|
CrO2Cl2
Chromyl chloride is an inorganic compound with the formula CrO2Cl2. It is a reddish brown compound that is a volatile liquid at room temperature, which is unusual for transition metal compounds. It is the dichloride of chromic acid. Preparation Chromyl chloride can be prepared by the reaction of potassium chromate or potassium dichromate with hydrogen chloride in the presence of concentrated sulfuric acid, followed by distillation. :K2Cr2O7 + 6 HCl → 2 CrO2Cl2 + 2 KCl + 3 H2O The sulfuric acid serves as a dehydration agent. It can also be prepared directly by exposing chromium trioxide to anhydrous hydrogen chloride gas. :CrO3 + 2 HCl ⇌ CrO2Cl2 + H2O Usage Test for the presence of chlorides The chromyl chloride test involves heating a sample suspected to contain chlorides with potassium dichromate and concentrated sulfuric acid. If a chloride is present then chromyl chloride forms which is indicated by the evolution of red smoke. No similar compound is formed in the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromyl Fluoride
Chromyl fluoride is an inorganic compound with the formula . It is a violet-red colored crystalline solid that melts to an orange-red liquid.Gard, G. L. (1986) "Chromium Difluoride Dioxide (Chromyl Fluoride)," '' Inorg. Synth.'', 24, 67-69, . Structure The liquid and gaseous have a tetrahedral geometry with C2v symmetry, much like chromyl chloride. Chromyl fluoride dimerizes via fluoride bridges (as ) in the solid state, crystallizing in the P21/c space group with Z = 4. The Cr=O bond lengths are about 157 pm, and the Cr–F bond lengths are 181.7, 186.7, and 209.4 pm. Chromium resides in a distorted octahedral position with a coordination number of 6. History and preparation Pure chromyl fluoride was first isolated in 1952 as reported by Alfred Engelbrecht and Aristid von Grosse.Engelbrecht, A.; von Grosse, A. (1952) "Pure Chromyl Fluoride," '' J. Am. Chem. Soc.'' 74(''21''), 5262–5264, . It was first observed as red vapor in the early 19th century upon heatin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
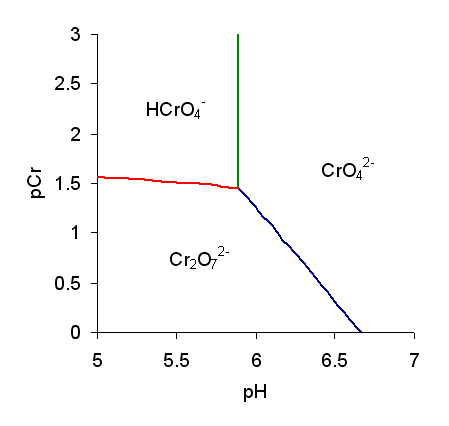
Chromic Acid
Chromic acid is a chemical compound with the chemical formula . It is also a jargon for a solution formed by the addition of sulfuric acid to aqueous solutions of dichromate. It consists at least in part of chromium trioxide. The term "chromic acid" is usually used for a mixture made by adding concentrated sulfuric acid to a dichromate, which may contain a variety of compounds, including solid chromium trioxide. This kind of chromic acid may be used as a cleaning mixture for glass. Chromic acid may also refer to the molecular species, of which the trioxide is the anhydride. Chromic acid features chromium in an oxidation state of +6 (and a valence of VI or 6). It is a strong and corrosive oxidizing agent and a moderate carcinogen. Molecular chromic acid Molecular chromic acid, , in principle, resembles sulfuric acid, . It would ionize accordingly: : The p''K''a for the equilibrium is not well characterized. Reported values vary between about −0.8 to 1.6. The structur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromine
Bromine is a chemical element; it has chemical symbol, symbol Br and atomic number 35. It is a volatile red-brown liquid at room temperature that evaporates readily to form a similarly coloured vapour. Its properties are intermediate between those of chlorine and iodine. Isolated independently by two chemists, Carl Jacob Löwig (in 1825) and Antoine Jérôme Balard (in 1826), its name was derived , referring to its sharp and pungent smell. Elemental bromine is very reactive and thus does not occur as a free element in nature. Instead, it can be isolated from colourless soluble crystalline mineral halide Ionic salt, salts analogous to table salt, a property it shares with the other halogens. While it is rather rare in the Earth's crust, the high solubility of the bromide ion (Br) has caused its Bromine cycle, accumulation in the oceans. Commercially the element is easily extracted from brine evaporation ponds, mostly in the United States and Israel. The mass of bromine in the oce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluoride
Fluoride (). According to this source, is a possible pronunciation in British English. is an Inorganic chemistry, inorganic, Monatomic ion, monatomic Ion#Anions and cations, anion of fluorine, with the chemical formula (also written ), whose salts are typically white or colorless. Fluoride salts typically have distinctive bitter tastes, and are odorless. Its salts and minerals are important Reagent, chemical reagents and industrial chemicals, mainly used in the production of hydrogen fluoride for fluorocarbons. Fluoride is classified as a weak base since it only partially associates in solution, but concentrated fluoride is corrosive and can attack the skin. Fluoride is the simplest fluorine anion. In terms of charge and size, the fluoride ion resembles the hydroxide ion. Fluoride ions occur on Earth in several minerals, particularly fluorite, but are present only in trace quantities in bodies of water in nature. Nomenclature Fluorides include compounds that contain ionic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromium(VI) Compounds
Chromium is a chemical element; it has symbol Cr and atomic number 24. It is the first element in group 6. It is a steely-grey, lustrous, hard, and brittle transition metal. Chromium is valued for its high corrosion resistance and hardness. A major development in steel production was the discovery that steel could be made highly resistant to corrosion and discoloration by adding metallic chromium to form stainless steel. Stainless steel and chrome plating (electroplating with chromium) together comprise 85% of the commercial use. Chromium is also greatly valued as a metal that is able to be highly polished while resisting tarnishing. Polished chromium reflects almost 70% of the visible spectrum, and almost 90% of infrared light. The name of the element is derived from the Greek word χρῶμα, ''chrōma'', meaning color, because many chromium compounds are intensely colored. Industrial production of chromium proceeds from chromite ore (mostly FeCr2O4) to produce ferrochrom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |