|
Germanium Dioxide
Germanium dioxide, also called germanium(IV) oxide, germania, and salt of germanium, is an inorganic compound with the chemical formula Ge O2. It is the main commercial source of germanium. It also forms as a passivation layer on pure germanium in contact with atmospheric oxygen. Structure The two predominant polymorphs of GeO2 are hexagonal and tetragonal. Hexagonal GeO2 has the same structure as α-quartz, with germanium having coordination number 4. Tetragonal GeO2 (the mineral argutite) has the rutile-like structure seen in stishovite. In this motif, germanium has the coordination number 6. An amorphous (glassy) form of GeO2 is similar to fused silica. Germanium dioxide can be prepared in both crystalline and amorphous forms. At ambient pressure the amorphous structure is formed by a network of GeO4 tetrahedra. At elevated pressure up to approximately 9 GPa the germanium average coordination number steadily increases from 4 to around 5 with a corresponding increase ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acid
An acid is a molecule or ion capable of either donating a proton (i.e. Hydron, hydrogen cation, H+), known as a Brønsted–Lowry acid–base theory, Brønsted–Lowry acid, or forming a covalent bond with an electron pair, known as a Lewis acid. The first category of acids are the proton donors, or Brønsted–Lowry acid–base theory, Brønsted–Lowry acids. In the special case of aqueous solutions, proton donors form the hydronium ion H3O+ and are known as Acid–base reaction#Arrhenius theory, Arrhenius acids. Johannes Nicolaus Brønsted, Brønsted and Martin Lowry, Lowry generalized the Arrhenius theory to include non-aqueous solvents. A Brønsted–Lowry or Arrhenius acid usually contains a hydrogen atom bonded to a chemical structure that is still energetically favorable after loss of H+. Aqueous Arrhenius acids have characteristic properties that provide a practical description of an acid. Acids form aqueous solutions with a sour taste, can turn blue litmus red, and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystalline
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macroscopic single crystals are usually identifiable by their geometrical shape, consisting of flat faces with specific, characteristic orientations. The scientific study of crystals and crystal formation is known as crystallography. The process of crystal formation via mechanisms of crystal growth is called crystallization or solidification. The word ''crystal'' derives from the Ancient Greek word (), meaning both "ice" and " rock crystal", from (), "icy cold, frost". Examples of large crystals include snowflakes, diamonds, and table salt. Most inorganic solids are not crystals but polycrystals, i.e. many microscopic crystals fused together into a single solid. Polycrystals include most metals, rocks, ceramics, and ice. A third categor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Optical Microscope
The optical microscope, also referred to as a light microscope, is a type of microscope that commonly uses visible light and a system of lenses to generate magnified images of small objects. Optical microscopes are the oldest design of microscope and were possibly invented in their present compound form in the 17th century. Basic optical microscopes can be very simple, although many complex designs aim to improve resolution and sample contrast. The object is placed on a stage and may be directly viewed through one or two eyepieces on the microscope. In high-power microscopes, both eyepieces typically show the same image, but with a stereo microscope, slightly different images are used to create a 3-D effect. A camera is typically used to capture the image (micrograph). The sample can be lit in a variety of ways. Transparent objects can be lit from below and solid objects can be lit with light coming through ( bright field) or around ( dark field) the objective lens. Polar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wide-angle Lens
In photography and cinematography, a wide-angle lens is a Photographic lens, lens covering a large angle of view. Conversely, its focal length is substantially smaller than that of a normal lens for a given film plane. This type of lens allows more of the Scene (perception), scene to be included in the photograph, which is useful in architectural, interior, and landscape photography where the photographer may not be able to move farther from the scene to photograph it. Another use is where the photographer wishes to emphasize the difference in size or distance between objects in the foreground and the background; nearby objects appear very large and objects at a moderate distance appear small and far away. This exaggeration of relative size can be used to make foreground objects more prominent and striking, while capturing expansive backgrounds. A wide-angle lens is also one that projects a substantially larger image circle than would be typical for a standard design lens of the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
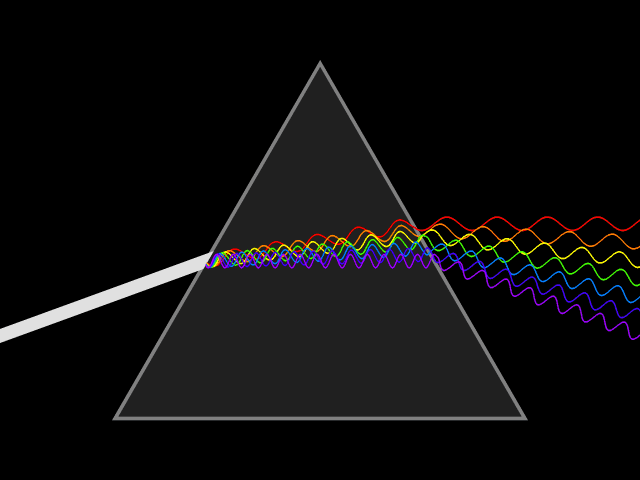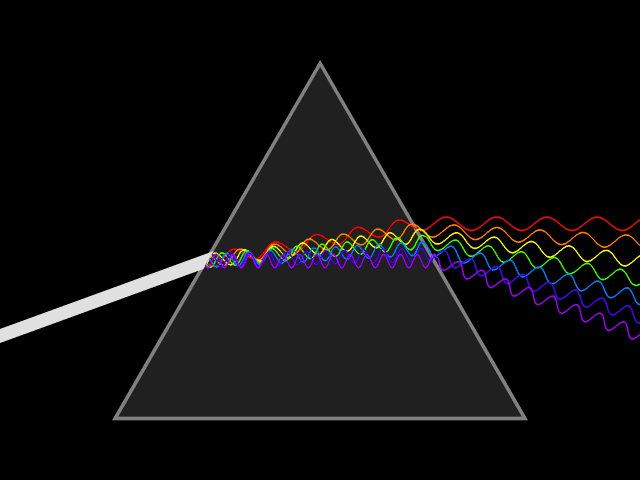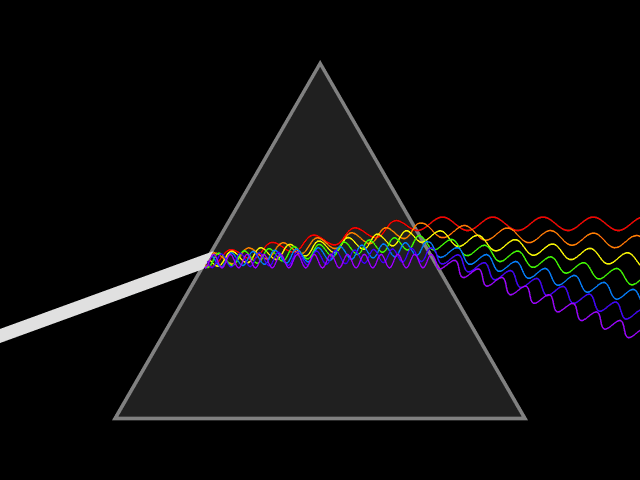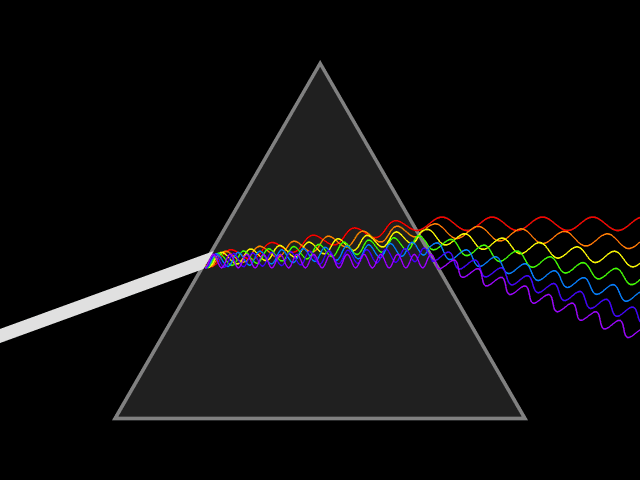
Dispersion (optics)
Dispersion is the phenomenon in which the phase velocity of a wave depends on its frequency. Sometimes the term chromatic dispersion is used to refer to optics specifically, as opposed to wave propagation in general. A medium having this common property may be termed a dispersive medium. Although the term is used in the field of optics to describe light and other electromagnetic waves, dispersion in the same sense can apply to any sort of wave motion such as acoustic dispersion in the case of sound and seismic waves, and in gravity waves (ocean waves). Within optics, dispersion is a property of telecommunication signals along transmission lines (such as microwaves in coaxial cable) or the Pulse (signal processing), pulses of light in optical fiber. In optics, one important and familiar consequence of dispersion is the change in the angle of refraction of different colors of light, as seen in the spectrum produced by a dispersive Prism (optics), prism and in chromatic aberration ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Refractive Index
In optics, the refractive index (or refraction index) of an optical medium is the ratio of the apparent speed of light in the air or vacuum to the speed in the medium. The refractive index determines how much the path of light is bent, or refraction, refracted, when entering a material. This is described by Snell's law of refraction, , where and are the angle of incidence (optics), angle of incidence and angle of refraction, respectively, of a ray crossing the interface between two media with refractive indices and . The refractive indices also determine the amount of light that is reflectivity, reflected when reaching the interface, as well as the critical angle for total internal reflection, their intensity (Fresnel equations) and Brewster's angle. The refractive index, n, can be seen as the factor by which the speed and the wavelength of the radiation are reduced with respect to their vacuum values: the speed of light in a medium is , and similarly the wavelength in that me ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Germanium Tetrachloride
Germanium tetrachloride is a colourless, fuming liquid with a peculiar, acidic odour. It is used as an intermediate in the production of purified germanium metal. In recent years, GeCl4 usage has increased substantially due to its use as a reagent for fiber optic production. Production Most commercial production of germanium is from treating flue-dusts of zinc- and copper-ore smelters, although a significant source is also found in the ash from the combustion of certain types of coal called vitrain. Germanium tetrachloride is an intermediate for the purification of germanium metal or its oxide, GeO2."Germanium" Mineral Commodity Profile, U.S. Geological Survey, 2005. Germanium tetrachloride can be generated directly from GeO2 ( germanium dioxide) by dissolution of the oxide in concentrated hydrochloric acid. The resulting mixture is fractionally distilled to purify and separate the germanium tetrachloride from other products and impurities."The Elements" C. R. Hammond, David R. L ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrochloric Acid
Hydrochloric acid, also known as muriatic acid or spirits of salt, is an aqueous solution of hydrogen chloride (HCl). It is a colorless solution with a distinctive pungency, pungent smell. It is classified as a acid strength, strong acid. It is a component of the gastric acid in the digestive systems of most animal species, including humans. Hydrochloric acid is an important laboratory reagent and industrial chemical. Etymology Because it was produced from halite, rock salt according to the methods of Johann Rudolph Glauber, hydrochloric acid was historically called by European alchemists ''spirits of salt'' or ''acidum salis'' (salt acid). Both names are still used, especially in other languages, such as , , , , , , , , , , (''ensan''), zh, 盐酸 (''yánsuān''), and (''yeomsan''). Gaseous HCl was called ''marine acid air''. The name ''muriatic acid'' has the same origin (''muriatic'' means "pertaining to brine or salt", hence ''muriate'' means hydrochloride), and this ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
O-diphenols
In organic chemistry, dihydroxybenzenes (benzenediols) are organic compounds in which two hydroxyl groups () are substituted onto a benzene ring (). These aromatic compounds are classed as phenols. There are three structural isomers: 1,2-dihydroxybenzene (the ''ortho'' isomer) is commonly known as catechol, 1,3-dihydroxybenzene (the ''meta'' isomer) is commonly known as resorcinol, and 1,4-dihydroxybenzene (the ''para'' isomer) is commonly known as hydroquinone. : All three of these compounds are colorless to white granular solids at room temperature and pressure, but upon exposure to oxygen they may darken. All three isomers have the chemical formula . Similar to other phenols, the hydroxyl groups on the aromatic ring of a benzenediol are weakly acidic. Each benzenediol can lose an from one of the hydroxyls to form a type of phenolate ion. The Dakin oxidation is an organic redox reaction in which an ''ortho''- or ''para''-hydroxylated phenyl aldehyde () or ketone () reacts w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyalcohol
Sugar alcohols (also called polyhydric alcohols, polyalcohols, alditols or glycitols) are organic compounds, typically derived from sugars, containing one hydroxyl group attached to each carbon atom. They are white, water-soluble solids that can occur naturally or be produced industrially by hydrogenating sugars. Since they contain multiple groups, they are classified as polyols. Sugar alcohols are used widely in the food industry as thickeners and sweeteners. In commercial foodstuffs, sugar alcohols are commonly used in place of table sugar (sucrose), often in combination with high-intensity artificial sweeteners, in order to offset their low sweetness. Xylitol and sorbitol are popular sugar alcohols in commercial foods. Structure Sugar alcohols have the general formula . In contrast, sugars have two fewer hydrogen atoms, for example, or . Like their parent sugars, sugar alcohols exist in diverse chain length. Most have five- or six-carbon chains, because they are derived ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carboxylic Acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an Substituent, R-group. The general formula of a carboxylic acid is often written as or , sometimes as with R referring to an organyl group (e.g., alkyl, alkenyl, aryl), or hydrogen, or other groups. Carboxylic acids occur widely. Important examples include the amino acids and fatty acids. Deprotonation of a carboxylic acid gives a carboxylate anion. Examples and nomenclature Carboxylic acids are commonly identified by their trivial names. They often have the suffix ''-ic acid''. IUPAC-recommended names also exist; in this system, carboxylic acids have an ''-oic acid'' suffix. For example, butyric acid () is butanoic acid by IUPAC guidelines. For nomenclature of complex molecules containing a carboxylic acid, the carboxyl can be considered position one of the parent chain even if there are other substituents, such as 3-chloropropanoic acid. Alternately, it can be named ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Germanate
In chemistry, germanate is a compound containing an oxyanion of germanium. In the naming of inorganic compounds it is a suffix that indicates a polyatomic anion with a central germanium atom, for example potassium hexafluorogermanate, K2GeF6.Egon Wiberg, Arnold Frederick Holleman (2001) ''Inorganic Chemistry'', Elsevier Germanate oxy compounds Germanium is similar to silicon forming many compounds with tetrahedral units although it can also exhibit 5 and 6 coordination. Analogues of all the major types of silicates and aluminosilicates have been prepared. For example, the compounds Mg2GeO4 (olivine and spinel forms), CaGeO3( perovskite structure), Be2GeO4 ( phenakite structure) show the resemblance to the silicates. BaGe4O9 has a complex structure containing 4 and 6 coordinate germanium. Germanates are important for geoscience as they possess similar structures to silicates and can be used as analogues for studying the behaviour of silicate minerals found in the Earth's mantl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |