|
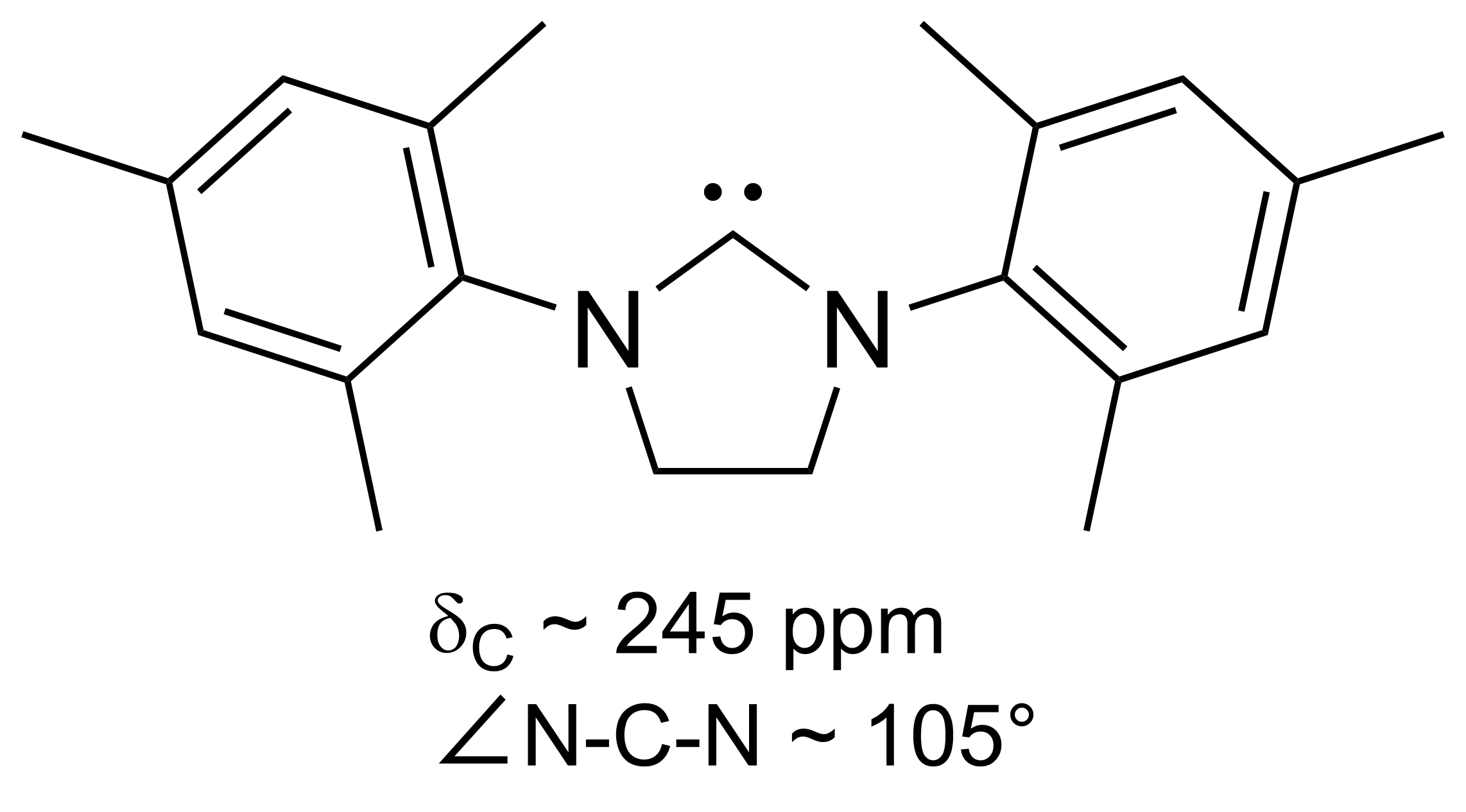
Arduengo Carbene
A persistent carbene (also known as stable carbene) is an organic molecule whose natural resonance structure has a carbon atom with incomplete octet (a carbene), but does not exhibit the tremendous instability typically associated with such moieties. The best-known examples and by far largest subgroup are the ''N''-heterocyclic carbenes (NHC) (sometimes called Arduengo carbenes), in which nitrogen atoms flank the formal carbene. Modern theoretical analysis suggests that the term "persistent carbene" is in fact a misnomer. Persistent carbenes do not in fact have a carbene electronic structure in their ground state, but instead an ylide stabilized by aromatic resonance or steric shielding. Excitation to a carbene structure then accounts for the carbene-like dimerization that some persistent carbenes undergo over the course of days. Persistent carbenes in general, and Arduengo carbenes in particular, are popular ligands in organometallic chemistry. History Early evidence In 1 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SIMes
SIMes (or H2Imes) is an N-heterocyclic carbene, ''N''-heterocyclic carbene. It is a white solid that dissolves in organic solvents. The compound is used as a ligand in organometallic chemistry. It is structurally related to the more common ligand IMes but with a saturated backbone (the S of SIMes indicates a saturated backbone). It is slightly more flexible and is a component in Grubbs' catalyst#Second generation catalyst, Grubbs II. It is prepared by alkylation of trimethylaniline by dibromoethane followed by ring closure and dehydrohalogenation. References [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzoin Condensation
In organic chemistry, the benzoin addition is an addition reaction involving two aldehydes (). The reaction generally occurs between aromatic compound, aromatic aldehydes or glyoxals (), and results in formation of an acyloin (). In the classic example, benzaldehyde is converted to Benzoin (organic compound), benzoin (). The benzoin condensation was first reported in 1832 by Justus von Liebig and Friedrich Wöhler during their research on almond, bitter almond oil. The catalytic version of the reaction involving cyanide was developed by Nikolay Zinin in the late 1830s. Reaction mechanism The reaction is catalysis, catalyzed by nucleophile, nucleophiles such as a cyanide or an persistent carbene, N-heterocyclic carbene (usually Thiazolium salt, thiazolium salts). The reaction mechanism was proposed in 1903 by Arthur Lapworth, A. J. Lapworth. In the first step in this reaction, the cyanide anion (as sodium cyanide) reacts with the aldehyde in a nucleophilic addition. Rearrangemen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vacuum Pyrolysis
Pyrolysis is a process involving the Bond cleavage, separation of covalent bonds in organic matter by thermal decomposition within an Chemically inert, inert environment without oxygen. Etymology The word ''pyrolysis'' is coined from the Greek language, Greek-derived morpheme, elements ''pyro-'' (from Ancient Greek : - "fire, heat, fever") and ''lysis'' ( : - "separation, loosening"). Applications Pyrolysis is most commonly used in the treatment of organic compound, organic materials. It is one of the processes involved in the charring of wood or pyrolysis of biomass. In general, pyrolysis of organic substances produces volatile products and leaves Char (chemistry), char, a carbon-rich solid residue. Extreme pyrolysis, which leaves mostly carbon as the residue, is called carbonization. Pyrolysis is considered one of the steps in the processes of gasification or combustion. Laypeople often confuse pyrolysis gas with syngas. Pyrolysis gas has a high percentage of heavy tar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dihydroimidazol-2-ylidene
Dihydroimidazol-2-ylidene is a hypothetical organic compound with formula C3H6N2. It would be a heterocyclic compound, formally derived from imidazolidine with two hydrogen atoms removed from carbon number 2, leaving two vacant chemical bonds — which makes it a carbene. Although carbenes in general are extremely short-lived, some derivatives of this compound are surprisingly stable, and form an important class of the persistent carbenes. They include the first stable carbenes postulated (but not isolated) by Hans-Werner Wanzlick around 1960. They also include an example of the (saturated) imidazolin-2-ylidene (carbene) reported by Anthony Joseph Arduengo, III, A.J. Arduengo in 1995. References Carbenes Imidazolines Hypothetical chemical compounds {{heterocyclic-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hans-Werner Wanzlick
Hans-Werner Wanzlick (1917-1988) was a German chemist. A Professor of chemistry at Technische Universität Berlin he is notable for work on persistent carbenes and for proposing the Wanzlick equilibrium between saturated imidazolin-2-ylidenes and their dimers — which he called "''das doppelte Lottchen''", after a 1949 novel by Erich Kästner about a pair of mischievous twins. Anthony J. Arduengo, III (1999) ''Looking for Stable Carbenes: The Difficulty in Starting Anew''. Accounts of Chemical Research, volume 32, number 11, pages 913–921. References * Hans-Werner Wanzlick (1958), letter to Linus Pauling Linus Carl Pauling ( ; February 28, 1901August 19, 1994) was an American chemist and peace activist. He published more than 1,200 papers and books, of which about 850 dealt with scientific topics. ''New Scientist'' called him one of the 20 gre ... (in German). Linus Pauling's Correspondence, item #444.6. Academic staff of Technische Universität B ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Equilibrium
In a chemical reaction, chemical equilibrium is the state in which both the Reagent, reactants and Product (chemistry), products are present in concentrations which have no further tendency to change with time, so that there is no observable change in the properties of the Thermodynamic system, system. This state results when the forward reaction proceeds at the same rate as the Reversible reaction, reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but they are equal. Thus, there are no net changes in the concentrations of the reactants and products. Such a state is known as dynamic equilibrium. It is the subject of study of ''equilibrium chemistry''. Historical introduction The Concept learning, concept of chemical equilibrium was developed in 1803, after Claude Louis Berthollet, Berthollet found that some chemical reactions are Reversible reaction, reversible. For any reaction mixture to exist at equilibrium, the reaction rate, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Deuteron
Deuterium (hydrogen-2, symbol H or D, also known as heavy hydrogen) is one of two Stable isotope ratio, stable isotopes of hydrogen; the other is protium, or hydrogen-1, H. The deuterium atomic nucleus, nucleus (deuteron) contains one proton and one neutron, whereas the far more common H has no neutrons. The name ''deuterium'' comes from Greek ''Wikt:δεύτερος, deuteros'', meaning "second". American chemist Harold Urey discovered deuterium in 1931. Urey and others produced samples of heavy water in which the H had been highly concentrated. The discovery of deuterium won Urey a List of Nobel laureates in Chemistry, Nobel Prize in 1934. Nearly all deuterium found in nature was Big Bang nucleosynthesis, synthesized in the Big Bang 13.8 billion years ago, forming the primordial ratio of H to H (~26 deuterium nuclei per 10 hydrogen nuclei). Deuterium is subsequently produced by the slow stellar proton–proton chain, but rapidly destroyed by exothermic Nuclear fusio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proton
A proton is a stable subatomic particle, symbol , Hydron (chemistry), H+, or 1H+ with a positive electric charge of +1 ''e'' (elementary charge). Its mass is slightly less than the mass of a neutron and approximately times the mass of an electron (the proton-to-electron mass ratio). Protons and neutrons, each with a mass of approximately one Dalton (unit), dalton, are jointly referred to as ''nucleons'' (particles present in atomic nuclei). One or more protons are present in the Atomic nucleus, nucleus of every atom. They provide the attractive electrostatic central force which binds the atomic electrons. The number of protons in the nucleus is the defining property of an element, and is referred to as the atomic number (represented by the symbol ''Z''). Since each chemical element, element is identified by the number of protons in its nucleus, each element has its own atomic number, which determines the number of atomic electrons and consequently the chemical characteristi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heavy Water
Heavy water (deuterium oxide, , ) is a form of water (molecule), water in which hydrogen atoms are all deuterium ( or D, also known as ''heavy hydrogen'') rather than the common hydrogen-1 isotope (, also called ''protium'') that makes up most of the hydrogen in normal water. The presence of the heavier isotope gives the water different nuclear properties, and the increase in mass gives it slightly different physical and chemical properties when compared to normal water. Deuterium is a heavy Isotopes of hydrogen, hydrogen isotope. Heavy water contains deuterium atoms and is used in nuclear reactors. Semiheavy water (HDO) is more common than pure heavy water, while heavy-oxygen water is denser but lacks unique properties. Tritiated water is radioactive due to tritium content. Heavy water has different physical properties from regular water, such as being 10.6% denser and having a higher melting point. Heavy water is less Dissociation (chemistry), dissociated at a given temperatur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiazolium
Thiazole (), or 1,3-thiazole, is a 5-membered heterocyclic compound that contains both sulfur and nitrogen. The term 'thiazole' also refers to a large family of derivatives. Thiazole itself is a pale yellow liquid with a pyridine-like odor and the molecular formula C3H3NS. The thiazole ring is notable as a component of the vitamin thiamine (B1). Molecular and electronic structure Thiazoles are members of the azoles, heterocycles that include imidazoles and oxazoles. Thiazole can also be considered a functional group when part of a larger molecule. Being planar thiazoles are characterized by significant pi-electron delocalization and have some degree of aromaticity, more so than the corresponding oxazoles. This aromaticity is evidenced by the 1H NMR chemical shift of the ring protons, which absorb between 7.27 and 8.77 ppm, indicating a strong diamagnetic ring current. The calculated pi-electron density marks C5 as the primary site for electrophilic substitution, and C2-H ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |