|
Double Bond Rule
In chemistry, the double bond rule states that elements with a principal quantum number (''n'') greater than 2 for their valence electrons ( period 3 elements and higher) tend not to form multiple bonds (e.g. double bonds and triple bonds). Double bonds for these heavier elements, when they exist, are often weak due to poor orbital overlap between the ''n''>2 orbitals of the two atoms. Although such compounds are not intrinsically unstable, they instead tend to dimerize or even polymerize In polymer chemistry, polymerization (American English), or polymerisation (British English), is a process of reacting monomer molecules together in a chemical reaction to form polymer chains or three-dimensional networks. There are many form .... Moreover, the multiple bonds of the elements with ''n''=2 are much stronger than usual, because lone pair repulsion weakens their sigma bonding but not their pi bonding. An example is the rapid polymerization that occurs upon condensation of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a physical science within the natural sciences that studies the chemical elements that make up matter and chemical compound, compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during chemical reaction, reactions with other chemical substance, substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both Basic research, basic and Applied science, applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the prop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diborene
Diborane(2), also known as diborene, is an inorganic compound with the formula B2H2. The number 2 in diborane(2) indicates the number of hydrogen atoms bonded to the boron complex. There are other forms of diborane with different numbers of hydrogen atoms, including diborane(4) and diborane(6). Diborane(2) is a highly reactive molecule that rapidly decomposes, making it a challenge to study experimentally under ambient conditions. To observe diborane(2) experimentally, high-vacuum and low temperature conditions using matrix isolation techniques are required, such as trapping the molecule in inert matrices like neon or argon. As a result of these difficult synthesis conditions, its properties and behaviour have been predominantly studied using theoretical models and computational simulations. Diborene also refers to a series of molecules with the formula R:(BH)=(BH):R or R-B=B-R where R is an organic group. Diborene derivatives are relatively stable and can be stored at room te ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitroso
In organic chemistry, nitroso refers to a functional group in which the nitric oxide () group is attached to an organic moiety. As such, various nitroso groups can be categorized as ''C''-nitroso compounds (e.g., nitrosoalkanes; ), ''S''-nitroso compounds ( nitrosothiols; ), ''N''-nitroso compounds (e.g., nitrosamines, ), and ''O''-nitroso compounds ( alkyl nitrites; ). Synthesis Nitroso compounds can be prepared by the reduction of nitro compounds or by the oxidation of hydroxylamines. Ortho-nitrosophenols may be produced by the Baudisch reaction. In the Fischer–Hepp rearrangement, aromatic 4-nitrosoanilines are prepared from the corresponding nitrosamines. Properties Nitrosoarenes typically participate in a monomer–dimer equilibrium. The azobenzene ''N'',''N'-''dioxide (Ar(–O)N+=+N(O–)Ar) dimers, which are often pale yellow, are generally favored in the solid state, whereas the deep-green monomers are favored in dilute solution or at higher temperatures. They ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
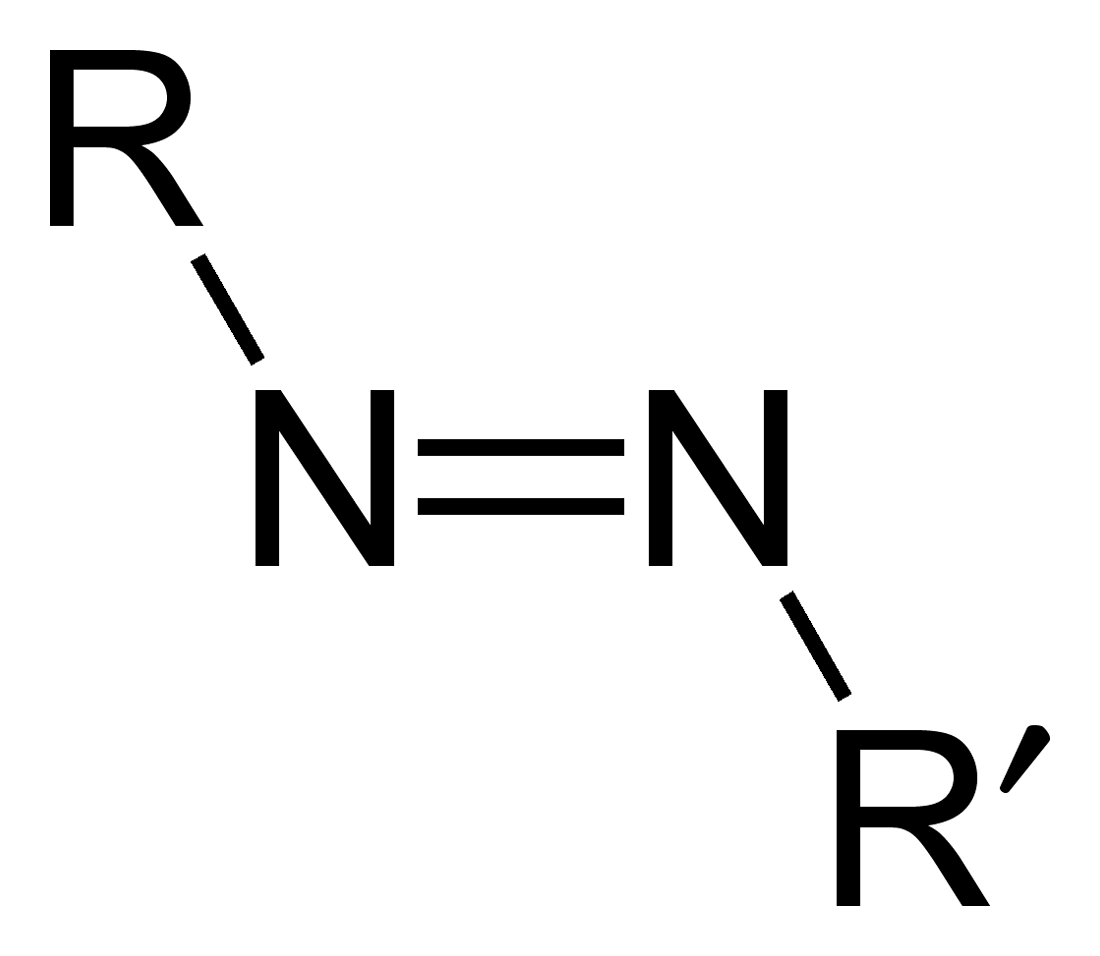
Azo Compound
Azo compounds are organic compounds bearing the functional group diazenyl (, in which R and R′ can be either aryl or alkyl groups). IUPAC defines azo compounds as: "Derivatives of diazene (diimide), , wherein both hydrogens are substituted by hydrocarbyl groups, e.g. azobenzene or diphenyldiazene.", where Ph stands for phenyl group. The more stable derivatives contain two aryl groups. The group is called an ''azo group'' (, ). Many textile and leather articles are dyed with azo dyes and pigments. Aryl azo compounds urinary tract infections">Phenazopyridine, an aryl azo compound, is used to treat urinary tract infections">150px Aryl azo compounds are usually stable, crystalline species. Azobenzene is the prototypical aromatic azo compound. It exists mainly as the Cis-trans isomerism, ''trans'' isomer, but upon illumination, converts to the Cis-trans isomerism, ''cis'' isomer. Aromatic azo compounds can be synthesized by azo coupling, which entails an electrophilic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thioketone
In organic chemistry, thioketones (; also known as thiones or thiocarbonyls) are organosulfur compounds related to conventional ketones in which the oxygen has been replaced by a sulfur. Instead of a structure of , thioketones have the structure , which is reflected by the prefix " thio-" in the name of the functional group. Thus the simplest thioketone is thioacetone, the sulfur analog of acetone. Unhindered alkylthioketones typically tend to form polymers or rings. Structure and bonding The C=S bond length of thiobenzophenone is 1.63 Å, which is comparable to 1.64 Å, the C=S bond length of thioformaldehyde, measured in the gas phase. Due to steric interactions, the phenyl groups are not coplanar and the dihedral angle SC-CC is 36°. Unhindered dialkylthiones polymerize or oligomerize but thiocamphor is well characterized red solid. Consistent with the double bond rule, most alkyl thioketones are unstable with respect to dimerization.Organosulfur Chemistry I: Topics in Cur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphaalkene
Phosphaalkenes (IUPAC name: alkylidenephosphanes) are organophosphorus compounds with double bonds between carbon and phosphorus(III) with the formula R2C=PR. In the compound phosphorine one carbon atom in benzene is replaced by phosphorus. The reactivity of phosphaalkenes is often compared to that of alkenes and not to that of imines because the HOMO of phosphaalkenes is not the phosphorus lone pair (as in imines the amine lone pair) but the double bond. Therefore like alkenes, phosphaalkenes engage in Wittig reactions, Peterson reactions, Cope rearrangements and Diels-Alder reactions. The first phosphaalkene discovered was a phosphabenzene, by Mërkl in 1969. The first localized phosphaalkene was reported in 1976 by Gerd Becker as a keto-enol tautomerism akin a Brook rearrangement: :: In the same year Harold Kroto established spectroscopically that thermolysis of Me2PH generates CH2=PMe. A general method for the synthesis of phosphaalkenes is by 1,2-elimination of suita ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silenes
In inorganic chemistry, silenes, or disilalkenes,Philip P. Power "pi-Bonding and the Lone Pair Effect in Multiple Bonds between Heavier Main Group Elements" Chemical Reviews, 1999, 99, 3462. are silicon compounds that contain double bonds, where the oxidation state of Si is +2. The parent molecule is disilene, Si2H4. Structure The first transient disilene was reported in 1972 by D. N. Roark and Garry J. D. Peddle. Simple disilenes easily polymerize. To suppress this tendency, bulky substituents are used. Indeed the first isolable disilene, tetramesityldisilene, was described in 1981 by West, Fink, and Michl. It was prepared by UV-photolysis of the related cyclic trisilane: : 2 i(mesityl)2sub>3 → 3 (mesityl)2Si=Si(mesityl)2 Structure of tetramesityldisilene Tetramesityldisilene is a yellow-orange solid. The Si=Si double bond lengths of disilenes vary between 2.14 and 2.29 Å and are nearly 5 to 10% shorter than the Si-Si single bond lengths of corresponding disilane ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonyl
In organic chemistry, a carbonyl group is a functional group with the formula , composed of a carbon atom double bond, double-bonded to an oxygen atom, and it is divalent at the C atom. It is common to several classes of organic compounds (such as aldehydes, ketones and carboxylic acid), as part of many larger functional groups. A compound containing a carbonyl group is often referred to as a carbonyl compound. The term carbonyl can also refer to carbon monoxide as a ligand in an inorganic or organometallic complex (a metal carbonyl, e.g. nickel carbonyl). The remainder of this article concerns itself with the organic chemistry definition of carbonyl, such that carbon and oxygen share a double bond. Carbonyl compounds In organic chemistry, a carbonyl group characterizes the following types of compounds: Other organic carbonyls are urea and the carbamates, the derivatives of acyl chlorides, chloroformates and phosgene, carbonate esters, thioesters, lactones, lactams, Hydro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Imine
In organic chemistry, an imine ( or ) is a functional group or organic compound containing a carbon–nitrogen double bond (). The nitrogen atom can be attached to a hydrogen or an organic group (R). The carbon atom has two additional single bonds. Imines are common in synthetic and naturally occurring compounds and they participate in many reactions. Distinction is sometimes made between aldimines and ketimines, derived from aldehydes and ketones, respectively. Structure In imines the five core atoms (C2C=NX, ketimine; and C(H)C=NX, aldimine; X = H or C) are coplanar. Planarity results from the sp2-hybridization of the mutually double-bonded carbon and the nitrogen atoms. The C=N distance is 1.29–1.31 Å for nonconjugated imines and 1.35 Å for conjugated imines. By contrast, C−N distances in amines and nitriles are 1.47 and 1.16 Å respectively. Rotation about the C=N bond is slow. Using NMR spectroscopy, both E–Z notation, ''E'' and ''Z'' isomers of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as Alpha-olefin, α-olefins. The International Union of Pure and Applied Chemistry (IUPAC) Preferred IUPAC name, recommends using the name "alkene" only for Open-chain compound, acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for Cyclic compound, cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with ''n'' being a >1 natural number (which is two hydrogens less than the corresponding alkane). When ''n'' is four or more, isomers are possible, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2,2,6,6-tetramethylpiperidine
2,2,6,6-Tetramethylpiperidine, abbreviated TMP, HTMP, or TMPH, is an organic compound of the amine class. In appearance, it is a colorless liquid and has a "fishy", amine-like odor. This amine is used in chemistry as a hindered base ( hindered amine). Although TMP finds limited use per se, its derivatives are a mainstay of hindered amine light stabilizers. TMP is the starting material for an even stronger base, lithium tetramethylpiperidide and the radical species TEMPO. Another non-nucleophilic base is N,N-diisopropylethylamine. Its aqueous p''K''aH (conjugate acid dissociation constant, a measure of basicity) is 11.07 at 25 °C, while its p''K''a (acid dissociation constant, a measure of acidity) is approximately 37. Preparation Many routes for the synthesis of TMP have been reported. One method starts with a conjugate addition reaction of ammonia to phorone. The intermediate triacetone amine is then reduced in a Wolff-Kishner reaction. See also * 2,6-Dimethylpi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mesityl
Mesitylene or 1,3,5-trimethylbenzene is a derivative of benzene with three methyl substituents positioned symmetrically around the ring. The other two isomeric trimethylbenzenes are 1,2,4-trimethylbenzene (pseudocumene) and 1,2,3-trimethylbenzene (hemimellitene). All three compounds have the formula C6H3(CH3)3, which is commonly abbreviated C6H3Me3. Mesitylene is a colorless liquid with sweet aromatic odor. It is a component of coal tar, which is its traditional source. It is a precursor to diverse fine chemicals. The mesityl group (Mes) is a substituent with the formula C6H2Me3 and is found in various other compounds. Preparation Mesitylene is prepared by transalkylation of xylene over solid acid catalyst:Karl Griesbaum, Arno Behr, Dieter Biedenkapp, Heinz-Werner Voges, Dorothea Garbe, Christian Paetz, Gerd Collin, Dieter Mayer, Hartmut Höke “Hydrocarbons” in Ullmann's Encyclopedia of Industrial Chemistry 2002 Wiley-VCH, Weinheim. . :2 C6H4(CH3)2 ⇌ C6H3(C ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |