wax produced by honey bees of the genus ''Apis''. The wax is formed into scales by eight wax-producing glands in the abdominal segments of worker bees, which discard it in o .... It has 10,660,307,791 constitutional isomers. References External links Hentriacontaneat Dr. Duke's Phytochemical and Ethnobotanical Databases {{Alkanes Alkanes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Combustion
Combustion, or burning, is a high-temperature exothermic redox chemical reaction between a fuel (the reductant) and an oxidant, usually atmospheric oxygen, that produces oxidized, often gaseous products, in a mixture termed as smoke. Combustion does not always result in fire, because a flame is only visible when substances undergoing combustion vaporize, but when it does, a flame is a characteristic indicator of the reaction. While activation energy must be supplied to initiate combustion (e.g., using a lit match to light a fire), the heat from a flame may provide enough energy to make the reaction self-sustaining. The study of combustion is known as combustion science. Combustion is often a complicated sequence of elementary reaction, elementary Radical (chemistry), radical reactions. Solid fuels, such as wood and coal, first undergo endothermic pyrolysis to produce gaseous fuels whose combustion then supplies the heat required to produce more of them. Combustion is often hot e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |