|
Nickel Aluminide
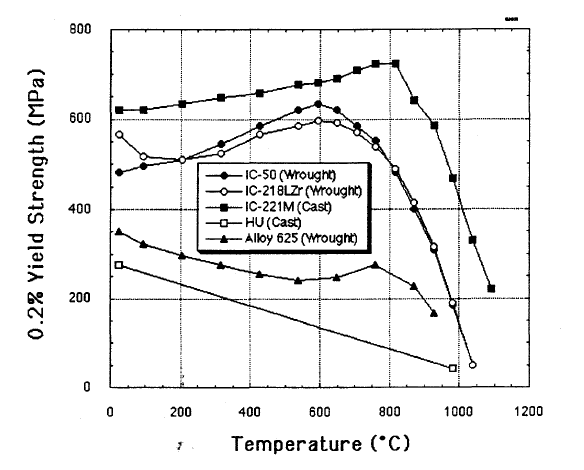
Nickel aluminide refers to either of two widely used intermetallic compounds, Ni3Al or NiAl, but the term is sometimes used to refer to any nickelŌĆōaluminium alloy. These alloys are widely used because of their high strength even at high temperature, low density, corrosion resistance, and ease of production. Ni3Al is of specific interest as a precipitate in nickel-based superalloys, where it is called the ╬│' (gamma prime) phase. It gives these alloys high strength and creep (deformation), creep resistance up to 0.7ŌĆō0.8 of its melting temperature. Meanwhile, NiAl displays excellent properties such as lower density and higher melting temperature than those of Ni3Al, and good thermal conductivity and oxidation resistance. These properties make it attractive for special high-temperature applications like coatings on blades in gas turbines and jet engines. However, both these alloys have the disadvantage of being quite brittle at room temperature, with Ni3Al remaining brittle at high ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Intermetallic
An intermetallic (also called intermetallic compound, intermetallic alloy, ordered intermetallic alloy, long-range-ordered alloy) is a type of metallic alloy that forms an ordered solid-state compound between two or more metallic elements. Intermetallics are generally hard and brittle, with good high-temperature mechanical properties. They can be classified as stoichiometric or nonstoichiometic. The term "intermetallic compounds" applied to solid phases has long been in use. However, Hume-Rothery argued that it misleads, suggesting a fixed stoichiometry and a clear decomposition into species. Definitions Research definition In 1967 defined intermetallic compounds as ''solid phases containing two or more metallic elements, with optionally one or more non-metallic elements, whose crystal structure differs from that of the other constituents''. This definition includes: * Electron (or Hume-Rothery) compounds * Size packing phases. e.g. Laves phases, FrankŌĆōKasper phases and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromium
Chromium is a chemical element; it has Symbol (chemistry), symbol Cr and atomic number 24. It is the first element in Group 6 element, group 6. It is a steely-grey, Luster (mineralogy), lustrous, hard, and brittle transition metal. Chromium is valued for its high corrosion resistance and hardness. A major development in steel production was the discovery that steel could be made highly resistant to corrosion and discoloration by adding metallic chromium to form stainless steel. Stainless steel and chrome plating (electroplating with chromium) together comprise 85% of the commercial use. Chromium is also greatly valued as a metal that is able to be highly polishing, polished while resisting tarnishing. Polished chromium reflects almost 70% of the visible spectrum, and almost 90% of infrared, infrared light. The name of the element is derived from the Ancient Greek, Greek word ŽćŽüß┐Č╬╝╬▒, ''chr┼Źma'', meaning color, because many chromium compounds are intensely colored. Indust ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Intermetallics
An intermetallic (also called intermetallic compound, intermetallic alloy, ordered intermetallic alloy, long-range-ordered alloy) is a type of metallic alloy that forms an ordered solid-state compound between two or more metallic elements. Intermetallics are generally hard and brittle, with good high-temperature mechanical properties. They can be classified as stoichiometric or nonstoichiometic. The term "intermetallic compounds" applied to solid phases has long been in use. However, Hume-Rothery argued that it misleads, suggesting a fixed stoichiometry and a clear decomposition into species. Definitions Research definition In 1967 defined intermetallic compounds as ''solid phases containing two or more metallic elements, with optionally one or more non-metallic elements, whose crystal structure differs from that of the other constituents''. This definition includes: * Electron (or Hume-Rothery) compounds * Size packing phases. e.g. Laves phases, FrankŌĆōKasper phases and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stainless Steel
Stainless steel, also known as inox, corrosion-resistant steel (CRES), or rustless steel, is an iron-based alloy that contains chromium, making it resistant to rust and corrosion. Stainless steel's resistance to corrosion comes from its chromium content of 11% or more, which forms a Passivation (chemistry), passive film that protects the material and can self-healing material, self-heal when exposed to oxygen. It can be further alloyed with elements like molybdenum, carbon, nickel and nitrogen to enhance specific properties for various applications. The alloy's properties, such as luster and resistance to corrosion, are useful in many applications. Stainless steel can be rolled into Sheet metal, sheets, plates, bars, wire, and tubing. These can be used in cookware, cutlery, surgical instruments, major appliances, vehicles, construction material in large buildings, industrial equipment (e.g., in paper mills, chemical plants, water treatment), and storage tanks and tankers for ch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metallurgical Furnace
A metallurgical furnace, often simply referred to as a furnace when the context is known, is an industrial furnace used to heat, melt, or otherwise process metals. Furnaces have been a central piece of equipment throughout the history of metallurgy; processing metals with heat is even its own engineering specialty known as pyrometallurgy. One important furnace application, especially in iron and steel production, is smelting, where metal ores are Redox, reduced under high heat to separate the metal content from mineral gangue. The heat energy to fuel a furnace may be supplied directly by fuel combustion or by electricity. Different processes and the unique properties of specific metals and ores have led to many different furnace types. Air blast furnaces Many furnace designs for smelting combine ore, fuel, and other reagents like flux in a single chamber. Mechanisms, such as bellows or motorized fans, then drive pressurized blasts of air into the chamber. These blasts make the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corrosion
Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials (usually a metal) by chemical or electrochemical reaction with their environment. Corrosion engineering is the field dedicated to controlling and preventing corrosion. In the most common use of the word, this means electrochemical oxidation of metal in reaction with an oxidant such as oxygen, hydrogen, or hydroxide. Rusting, the formation of red-orange iron oxides, is a well-known example of electrochemical corrosion. This type of corrosion typically produces oxides or salts of the original metal and results in a distinctive coloration. Corrosion can also occur in materials other than metals, such as ceramics or polymers, although in this context, the term "degradation" is more common. Corrosion degrades the useful properties of materials and structures including mechanical strength, appearance, and permeability to liquids and ga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SAE 304 Stainless Steel
SAE 304 stainless steel is the most common stainless steel. It is an alloy of iron, carbon, chromium and nickel. It is an austenitic stainless steel, and is therefore not magnetic. It is less electrically and thermally conductive than carbon steel. It has a higher corrosion resistance than regular steel and is widely used because of the ease in which it is formed into various shapes.Data sheet on SAE 304 stainless steel . The composition was developed by W. H. Hatfield at Firth Brown in 1924 and was marketed under ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
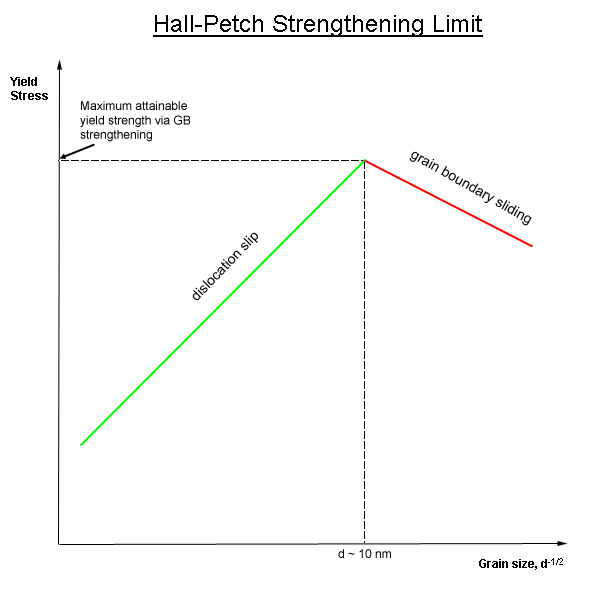
Hall-Petch
In materials science, grain-boundary strengthening (or HallŌĆōPetch strengthening) is a method of strengthening materials by changing their average crystallite (grain) size. It is based on the observation that grain boundaries are insurmountable borders for dislocations and that the number of dislocations within a grain has an effect on how stress builds up in the adjacent grain, which will eventually activate dislocation sources and thus enabling deformation in the neighbouring grain as well. By changing grain size, one can influence the number of dislocations piled up at the grain boundary and yield strength. For example, heat treatment after plastic deformation and changing the rate of solidification are ways to alter grain size.W.D. Callister. Fundamentals of Materials Science and Engineering, 2nd ed. Wiley & Sons. pp. 252. Theory In grain-boundary strengthening, the grain boundaries act as pinning points impeding further dislocation propagation. Since the lattice structure ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Grain Boundary
In materials science, a grain boundary is the interface between two grains, or crystallites, in a polycrystalline material. Grain boundaries are two-dimensional defects in the crystal structure, and tend to decrease the electrical and thermal conductivity of the material. Most grain boundaries are preferred sites for the onset of corrosion and for the precipitation of new phases from the solid. They are also important to many of the mechanisms of creep. On the other hand, grain boundaries disrupt the motion of dislocations through a material, so reducing crystallite size is a common way to improve mechanical strength, as described by the HallŌĆōPetch relationship. High and low angle boundaries It is convenient to categorize grain boundaries according to the extent of misorientation between the two grains. ''Low-angle grain boundaries'' (''LAGB'') or ''subgrain boundaries'' are those with a misorientation less than about 15 degrees. Generally speaking they are composed of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ductility
Ductility refers to the ability of a material to sustain significant plastic Deformation (engineering), deformation before fracture. Plastic deformation is the permanent distortion of a material under applied stress, as opposed to elastic deformation, which is reversible upon removing the stress. Ductility is a critical mechanical performance indicator, particularly in applications that require materials to bend, stretch, or deform in other ways without breaking. The extent of ductility can be quantitatively assessed using the percent elongation at break, given by the equation: \% \mathrm= \left ( \frac \right )\times100 where l_ is the length of the material after fracture and l_0 is the original length before testing. This formula helps in quantifying how much a material can stretch under tensile stress before failure, providing key insights into its ductile behavior. Ductility is an important consideration in engineering and manufacturing. It defines a material's suitabil ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |