|
Germain Hess
Germain Henri Hess (; 7 August 1802 – ) was a Swiss-Russian chemist and doctor who formulated Hess' law, an early principle of thermochemistry. Early life and education Hess was born on 7 August 1802 in Geneva, France (Switzerland after 1815). His father was an artist and in 1805 moved the family to the Russian Empire to work as a tutor to a rich family. His mother was a tutor as well and Hess learned German and French at home. In 1817, his family moved to Dorpat, Russian Empire (now Tartu, Estonia), where he went to a private school for two years, and then to Dorpat Gymnasium, which he finished in 1822. In autumn of the same year Hess studied medicine at the University of Dorpat. During that time, the chemistry department was responsible for the Chemistry courses of the Medicine and Pharmacy departments and Professor Gottfried W. Osann was giving the lectures in German (an obvious advance for Hess). Under Osann's supervision, Hess made chemical analyses, but also had an inte ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Geneva
Geneva ( , ; ) ; ; . is the List of cities in Switzerland, second-most populous city in Switzerland and the most populous in French-speaking Romandy. Situated in the southwest of the country, where the Rhône exits Lake Geneva, it is the capital of the Canton of Geneva, Republic and Canton of Geneva, and a centre for international diplomacy. Geneva hosts the highest number of International organization, international organizations in the world, and has been referred to as the world's most compact metropolis and the "Peace Capital". Geneva is a global city, an international financial centre, and a worldwide centre for diplomacy hosting the highest number of international organizations in the world, including the headquarters of many agencies of the United Nations and the International Committee of the Red Cross, ICRC and International Federation of Red Cross and Red Crescent Societies, IFRC of the International Red Cross and Red Crescent Movement, Red Cross. In the aftermath ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ural Mountains
The Ural Mountains ( ),; , ; , or simply the Urals, are a mountain range in Eurasia that runs north–south mostly through Russia, from the coast of the Arctic Ocean to the river Ural (river), Ural and northwestern Kazakhstan.Ural Mountains , Encyclopædia Britannica on-line The mountain range forms part of the Boundaries between the continents of Earth, conventional boundary between the continents of Europe and Asia, marking the separation between European Russia and Siberia. Vaygach Island and the islands of Novaya Zemlya form a further continuation of the chain to the north into the Arctic Ocean. The average altitudes of the Urals are around , the highest point being Mount Narodnaya, which reaches a height of . The mountains lie within the Ural (region), Ural geographical region and significantl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dissociation (chemistry)
Dissociation in chemistry is a general process in which molecules (or ionic compounds such as salt (chemistry), salts, or coordination complex, complexes) separate or split into other things such as atoms, ions, or radical (chemistry), radicals, usually in a reversible manner. For instance, when an acid dissolves in water, a covalent bond between an electronegativity, electronegative atom and a hydrogen atom is broken by heterolytic fission, which gives a proton (H+) and a negative ion. Dissociation is the opposite of association or recombination. Dissociation constant For reversible dissociations in a chemical equilibrium :AB A + B the dissociation constant ''K''d is the ratio of dissociated to undissociated compound :K_d = \mathrm where the brackets denote the equilibrium concentrations of the species. Dissociation degree The dissociation degree \alpha is the fraction of original solute molecules that have dissociated. It is usually indicated by the Greek symbol α. More acc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aqueous Solution
An aqueous solution is a solution in which the solvent is water. It is mostly shown in chemical equations by appending (aq) to the relevant chemical formula. For example, a solution of table salt, also known as sodium chloride (NaCl), in water would be represented as . The word ''aqueous'' (which comes from ''aqua'') means pertaining to, related to, similar to, or dissolved in, water. As water is an excellent solvent and is also naturally abundant, it is a ubiquitous solvent in chemistry. Since water is frequently used as the solvent in experiments, the word solution refers to an aqueous solution, unless the solvent is specified. A ''non-aqueous solution'' is a solution in which the solvent is a liquid, but is not water. Characteristics Substances that are ''hydrophobic'' ('water-fearing') do not dissolve well in water, whereas those that are '' hydrophilic'' ('water-friendly') do. An example of a hydrophilic substance is sodium chloride. In an aqueous solution the hydrogen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Salt (chemistry)
In chemistry, a salt or ionic compound is a chemical compound consisting of an assembly of positively charged ions ( cations) and negatively charged ions ( anions), which results in a compound with no net electric charge (electrically neutral). The constituent ions are held together by electrostatic forces termed ionic bonds. The component ions in a salt can be either inorganic, such as chloride (Cl−), or organic, such as acetate (). Each ion can be either monatomic, such as sodium (Na+) and chloride (Cl−) in sodium chloride, or polyatomic, such as ammonium () and carbonate () ions in ammonium carbonate. Salts containing basic ions hydroxide (OH−) or oxide (O2−) are classified as bases, such as sodium hydroxide and potassium oxide. Individual ions within a salt usually have multiple near neighbours, so they are not considered to be part of molecules, but instead part of a continuous three-dimensional network. Salts usually form crystalline structures ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Exchange Reactions
Exchange or exchanged may refer to: Arts, entertainment and media Film and television * Exchange (film), or ''Deep Trap'', 2015 South Korean psychological thriller * Exchanged (film), 2019 Peruvian fantasy comedy * Exchange (TV program), 2021 South Korean dating reality show Gaming * Exchange (chess), closely related captures of pieces of both players in chess **The exchange (chess), an exchange of a minor piece for a rook Music * Exchange, a new-age jazz band of Steve Sexton and Gerald O'Brien, and their 1992 self-titled album * ''Exchange'' (EP), by Against All Authority and The Criminals, 1999 * "Exchange" (song), by Bryson Tiller, 2015 * "Exchange" and "(Exchange)", songs on Massive Attack's 1998 album ''Mezzanine'' (album) Business and economics * Bureau de change, or currency exchange * Cryptocurrency exchange, to trade cryptocurrencies or digital currencies * Exchange (economics), in a market economy * Exchange (organized market), where securities etc are bought and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Julius Von Mayer
Julius Robert von Mayer (25 November 1814 – 20 March 1878) was a German physician, chemist, and physicist and one of the founders of thermodynamics. He is best known for enunciating in 1841 one of the original statements of the conservation of energy or what is now known as one of the first versions of the first law of thermodynamics, namely that "energy can be neither created nor destroyed". In 1842, Mayer described the vital chemical process now referred to as oxidation as the primary source of energy for any living creature. He also proposed that plants convert light into chemical energy. His achievements were overlooked and priority for the discovery in 1842 of the ''mechanical equivalent of heat'' was attributed to James Joule in the following year. Early life Mayer was born on 25 November 1814 in Heilbronn, Württemberg (Baden-Württemberg, modern day Germany), the son of a pharmacist. He grew up in Heilbronn. After completing his ''Abitur'', he studied medicine at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Conservation Of Energy
The law of conservation of energy states that the total energy of an isolated system remains constant; it is said to be Conservation law, ''conserved'' over time. In the case of a Closed system#In thermodynamics, closed system, the principle says that the total amount of energy within the system can only be changed through energy entering or leaving the system. Energy can neither be created nor destroyed; rather, it can only be transformed or transferred from one form to another. For instance, chemical energy is Energy conversion, converted to kinetic energy when a stick of dynamite explodes. If one adds up all forms of energy that were released in the explosion, such as the kinetic energy and potential energy of the pieces, as well as heat and sound, one will get the exact decrease of chemical energy in the combustion of the dynamite. Classically, the conservation of energy was distinct from the conservation of mass. However, special relativity shows that mass is related to en ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
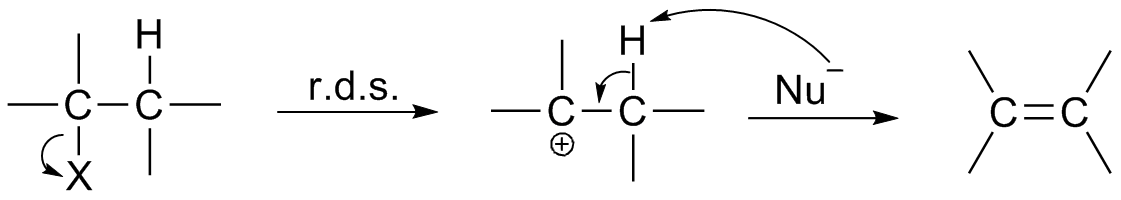
Reaction Intermediate
In chemistry, a reaction intermediate, or intermediate, is a molecular entity arising within the sequence of a stepwise chemical reaction. It is formed as the reaction product of an elementary step, from the reactants and/or preceding intermediates, but is consumed in a later step. It does not appear in the chemical equation for the overall reaction. For example, consider this hypothetical reaction: :A + B → C + D If this overall reaction comprises two elementary steps thus: :A + B → X :X → C + D then X is a reaction intermediate. The phrase ''reaction intermediate'' is often abbreviated to the single word ''intermediate'', and this is IUPAC's preferred form of the term. But this shorter form has other uses. It often refers to reactive intermediates. It is also used more widely for chemicals such as cumene which are traded within the chemical industry but are not generally of value outside it. IUPAC definition The IUPAC Gold Book defines an ''intermediate'' as a co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfuric Acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, and hydrogen, with the molecular formula . It is a colorless, odorless, and Viscosity, viscous liquid that is Miscibility, miscible with water. Pure sulfuric acid does not occur naturally due to its Dehydration reaction, strong affinity to water vapor; it is Hygroscopy, hygroscopic and readily absorbs water vapor from the Atmosphere of Earth, air. Concentrated sulfuric acid is a strong oxidant with powerful dehydrating properties, making it highly corrosive towards other materials, from rocks to metals. Phosphorus pentoxide is a notable exception in that it is not dehydrated by sulfuric acid but, to the contrary, dehydrates sulfuric acid to sulfur trioxide. Upon addition of sulfuric acid to water, a considerable amount of heat is releas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrate
In chemistry, a hydrate is a substance that contains water or its constituent elements. The chemical state of the water varies widely between different classes of hydrates, some of which were so labeled before their chemical structure was understood. Chemical nature Inorganic chemistry Hydrates are not inorganic salts "containing water molecules combined in a definite ratio as an integral part of the crystal" that are either bound to a metal center or that have crystallized with the metal complex. Such hydrates are also said to contain '' water of crystallization'' or ''water of hydration''. If the water is heavy water in which the constituent hydrogen is the isotope deuterium, then the term ''deuterate'' may be used in place of ''hydrate''. A colorful example is cobalt(II) chloride, which turns from blue to red upon hydration, and can therefore be used as a water indicator. The notation "''hydrated compound''⋅''n''", where ''n'' is the number of water molecules per form ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |