|
Phosphorus Oxoacids
In chemistry, phosphorus oxoacid (or phosphorus acid) is a generic name for any acid whose molecule consists of atoms of phosphorus, oxygen, and hydrogen. There is a potentially infinite number of such compounds. Some of them are unstable and have not been isolated, but the derived anions and functional group, organic groups are present in stable salt (chemistry), salts and esters. The most important ones—in biology, geology, industry, and chemical research—are the phosphoric acids, whose esters and salts are the phosphates. In general, any hydrogen atom bonded to an oxygen atom is acidic, meaning that the –OH group can lose a proton leaving a negatively charged – group and thus turning the acid into a phosphorus oxoanion. Each additional proton lost has an associated acid dissociation constant K''a''1, K''a''2 K''a''3, ..., often expressed by its cologarithm (pK''a''1, pK''a''2, pK''a''3, ...). Hydrogen atoms bonded directly to phosphorus are generally not acidic. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a physical science within the natural sciences that studies the chemical elements that make up matter and chemical compound, compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during chemical reaction, reactions with other chemical substance, substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both Basic research, basic and Applied science, applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the prop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
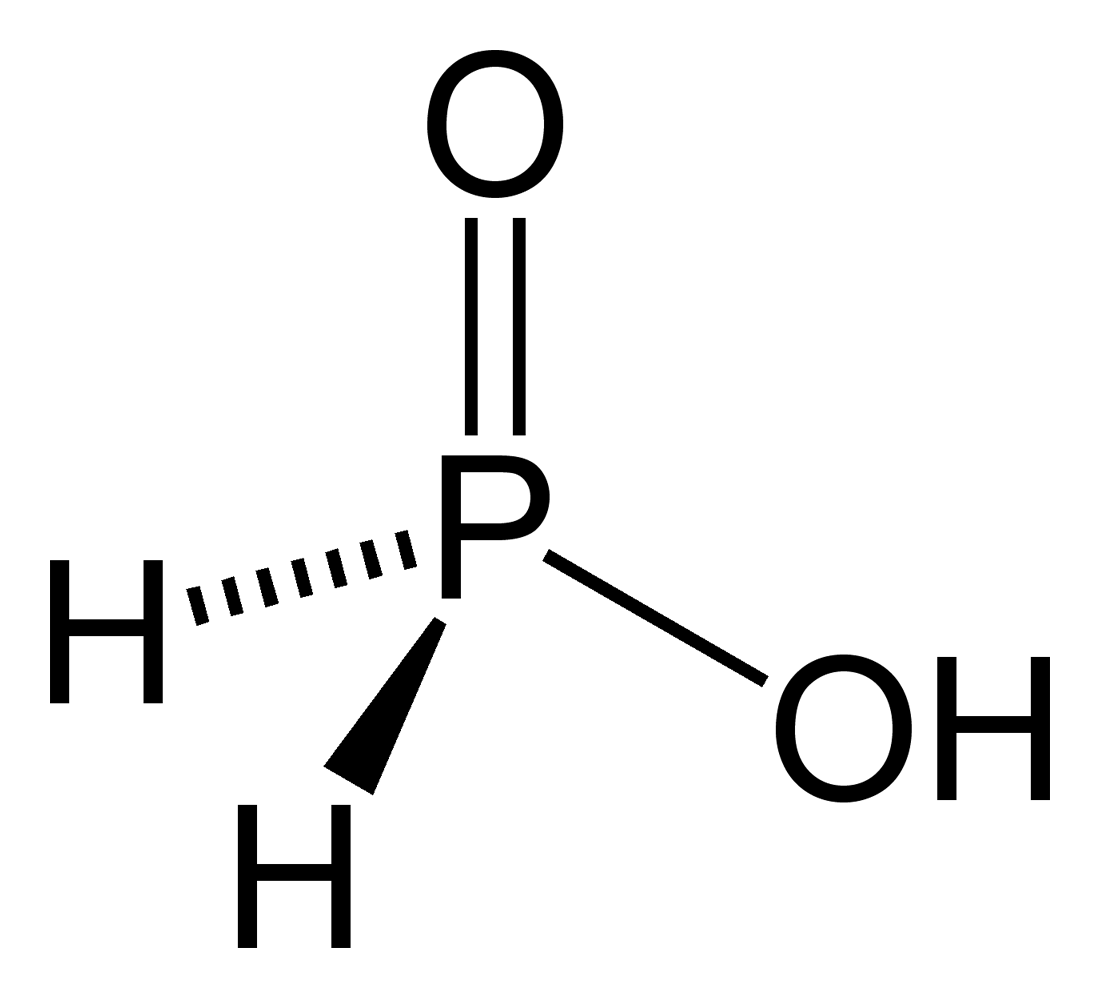
Hypophosphorous Acid
Hypophosphorous acid (HPA), or phosphinic acid, is a phosphorus oxyacid and a powerful reducing agent with molecular formula H3PO2. It is a colorless low-melting compound, which is soluble in water, dioxane and alcohols. The formula for this acid is generally written H3PO2, but a more descriptive presentation is HOP(O)H2, which highlights its monoprotic character. Salts derived from this acid are called hypophosphites. HOP(O)H2 exists in equilibrium with the minor tautomer HP(OH)2. Sometimes the minor tautomer is called hypophosphorous acid and the major tautomer is called phosphinic acid. Preparation and availability Hypophosphorous acid was first prepared in 1816 by the French chemist Pierre Louis Dulong (1785–1838). The acid is prepared industrially via a two step process: Firstly, elemental white phosphorus reacts with alkali and alkaline earth hydroxides to give an aqueous solution of hypophosphites: :P4 + 4 OH− + 4 H2O → 4 + 2 H2 Any pho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cycle Basis
In graph theory, a branch of mathematics, a cycle basis of an undirected graph is a set of simple cycles that forms a basis of the cycle space of the graph. That is, it is a minimal set of cycles that allows every even-degree subgraph to be expressed as a symmetric difference of basis cycles. A fundamental cycle basis may be formed from any spanning tree or spanning forest of the given graph, by selecting the cycles formed by the combination of a path in the tree and a single edge outside the tree. Alternatively, if the edges of the graph have positive weights, the minimum weight cycle basis may be constructed in polynomial time. In planar graphs, the set of bounded cycles of an embedding of the graph forms a cycle basis. The minimum weight cycle basis of a planar graph corresponds to the Gomory–Hu tree of the dual graph. Definitions A spanning subgraph of a given graph ''G'' has the same set of vertices as ''G'' itself but, possibly, fewer edges. A graph ''G'', or one of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrahedron
In geometry, a tetrahedron (: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular Face (geometry), faces, six straight Edge (geometry), edges, and four vertex (geometry), vertices. The tetrahedron is the simplest of all the ordinary convex polytope, convex polyhedra. The tetrahedron is the three-dimensional case of the more general concept of a Euclidean geometry, Euclidean simplex, and may thus also be called a 3-simplex. The tetrahedron is one kind of pyramid (geometry), pyramid, which is a polyhedron with a flat polygon base and triangular faces connecting the base to a common point. In the case of a tetrahedron, the base is a triangle (any of the four faces can be considered the base), so a tetrahedron is also known as a "triangular pyramid". Like all convex polyhedra, a tetrahedron can be folded from a single sheet of paper. It has two such net (polyhedron), nets. For any tetrahedron there exists a sphere (called th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Double Bond
In chemistry, a double bond is a covalent bond between two atoms involving four bonding electrons as opposed to two in a single bond. Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist between two different elements: for example, in a carbonyl group between a carbon atom and an oxygen atom. Other common double bonds are found in azo compounds (N=N), imines (C=N), and sulfoxides (S=O). In a skeletal formula, a double bond is drawn as two parallel lines (=) between the two connected atoms; typographically, the equals sign is used for this. Double bonds were introduced in chemical notation by Russian chemist Alexander Butlerov. Double bonds involving carbon are stronger and shorter than single bonds. The bond order is two. Double bonds are also electron-rich, which makes them potentially more reactive in the presence of a strong electron acceptor (as in addition reactions of the halogens). File:Ethene structural.svg, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Bond
A chemical bond is the association of atoms or ions to form molecules, crystals, and other structures. The bond may result from the electrostatic force between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bonds, or some combination of these effects. Chemical bonds are described as having different strengths: there are "strong bonds" or "primary bonds" such as covalent, ionic and metallic bonds, and "weak bonds" or "secondary bonds" such as dipole–dipole interactions, the London dispersion force, and hydrogen bonding. Since opposite electric charges attract, the negatively charged electrons surrounding the nucleus and the positively charged protons within a nucleus attract each other. Electrons shared between two nuclei will be attracted to both of them. "Constructive quantum mechanical wavefunction interference" stabilizes the paired nuclei (see Theories of chemical bonding). Bonded nuclei maintain an optima ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphoric Acids And Phosphates
In chemistry, a phosphoric acid, in the general sense, is a phosphorus oxoacid in which each phosphorus (P) atom is in the oxidation state +5, and is bonded to four oxygen (O) atoms, one of them through a double bond, arranged as the corners of a tetrahedron. Two or more of these tetrahedra may be connected by shared single-bonded oxygens, forming linear or branched chains, cycles, or more complex structures. The single-bonded oxygen atoms that are not shared are completed with acidic hydrogen atoms. The general formula of a phosphoric acid is , where ''n'' is the number of phosphorus atoms and ''x'' is the number of fundamental cycles in the molecule's structure, between 0 and . Removal of protons () from ''k'' hydroxyl groups –OH leaves anions generically called phosphates (if ) or hydrogen phosphates (if ''k'' is between 1 and ), with general formula . The fully dissociated anion () has formula . The term phosphate is also used in organic chemistry for the funct ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hypophosphate
Hypophosphoric acid is a mineral acid with the formula H4P2O6, with phosphorus in a formal oxidation state of +4. In the solid state it is present as the dihydrate, H4P2O6·2H2O. In hypophosphoric acid the phosphorus atoms are identical and joined directly with a P−P bond. Isohypophosphoric acid is a structural isomer of hypophosphoric acid in which one phosphorus has a hydrogen directedly bonded to it and that phosphorus atom is linked to the other one by an oxygen bridge to give a phosphorous acid/phosphoric acid mixed anhydride. The two phosphorus atoms are in the +3 and +5 oxidation states, respectively. Preparation and reactions Hypophosphoric acid can be prepared by the reaction of red phosphorus with sodium chlorite at room temperature.Phosphorus: Chemistry, Biochemistry and Technology, Sixth Edition, 2013, D.E.C. Corbridge, CRC Pres, Taylor Francis Group, :2 P + 2 NaClO2 + 2 H2O → Na2H2P2O6 + 2 HCl A mixture of hypophosphoric acid, phosphorous acid (H3PO3) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphite
The general structure of a phosphite ester showing the lone pairs on the P In organic chemistry, a phosphite ester or organophosphite usually refers to an organophosphorous compound with the formula P(OR)3. They can be considered as esters of an unobserved tautomer phosphorous acid, H3PO3, with the simplest example being trimethylphosphite, P(OCH3)3. Some phosphites can be considered esters of the dominant tautomer of phosphorous acid (HP(O)(OH)2). The simplest representative is dimethylphosphite with the formula HP(O)(OCH3)2. Both classes of phosphites are usually colorless liquids. Synthesis ;From PCl3 Phosphite esters are typically prepared by treating phosphorus trichloride with an alcohol. For alkyl alcohols the displaced chloride ion can attack the phosphite, causing dealkylation to give a dialkylphosphite and an organochlorine compound. The overall reaction is as follows: :PCl3 + 3 C2H5OH → (C2H5O)2P(O)H + 2 HCl + C2H5Cl Alternatively, when the alcoh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diprotic Acid
An acid is a molecule or ion capable of either donating a proton (i.e. hydrogen cation, H+), known as a Brønsted–Lowry acid, or forming a covalent bond with an electron pair, known as a Lewis acid. The first category of acids are the proton donors, or Brønsted–Lowry acids. In the special case of aqueous solutions, proton donors form the hydronium ion H3O+ and are known as Arrhenius acids. Brønsted and Lowry generalized the Arrhenius theory to include non-aqueous solvents. A Brønsted–Lowry or Arrhenius acid usually contains a hydrogen atom bonded to a chemical structure that is still energetically favorable after loss of H+. Aqueous Arrhenius acids have characteristic properties that provide a practical description of an acid. Acids form aqueous solutions with a sour taste, can turn blue litmus red, and react with bases and certain metals (like calcium) to form salts. The word ''acid'' is derived from the Latin , meaning 'sour'. An aqueous solution of an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorous Acid
Phosphorous acid (or phosphonic acid) is the Compound (chemistry), compound described by the chemical formula, formula . It is diprotic (readily ionizes two protons), not triprotic as might be suggested by its formula. Phosphorous acid is an intermediate in the preparation of other phosphorus compounds. Organic derivatives of phosphorous acid, compounds with the formula , are called phosphonic acids. Nomenclature and tautomerism Solid has tetrahedral geometry about the central phosphorus atom, with a bond of 132 picometer, pm, one double bond of 148 pm and two longer single bonds of 154 pm. In common with other phosphorus oxides with bonds (e.g.hypophosphorous acid and dialkyl phosphites), it exists in equilibrium with an extremely minor tautomer . (In contrast, arsenous acid's major tautomer is the trihydroxy form.) IUPAC recommends that the trihydroxy form be called phosphorous acid, and the dihydroxy form phosphonic acid.. Only the reduced phosphorus c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |