|
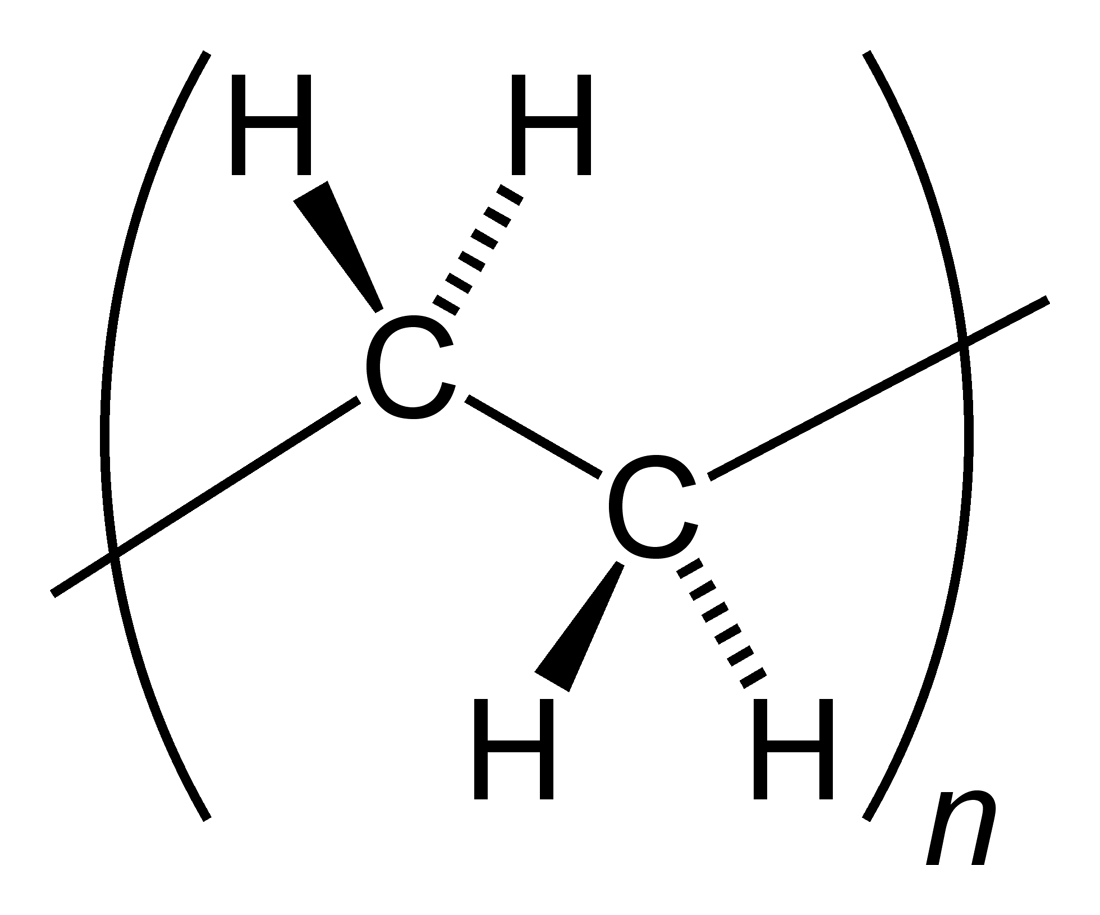
Polyolefins
A polyolefin is a type of polymer with the general formula (CH2CHR)n where R is an alkyl group. They are usually derived from a small set of simple olefins ( alkenes). Dominant in a commercial sense are polyethylene and polypropylene. More specialized polyolefins include polyisobutylene and polymethylpentene. They are all colorless or white oils or solids. Many copolymers are known, such as polybutene, which derives from a mixture of different butene isomers. The name of each polyolefin indicates the olefin from which it is prepared; for example, polyethylene is derived from ethylene, and polymethylpentene is derived from 4-methyl-1-pentene. Polyolefins are not olefins themselves because the double bond of each olefin monomer is opened in order to form the polymer. Monomers having more than one double bond such as butadiene and isoprene yield polymers that contain double bonds ( polybutadiene and polyisoprene) and are usually not considered polyolefins. Polyolefins are t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymethylpentene
Polymethylpentene (PMP), also known as poly(4-methyl-1-pentene). It is used for gas-permeable packaging, autoclavable medical and laboratory equipment, microwave components, and cookware. It is commonly called TPX, which is a trademark of Mitsui Chemicals. Production Polymethylpentene is a 4-methyl-1-pentene-derived linear isotactic polyolefin and is made by Ziegler–Natta type catalysis. The commercially available grades are usually copolymers. It can be extruded and moulded (by injection moulding or blow moulding). Physical properties Polymethylpentene melts at ≈ 235 °C. It has a relatively low density (0.84 g/cm3) among plastics and is transparent. It has low moisture absorption, and exceptional acoustical and electrical properties. Its properties are reasonably similar to those of other polyolefins, although it is more brittle and more gas permeable. The polymer also has a high thermal stability, excellent dielectric characteristics and a high chemical re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyethylene
Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a polymer, primarily used for packaging (plastic bags, plastic films, geomembranes and containers including bottles, cups, jars, etc.). , over 100 million tonnes of polyethylene resins are being produced annually, accounting for 34% of the total plastics market. Many kinds of polyethylene are known, with most having the chemical formula (C2H4)''n''. PE is usually a mixture of similar polymers of ethylene, with various values of ''n''. It can be ''low-density'' or ''high-density'' and many variations thereof. Its properties can be modified further by crosslinking or copolymerization. All forms are nontoxic as well as chemically resilient, contributing to polyethylene's popularity as a multi-use plastic. However, polyethylene's chemical resilience also makes it a long-lived and decomposition-resistant pollutant when disposed of improperly. Being a h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymer
A polymer () is a chemical substance, substance or material that consists of very large molecules, or macromolecules, that are constituted by many repeat unit, repeating subunits derived from one or more species of monomers. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compound (chemistry), compounds, produces unique physical property, physical properties including toughness, high rubber elasticity, elasticity, viscoelasticity, and a tendency to form Amorphous solid, amorphous and crystallization of polymers, semicrystalline structures rath ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Comonomer
In polymer chemistry, a comonomer refers to a polymerizable precursor to a copolymer aside from the principal monomer. In some cases, only small amounts of a comonomer are employed, in other cases substantial amounts of comonomers are used. Furthermore, in some cases, the comonomers are statistically incorporated within the polymer chain, whereas in other cases, they aggregate. The distribution of comonomers is referred to as the " blockiness" of a copolymer. Polyolefins 1-Octene, 1-hexene, and 1-butene are used comonomers in the manufacture of polyethylene Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a polymer, primarily used for packaging (plastic bags, plastic films, geomembranes and containers including bott ...s. The advantages to such copolymers has led to a focus on catalysts that facilitate the incorporation of these comonomers, e.g., constrained geometry complexes. Comonome ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polypropylene
Polypropylene (PP), also known as polypropene, is a thermoplastic polymer used in a wide variety of applications. It is produced via chain-growth polymerization from the monomer Propene, propylene. Polypropylene belongs to the group of polyolefins and is Crystallization of polymers#Degree of crystallinity, partially crystalline and Chemical polarity#Nonpolar molecules, non-polar. Its properties are similar to polyethylene, but it is slightly harder and more heat-resistant. It is a white, mechanically rugged material and has a high chemical resistance. Polypropylene is the second-most widely produced Commodity plastics, commodity plastic (after polyethylene). History Phillips Petroleum chemists J. Paul Hogan and Robert Banks (chemist), Robert Banks first demonstrated the polymerization of propylene in 1951. The stereoselective polymerization to the isotactic was discovered by Giulio Natta and Karl Rehn in March 1954. This pioneering discovery led to large-scale commercial producti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyisobutene
Polyisobutene (polyisobutylene) is a class of organic polymers prepared by polymerization of isobutene. The polymers often have the formula Me3C H2CMe2sub>nH (Me = CH3). They are typically colorless gummy solids. Cationic polymerization, initiated with a strong Brønsted or Lewis acid, is the typical method for its production. The molecular weight (MW) of the resulting polymer determines the applications. Low MW polyisobutene, a mixture of oligomers with Mns of about 500, is used as plasticizers. Medium and high MW polyisobutenes, with Mn ≥ 20,000, are components of commercial adhesive Adhesive, also known as glue, cement, mucilage, or paste, is any non-metallic substance applied to one or both surfaces of two separate items that binds them together and resists their separation. The use of adhesives offers certain advantage ...s.{{cite encyclopedia , author1=Kenneth S. Whiteley , author2=T. Geoffrey Heggs , author3=Hartmut Koch , author4=Ralph L. Mawer , author5=Wol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polybutene
Polybutene is an organic polymer made from a mixture of 1-butene, 2-butene, and isobutylene. Ethylene steam cracker C4s are also used as supplemental feed for polybutene. It is similar to polyisobutylene (PIB), which is produced from essentially pure isobutylene made in a C4 complex of a major refinery. The presence of isomers other than isobutylene can have several effects including: 1) lower reactivity due to steric hindrance at the terminal carbon in, e.g., manufacture of polyisobutenyl succinic anhydride (PIBSA) dispersant manufacture; 2) the molecular weight—viscosity relationships of the two materials may also be somewhat different. Applications Industrial product applications include sealants, adhesives, extenders for putties used for sealing roofs and windows, coatings, polymer modification, tackified polyethylene films, personal care, polybutene emulsions. Hydrogenated polybutenes are used in a wide variety of cosmetic preparations, such as lipstick and lip gloss ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triethylaluminium
Triethylaluminium is one of the simplest examples of an organoaluminium compound. Despite its name the compound has the formula Al2( C2H5)6 (abbreviated as Al2Et6 or TEA). This colorless liquid is pyrophoric. It is an industrially important compound, closely related to trimethylaluminium. Structure and bonding The structure and bonding in Al2R6 and diborane are analogous (R = alkyl). Referring to Al2Me6, the Al-C(terminal) and Al-C(bridging) distances are 1.97 and 2.14 Å, respectively. The Al center is tetrahedral. The carbon atoms of the bridging ethyl groups are each surrounded by five neighbors: carbon, two hydrogen atoms and two aluminium atoms. The ethyl groups interchange readily intramolecularly. At higher temperatures, the dimer cracks into monomeric AlEt3. Synthesis and reactions Triethylaluminium can be formed via several routes. The discovery of an efficient route was a significant technological achievement. The multistep process uses aluminium, hydrogen gas, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phillips Catalyst
The Phillips catalyst, or the Phillips supported chromium catalyst, is the catalyst used to produce approximately half of the world's polyethylene. A heterogeneous catalyst, it consists of a chromium oxide supported on silica gel. Polyethylene, the most-produced synthetic polymer, is produced industrially by the polymerization of ethylene: :n C2H4 → (C2H4)n Although exergonic (i.e., thermodynamically favorable), the reaction requires catalysts. Three main catalysts are employed commercially: the Phillips catalyst, Ziegler–Natta catalysts (based on titanium trichloride), and, for specialty polymers, metallocene-based catalysts. Preparation and mechanism of action The Phillips catalyst is prepared by impregnating high surface area silica gel with chromium trioxide or related chromium compounds. The solid precatalyst is then calcined in air to give the active catalyst. Only a fraction of the chromium is catalytically active, a fact that interferes with elucidation of t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kaminsky Catalyst
A Kaminsky catalyst is a catalytic system for alkene polymerization. Kaminsky catalysts are based on metallocenes of group 4 transition metals (Ti, Zr, Hf) activated with methylaluminoxane (MAO). These and other innovations have inspired development of new classes of catalysts that in turn led to commercialization of novel engineering polyolefins. : __TOC__ Catalyst development The catalyst is named after German chemist Walter Kaminsky, who first described it in 1980 along with Hansjörg Sinn and others. Prior to Kaminsky's work, titanium chlorides supported on various materials were widely used (and still are) as heterogeneous catalysts for alkene polymerization. These halides are typically activated by treatment with trimethylaluminium. Kaminsky discovered that titanocene and related complexes emulated some aspects of these Ziegler–Natta catalysts but with low activity. He subsequently found that high activity could be achieved upon activation of these metallocenes with me ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tacticity
Tacticity (from , "relating to arrangement or order") is the relative stereochemistry of adjacent chiral centers within a macromolecule. The practical significance of tacticity rests on the effects on the physical properties of the polymer. The regularity of the macromolecular structure influences the degree to which it has rigid, crystalline long range order or flexible, amorphous long range disorder. Precise knowledge of tacticity of a polymer also helps understanding at what temperature a polymer melts, how soluble it is in a solvent, as well as its mechanical properties. A tactic macromolecule in the IUPAC definition is a macromolecule in which essentially all the configurational (repeating) units are identical. In a hydrocarbon macromolecule with all carbon atoms making up the backbone in a tetrahedral molecular geometry, the zigzag backbone is in the paper plane with the substituents either sticking out of the paper or retreating into the paper;, this projection is called ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |