Synapsid on:
[Wikipedia]
[Google]
[Amazon]
Synapsids + (, 'arch') > () "having a fused arch"; synonymous with ''theropsids'' (Greek, "beast-face") are one of the two major groups of animals that evolved from basal
 Synapsids evolved a temporal fenestra behind each eye
Synapsids evolved a temporal fenestra behind each eye
 Synapsids are characterized by having differentiated teeth. These include the
Synapsids are characterized by having differentiated teeth. These include the
 In addition to the glandular skin covered in fur found in most modern mammals, modern and extinct synapsids possess a variety of modified skin coverings, including
In addition to the glandular skin covered in fur found in most modern mammals, modern and extinct synapsids possess a variety of modified skin coverings, including
See also the news item at (see below, however). More primitive members of the Cynodontia are also hypothesized to have had fur or a fur-like covering based on their inferred warm-blooded metabolism. While more direct evidence of fur in early cynodonts has been proposed in the form of small pits on the snout possibly associated with
 '' Asaphestera'', '' Archaeothyris'' and ''
'' Asaphestera'', '' Archaeothyris'' and '' The therapsids, a more advanced group of synapsids, appeared during the
The therapsids, a more advanced group of synapsids, appeared during the 
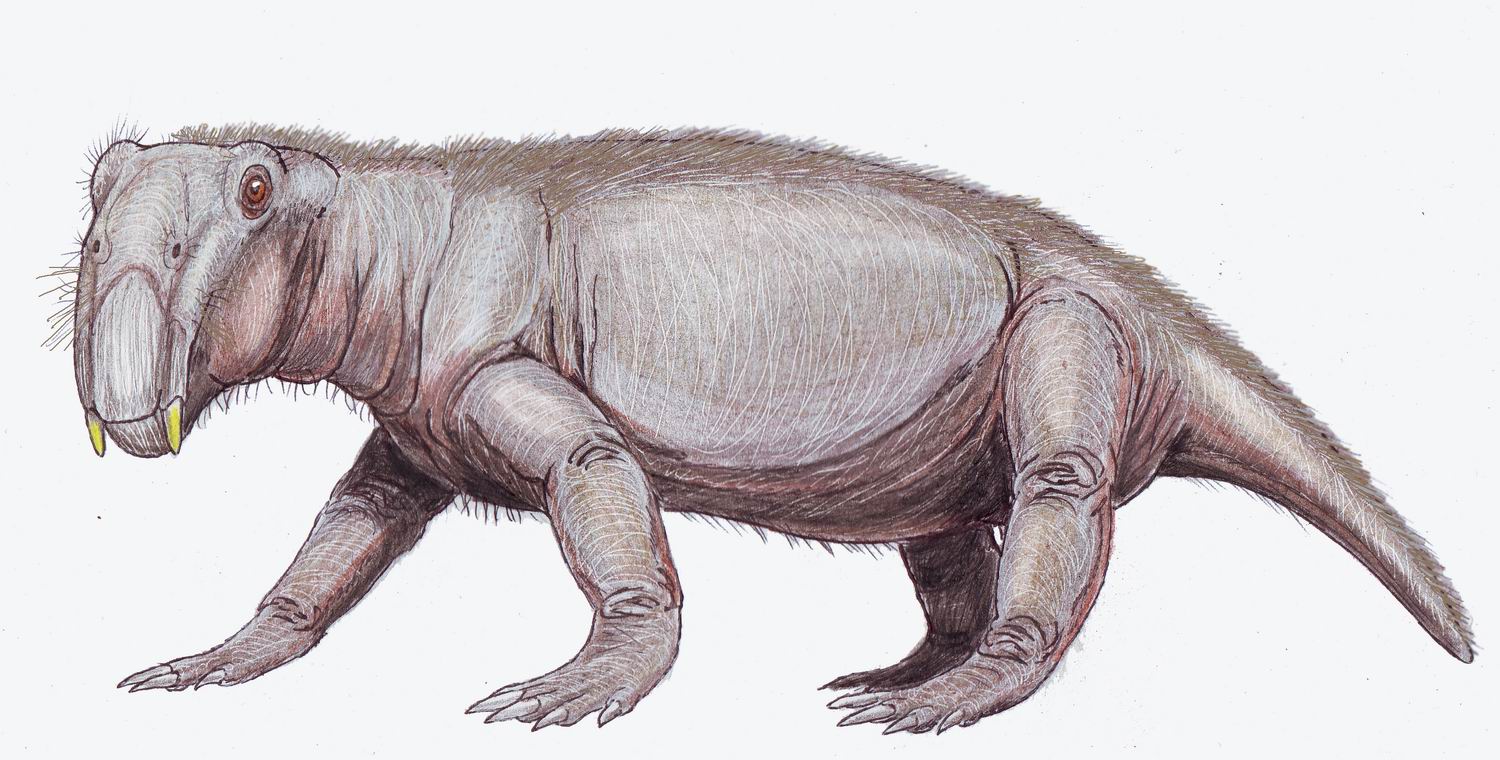 Only a few therapsids went on to be successful in the new early
Only a few therapsids went on to be successful in the new early  Unlike the dicynodonts, which were large, the cynodonts became progressively smaller and more mammal-like as the Triassic progressed, though some forms like '' Trucidocynodon'' remained large. The first mammaliaforms evolved from the cynodonts during the early
Unlike the dicynodonts, which were large, the cynodonts became progressively smaller and more mammal-like as the Triassic progressed, though some forms like '' Trucidocynodon'' remained large. The first mammaliaforms evolved from the cynodonts during the early  Whether through climate change, vegetation change, ecological competition, or a combination of factors, most of the remaining large cynodonts (belonging to the Traversodontidae) and dicynodonts (of the family Kannemeyeriidae) had disappeared by the
Whether through climate change, vegetation change, ecological competition, or a combination of factors, most of the remaining large cynodonts (belonging to the Traversodontidae) and dicynodonts (of the family Kannemeyeriidae) had disappeared by the 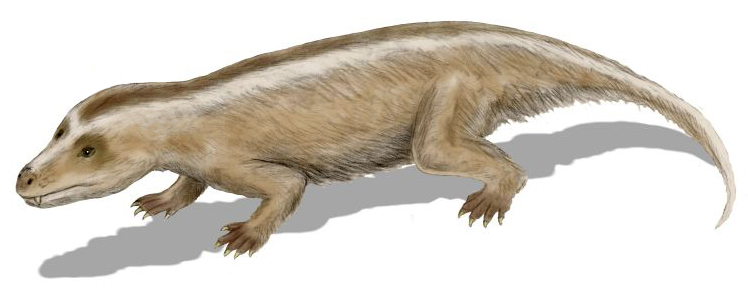 During the Jurassic and Cretaceous, the remaining non-mammalian cynodonts were small, such as ''
During the Jurassic and Cretaceous, the remaining non-mammalian cynodonts were small, such as ''
Synapsida - Pelycosauria
- at Palaeos
- includes description of important transitional genera in the evolutionary sequence linking primitive synapsids to mammals {{Authority control Tetrapod unranked clades Extant Pennsylvanian first appearances Taxa named by Henry Fairfield Osborn
amniote
Amniotes are a clade of tetrapod vertebrates that comprises sauropsids (including all reptiles and birds, and extinct parareptiles and non-avian dinosaurs) and synapsids (including pelycosaurs and therapsids such as mammals). They are dis ...
s, the other being the sauropsids
Sauropsida ("lizard faces") is a clade of amniotes, broadly equivalent to the class Reptilia. Sauropsida is the sister taxon to Synapsida, the other clade of amniotes which includes mammals as its only modern representatives. Although early syn ...
, the group that includes reptiles
Reptiles, as most commonly defined are the animals in the class Reptilia ( ), a paraphyletic grouping comprising all sauropsids except birds. Living reptiles comprise turtles, crocodilians, squamates ( lizards and snakes) and rhynchoceph ...
and birds. The group includes mammal
Mammals () are a group of vertebrate animals constituting the class Mammalia (), characterized by the presence of mammary glands which in females produce milk for feeding (nursing) their young, a neocortex (a region of the brain), fur ...
s and every animal more closely related to mammals than to sauropsids. Unlike other amniotes, synapsids have a single temporal fenestra, an opening low in the skull roof
The skull roof, or the roofing bones of the skull, are a set of bones covering the brain, eyes and nostrils in bony fishes and all land-living vertebrates. The bones are derived from dermal bone and are part of the dermatocranium.
In compar ...
behind each eye orbit
In celestial mechanics, an orbit is the curved trajectory of an object such as the trajectory of a planet around a star, or of a natural satellite around a planet, or of an artificial satellite around an object or position in space such as ...
, leaving a bony arch beneath each; this accounts for their name. The distinctive temporal fenestra developed about 318 million years ago during the Late Carboniferous
Late may refer to:
* LATE, an acronym which could stand for:
** Limbic-predominant age-related TDP-43 encephalopathy, a proposed form of dementia
** Local-authority trading enterprise, a New Zealand business law
** Local average treatment effe ...
period, when synapsids and sauropsids diverged, but was subsequently merged with the orbit in early mammals.
Traditionally, non-mammalian synapsids were believed to have evolved from reptiles, and therefore described as mammal-like reptiles in classical systematics, and primitive synapsids were also referred to as pelycosaur
Pelycosaur ( ) is an older term for basal or primitive Late Paleozoic synapsids, excluding the therapsids and their descendants. Previously, the term ''mammal-like reptile'' had been used, and pelycosaur was considered an order, but this is ...
s, or pelycosaur- grade synapsids. These paraphyletic
In taxonomy, a group is paraphyletic if it consists of the group's last common ancestor and most of its descendants, excluding a few monophyletic subgroups. The group is said to be paraphyletic ''with respect to'' the excluded subgroups. In ...
terms have now fallen into disfavor and are only used informally (if at all) in modern literature, because it is now known that synapsids are not reptiles, nor are they part of reptilian lineage in a cladistical
Cladistics (; ) is an approach to biological classification in which organisms are categorized in groups (" clades") based on hypotheses of most recent common ancestry. The evidence for hypothesized relationships is typically shared derived cha ...
sense. They are now more correctly referred to as stem mammals, and sometimes as proto-mammals, or paramammals.
Synapsids were the largest terrestrial vertebrates in the Permian
The Permian ( ) is a geologic period and stratigraphic system which spans 47 million years from the end of the Carboniferous Period million years ago (Mya), to the beginning of the Triassic Period 251.9 Mya. It is the last period of the Paleo ...
period, 299 to 251 million years ago, equalled only by some large pareiasaur
Pareiasaurs (meaning "cheek lizards") are an extinct clade of large, herbivorous parareptiles. Members of the group were armoured with scutes which covered large areas of the body. They first appeared in southern Pangea during the Middle Permi ...
s at the end of the Permian. Most lineages of pelycosaur-grade synapsids were replaced at the end of Early Permian by the more advanced therapsids
Therapsida is a major group of eupelycosaurian synapsids that includes mammals, their ancestors and relatives. Many of the traits today seen as unique to mammals had their origin within early therapsids, including limbs that were oriented mo ...
. Synapsid numbers and variety were severely reduced by the Permian–Triassic extinction. Only the therapsid dicynodont
Dicynodontia is an extinct clade of anomodonts, an extinct type of non-mammalian therapsid. Dicynodonts were herbivorous animals with a pair of tusks, hence their name, which means 'two dog tooth'. Members of the group possessed a horny, typic ...
s and eutheriodonts
Eutheriodontia is a clade of therapsids which appear during the Middle Permian and which includes therocephalians and cynodonts, this latter group including mammals and related forms.
With the dicynodonts, they form one of two lineages of thera ...
(consisting of Therocephalia
Therocephalia is an extinct suborder of eutheriodont therapsids (mammals and their close relatives) from the Permian and Triassic. The therocephalians ("beast-heads") are named after their large skulls, which, along with the structure of thei ...
and Cynodont
The cynodonts () (clade Cynodontia) are a clade of eutheriodont therapsids that first appeared in the Late Permian (approximately 260 mya), and extensively diversified after the Permian–Triassic extinction event. Cynodonts had a wide varie ...
ia) are known to have continued into the Triassic
The Triassic ( ) is a geologic period and system which spans 50.6 million years from the end of the Permian Period 251.902 million years ago ( Mya), to the beginning of the Jurassic Period 201.36 Mya. The Triassic is the first and shortest per ...
period. The cynodont group Probainognathia
Probainognathia is one of the two major subgroups of the clade Eucynodontia, the other being Cynognathia. The earliest forms were carnivorous and insectivorous, though some groups eventually also evolved herbivorous diets. The earliest and m ...
, which includes Mammaliaformes
Mammaliaformes ("mammalian forms") is a clade that contains the crown group mammals and their closest extinct relatives; the group radiated from earlier probainognathian cynodonts. It is defined as the clade originating from the most recent c ...
(mammals and their closer ancestors), were the only synapsids to survive beyond the Triassic.
During the Triassic, the sauropsid archosaur
Archosauria () is a clade of diapsids, with birds and crocodilians as the only living representatives. Archosaurs are broadly classified as reptiles, in the cladistic sense of the term which includes birds. Extinct archosaurs include non-avia ...
s became the largest and most numerous land vertebrates, and gave rise to the dinosaurs
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the evolution of dinosaurs is the ...
. When all non-avian dinosaurs were wiped out by the Cretaceous–Paleogene extinction event
The Cretaceous–Paleogene (K–Pg) extinction event (also known as the Cretaceous–Tertiary extinction) was a sudden mass extinction of three-quarters of the plant and animal species on Earth, approximately 66 million years ago. With the ...
, the mammalian synapsids diversified again to become the largest land and marine animals on Earth.
Linnaean and cladistic classifications
At the turn of the 20th century synapsids were thought to be one of the four main subclasses ofreptile
Reptiles, as most commonly defined are the animals in the class Reptilia ( ), a paraphyletic grouping comprising all sauropsids except birds. Living reptiles comprise turtles, crocodilians, squamates ( lizards and snakes) and rhynchocephalia ...
s. However, this notion was disproved upon closer inspection of skeletal remains, as synapsids are differentiated from reptiles by their distinctive temporal openings. These openings in the skull
The skull is a bone protective cavity for the brain. The skull is composed of four types of bone i.e., cranial bones, facial bones, ear ossicles and hyoid bone. However two parts are more prominent: the cranium and the mandible. In humans, th ...
bones allowed the attachment of larger jaw muscles, hence a more efficient bite.
Synapsids were subsequently considered to be a later reptilian lineage that became mammals by gradually evolving increasingly mammalian features, hence the name "mammal-like reptiles" (also known as pelycosaur
Pelycosaur ( ) is an older term for basal or primitive Late Paleozoic synapsids, excluding the therapsids and their descendants. Previously, the term ''mammal-like reptile'' had been used, and pelycosaur was considered an order, but this is ...
s). These became the traditional terms for all Paleozoic
The Paleozoic (or Palaeozoic) Era is the earliest of three geologic eras of the Phanerozoic Eon.
The name ''Paleozoic'' ( ;) was coined by the British geologist Adam Sedgwick in 1838
by combining the Greek words ''palaiós'' (, "old") and ...
(early) synapsids. More recent studies have debunked this notion as well, and reptiles are now classified within Sauropsida
Sauropsida ("lizard faces") is a clade of amniotes, broadly equivalent to the class Reptilia. Sauropsida is the sister taxon to Synapsida, the other clade of amniotes which includes mammals as its only modern representatives. Although early sy ...
(sauropsids), the sister group to synapsids, thus making synapsids their own taxonomic group.
As a result, the paraphyletic
In taxonomy, a group is paraphyletic if it consists of the group's last common ancestor and most of its descendants, excluding a few monophyletic subgroups. The group is said to be paraphyletic ''with respect to'' the excluded subgroups. In ...
terms "mammal-like reptile" and "pelycosaur" are seen as outdated and disfavored in technical literature, and the term stem mammal (or sometimes protomammal or paramammal) is used instead. Phylogenetically
In biology, phylogenetics (; from Greek φυλή/ φῦλον [] "tribe, clan, race", and wikt:γενετικός, γενετικός [] "origin, source, birth") is the study of the evolutionary history and relationships among or within groups ...
, it is now understood that synapsids comprise an independent branch of the tree of life
The tree of life is a fundamental archetype in many of the world's mythological, religious, and philosophical traditions. It is closely related to the concept of the sacred tree.Giovino, Mariana (2007). ''The Assyrian Sacred Tree: A Hist ...
. The monophyly
In cladistics for a group of organisms, monophyly is the condition of being a clade—that is, a group of taxa composed only of a common ancestor (or more precisely an ancestral population) and all of its lineal descendants. Monophyletic ...
of Synapsida is not in doubt, and the expressions such as "Synapsida contains the mammals" and "synapsids gave rise to the mammals" both express the same phylogenetic hypothesis. This terminology reflects the modern cladistical
Cladistics (; ) is an approach to biological classification in which organisms are categorized in groups (" clades") based on hypotheses of most recent common ancestry. The evidence for hypothesized relationships is typically shared derived cha ...
approach to animal relationships, according to which the only valid groups are those that include all of the descendants of a common ancestor: these are known as monophyletic
In cladistics for a group of organisms, monophyly is the condition of being a clade—that is, a group of taxa composed only of a common ancestor (or more precisely an ancestral population) and all of its lineal descendants. Monophyletic gr ...
groups, or clade
A clade (), also known as a monophyletic group or natural group, is a group of organisms that are monophyletic – that is, composed of a common ancestor and all its lineal descendants – on a phylogenetic tree. Rather than the English ter ...
s.
Additionally, Reptilia (reptiles) has been revised into a monophyletic group and is considered entirely distinct from Synapsida, falling within Sauropsida
Sauropsida ("lizard faces") is a clade of amniotes, broadly equivalent to the class Reptilia. Sauropsida is the sister taxon to Synapsida, the other clade of amniotes which includes mammals as its only modern representatives. Although early sy ...
, the sister group of Synapsida within Amniota.
Primitive and advanced synapsids
The synapsids are traditionally divided for convenience, intotherapsid
Therapsida is a major group of eupelycosaurian synapsids that includes mammals, their ancestors and relatives. Many of the traits today seen as unique to mammals had their origin within early therapsids, including limbs that were oriented more ...
s, an advanced group of synapsids and the branch within which mammals evolved, and stem mammals, (previously known as pelycosaur
Pelycosaur ( ) is an older term for basal or primitive Late Paleozoic synapsids, excluding the therapsids and their descendants. Previously, the term ''mammal-like reptile'' had been used, and pelycosaur was considered an order, but this is ...
s), comprising the other six more primitive families of synapsids. Stem mammals were all rather lizard-like, with sprawling gait and possibly horny scute
A scute or scutum (Latin: ''scutum''; plural: ''scuta'' "shield") is a bony external plate or scale overlaid with horn, as on the shell of a turtle, the skin of crocodilians, and the feet of birds. The term is also used to describe the anterior po ...
s, while therapsids tended to have a more erect pose and possibly hair, at least in some forms. In traditional taxonomy, the Synapsida encompasses two distinct grades: the low-slung stem mammals have given rise to the more erect therapsids, who in their turn have given rise to the mammals. In traditional vertebrate classification, the stem mammals and therapsids were both considered orders
Order, ORDER or Orders may refer to:
* Categorization, the process in which ideas and objects are recognized, differentiated, and understood
* Heterarchy, a system of organization wherein the elements have the potential to be ranked a number of ...
of the subclass Synapsida.Practical versus phylogenetic usage of “synapsid” and “therapsid”
Inphylogenetic nomenclature
Phylogenetic nomenclature is a method of nomenclature for taxa in biology that uses phylogenetic definitions for taxon names as explained below. This contrasts with the traditional approach, in which taxon names are defined by a '' type'', which ...
, the terms are used somewhat differently, as the daughter clades are included. Most papers published during the 21st century have treated "Pelycosaur" as an informal grouping of primitive members. Therapsida has remained in use as a clade containing both the traditional therapsid families and mammals.
Although Synapsida and Therapsida includes modern mammals, in practical usage, those two terms are used almost exclusively when referring to the more basal members that lie outside of Mammaliaformes
Mammaliaformes ("mammalian forms") is a clade that contains the crown group mammals and their closest extinct relatives; the group radiated from earlier probainognathian cynodonts. It is defined as the clade originating from the most recent c ...
.
Characteristics
Temporal openings
orbit
In celestial mechanics, an orbit is the curved trajectory of an object such as the trajectory of a planet around a star, or of a natural satellite around a planet, or of an artificial satellite around an object or position in space such as ...
on the lateral surface of the skull. It may have provided new attachment sites for jaw muscles. A similar development took place in the diapsid
Diapsids ("two arches") are a clade of sauropsids, distinguished from more primitive eureptiles by the presence of two holes, known as temporal fenestrae, in each side of their skulls. The group first appeared about three hundred million years a ...
s, which evolved two rather than one opening behind each eye. Originally, the openings in the skull left the inner cranium covered only by the jaw muscles, but in higher therapsids and mammals, the sphenoid bone
The sphenoid bone is an unpaired bone of the neurocranium. It is situated in the middle of the skull towards the front, in front of the basilar part of the occipital bone. The sphenoid bone is one of the seven bones that articulate to form the o ...
has expanded to close the opening. This has left the lower margin of the opening as an arch extending from the lower edges of the braincase.
Teeth
 Synapsids are characterized by having differentiated teeth. These include the
Synapsids are characterized by having differentiated teeth. These include the canine
Canine may refer to:
Zoology and anatomy
* a dog-like Canid animal in the subfamily Caninae
** ''Canis'', a genus including dogs, wolves, coyotes, and jackals
** Dog, the domestic dog
* Canine tooth, in mammalian oral anatomy
People with the surn ...
s, molars, and incisor
Incisors (from Latin ''incidere'', "to cut") are the front teeth present in most mammals. They are located in the premaxilla above and on the mandible below. Humans have a total of eight (two on each side, top and bottom). Opossums have 18, wher ...
s. The trend towards differentiation is found in some labyrinthodont
"Labyrinthodontia" (Greek, 'maze-toothed') is an informal grouping of extinct predatory amphibians which were major components of ecosystems in the late Paleozoic and early Mesozoic eras (about 390 to 150 million years ago). Traditionally conside ...
s and early anapsid
An anapsid is an amniote whose skull lacks one or more skull openings (fenestra, or fossae) near the temples. Traditionally, the Anapsida are the most primitive subclass of amniotes, the ancestral stock from which Synapsida and Diapsida evolv ...
reptilians in the form of enlargement of the first teeth on the maxilla
The maxilla (plural: ''maxillae'' ) in vertebrates is the upper fixed (not fixed in Neopterygii) bone of the jaw formed from the fusion of two maxillary bones. In humans, the upper jaw includes the hard palate in the front of the mouth. T ...
, forming a form of protocanines. This trait was subsequently lost in the diapsid
Diapsids ("two arches") are a clade of sauropsids, distinguished from more primitive eureptiles by the presence of two holes, known as temporal fenestrae, in each side of their skulls. The group first appeared about three hundred million years a ...
line, but developed further in the synapsids. Early synapsids could have two or even three enlarged "canines", but in the therapsids, the pattern had settled to one canine in each upper jaw half. The lower canines developed later.
Jaw
The jaw transition is a goodclassification Classification is a process related to categorization, the process in which ideas and objects are recognized, differentiated and understood.
Classification is the grouping of related facts into classes.
It may also refer to:
Business, organizat ...
tool, as most other fossilized features that make a chronological progression from a reptile-like to a mammalian condition follow the progression of the jaw transition. The mandible
In anatomy, the mandible, lower jaw or jawbone is the largest, strongest and lowest bone in the human facial skeleton. It forms the lower jaw and holds the lower teeth in place. The mandible sits beneath the maxilla. It is the only movable bone ...
, or lower jaw, consists of a single, tooth-bearing bone in mammals (the dentary), whereas the lower jaw of modern and prehistoric reptiles consists of a conglomeration of smaller bones (including the dentary, articular, and others). As they evolved in synapsids, these jaw bones were reduced in size and either lost or, in the case of the articular, gradually moved into the ear, forming one of the middle ear bones: while modern mammals possess the malleus, incus and stapes
The ''stapes'' or stirrup is a bone in the middle ear of humans and other animals which is involved in the conduction of sound vibrations to the inner ear. This bone is connected to the oval window by its annular ligament, which allows the foo ...
, basal synapsids (like all other tetrapods) possess only a stapes. The malleus is derived from the articular (a lower jaw bone), while the incus is derived from the quadrate (a cranial bone).Salentijn, L. ''Biology of Mineralized Tissues: Prenatal Skull Development'', Columbia University College of Dental Medicine post-graduate dental lecture series, 2007
Mammalian jaw structures are also set apart by the dentary-squamosal jaw joint
In anatomy, the temporomandibular joints (TMJ) are the two joints connecting the jawbone to the skull. It is a bilateral synovial articulation between the temporal bone of the skull above and the mandible below; it is from these bones that it ...
. In this form of jaw joint, the dentary forms a connection with a depression in the squamosal The squamosal is a skull bone found in most reptiles, amphibians, and birds. In fishes, it is also called the pterotic bone.
In most tetrapods, the squamosal and quadratojugal bones form the cheek series of the skull. The bone forms an ancestral co ...
known as the glenoid cavity. In contrast, all other jawed vertebrates, including reptiles and nonmammalian synapsids, possess a jaw joint in which one of the smaller bones of the lower jaw, the articular, makes a connection with a bone of the cranium
The skull is a bone protective cavity for the brain. The skull is composed of four types of bone i.e., cranial bones, facial bones, ear ossicles and hyoid bone. However two parts are more prominent: the cranium and the mandible. In humans, th ...
called the quadrate bone to form the articular-quadrate jaw joint. In forms transitional to mammals, the jaw joint is composed of a large, lower jaw bone (similar to the dentary found in mammals) that does not connect to the squamosal, but connects to the quadrate with a receding articular bone.
Palate
Over time, as synapsids became more mammalian and less 'reptilian', they began to develop asecondary palate
The secondary palate is an anatomical structure that divides the nasal cavity from the oral cavity in many vertebrates.
In human embryology, it refers to that portion of the hard palate that is formed by the growth of the two palatine shelves medi ...
, separating the mouth and nasal cavity
The nasal cavity is a large, air-filled space above and behind the nose in the middle of the face. The nasal septum divides the cavity into two cavities, also known as fossae. Each cavity is the continuation of one of the two nostrils. The nasal ...
. In early synapsids, a secondary palate began to form on the sides of the maxilla
The maxilla (plural: ''maxillae'' ) in vertebrates is the upper fixed (not fixed in Neopterygii) bone of the jaw formed from the fusion of two maxillary bones. In humans, the upper jaw includes the hard palate in the front of the mouth. T ...
, still leaving the mouth and nostril connected.
Eventually, the two sides of the palate began to curve together, forming a U shape instead of a C shape. The palate also began to extend back toward the throat, securing the entire mouth and creating a full palatine bone
In anatomy, the palatine bones () are two irregular bones of the facial skeleton in many animal species, located above the uvula in the throat. Together with the maxillae, they comprise the hard palate. (''Palate'' is derived from the Latin ...
. The maxilla is also closed completely. In fossils of one of the first eutheriodont
Eutheriodontia is a clade of therapsids which appear during the Middle Permian and which includes therocephalians and cynodonts, this latter group including mammals and related forms.
With the dicynodonts, they form one of two lineages of thera ...
s, the beginnings of a palate are clearly visible. The later '' Thrinaxodon'' has a full and completely closed palate, forming a clear progression.
Skin and fur
 In addition to the glandular skin covered in fur found in most modern mammals, modern and extinct synapsids possess a variety of modified skin coverings, including
In addition to the glandular skin covered in fur found in most modern mammals, modern and extinct synapsids possess a variety of modified skin coverings, including osteoderm
Osteoderms are bony deposits forming scales, plates, or other structures based in the dermis. Osteoderms are found in many groups of extant and extinct reptiles and amphibians, including lizards, crocodilians, frogs, temnospondyls (extinct amp ...
s (bony armor embedded in the skin), scute
A scute or scutum (Latin: ''scutum''; plural: ''scuta'' "shield") is a bony external plate or scale overlaid with horn, as on the shell of a turtle, the skin of crocodilians, and the feet of birds. The term is also used to describe the anterior po ...
s (protective structures of the dermis often with a horny covering), hair or fur, and scale-like structures (often formed from modified hair, as in pangolin
Pangolins, sometimes known as scaly anteaters, are mammals of the order Pholidota (, from Ancient Greek ϕολιδωτός – "clad in scales"). The one extant family, the Manidae, has three genera: '' Manis'', ''Phataginus'', and '' Smuts ...
s and some rodent
Rodents (from Latin , 'to gnaw') are mammals of the order Rodentia (), which are characterized by a single pair of continuously growing incisors in each of the upper and lower jaws. About 40% of all mammal species are rodents. They are n ...
s). While the skin of reptiles is rather thin, that of mammals has a thick dermal layer.
The ancestral skin type of synapsids has been subject to discussion. Among the early synapsids, only two species of small varanopids have been found to possess scute
A scute or scutum (Latin: ''scutum''; plural: ''scuta'' "shield") is a bony external plate or scale overlaid with horn, as on the shell of a turtle, the skin of crocodilians, and the feet of birds. The term is also used to describe the anterior po ...
s; fossilized rows of osteoderm
Osteoderms are bony deposits forming scales, plates, or other structures based in the dermis. Osteoderms are found in many groups of extant and extinct reptiles and amphibians, including lizards, crocodilians, frogs, temnospondyls (extinct amp ...
s indicate horny armour on the neck and back, and skin impressions indicate some possessed rectangular scutes on their undersides and tails. The pelycosaur scutes probably were nonoverlapping dermal structures with a horny overlay, like those found in modern crocodile
Crocodiles (family Crocodylidae) or true crocodiles are large semiaquatic reptiles that live throughout the tropics in Africa, Asia, the Americas and Australia. The term crocodile is sometimes used even more loosely to include all extant me ...
s and turtle
Turtles are an order of reptiles known as Testudines, characterized by a special shell developed mainly from their ribs. Modern turtles are divided into two major groups, the Pleurodira (side necked turtles) and Cryptodira (hidden necked t ...
s. These differed in structure from the scales of lizards and snakes, which are an epidermal feature (like mammalian hair or avian feathers). Recently, skin impressions from the genus '' Ascendonanus'' suggest that at least varanopsids developed scales similar to those of squamates.
It is currently unknown exactly when mammalian characteristics such as body hair
Body hair, or androgenic hair, is the terminal hair that develops on the human body during and after puberty. It is differentiated from the head hair and less visible vellus hair, which is much finer and lighter in color. The growth of androge ...
and mammary gland
A mammary gland is an exocrine gland in humans and other mammals that produces milk to feed young offspring. Mammals get their name from the Latin word ''mamma'', "breast". The mammary glands are arranged in organs such as the breasts in ...
s first appeared, as the fossils only rarely provide direct evidence for soft tissues. An exceptionally well-preserved skull of ''Estemmenosuchus
''Estemmenosuchus'' (meaning "crowned crocodile" in Greek) is an extinct genus of large, early omnivorous therapsid. It is believed and interpreted to have lived during the middle part of the Middle Permian around 267 million years ago. The two s ...
'', a therapsid from the Upper Permian, preserves smooth skin with what appear to be glandular depressions, an animal noted as being semi- aquatic. The oldest known fossil showing unambiguous imprints of hair is the Callovian (late middle Jurassic
The Jurassic ( ) is a geologic period and stratigraphic system that spanned from the end of the Triassic Period million years ago (Mya) to the beginning of the Cretaceous Period, approximately Mya. The Jurassic constitutes the middle period of ...
) ''Castorocauda
''Castorocauda'' is an extinct, semi-aquatic, superficially otter-like genus of docodont mammaliaforms with one species, ''C. lutrasimilis''. It is part of the Yanliao Biota, found in the Daohugou Beds of Inner Mongolia, China dating to the M ...
'' and several contemporary haramiyida
Haramiyida ("thief" from Arabic الحرامية (al ḥarāmiyah), "thief, bandit") is a possibly polyphyletic order of mammaliaform cynodonts or mammals of controversial taxonomic affinites. Their teeth, which are by far the most common remai ...
ns, both non-mammalian mammaliaformSee also the news item at (see below, however). More primitive members of the Cynodontia are also hypothesized to have had fur or a fur-like covering based on their inferred warm-blooded metabolism. While more direct evidence of fur in early cynodonts has been proposed in the form of small pits on the snout possibly associated with
whisker
Vibrissae (; singular: vibrissa; ), more generally called Whiskers, are a type of stiff, functional hair used by mammals to sense their environment. These hairs are finely specialised for this purpose, whereas other types of hair are coars ...
s, such pits are also found in some reptiles that lack whiskers. There is evidence that some other non-mammalian cynodonts more basal than ''Castorocauda'', such as ''Morganucodon
''Morganucodon'' ("Glamorgan tooth") is an early mammaliaform genus that lived from the Late Triassic to the Middle Jurassic. It first appeared about 205 million years ago. Unlike many other early mammaliaforms, ''Morganucodon'' is well represent ...
'', had Harderian glands
The Harderian gland is a gland found within the eye's orbit that occurs in tetrapods (reptiles, amphibians, birds and mammals) that possess a nictitating membrane.
The gland can be compound tubular or compound tubuloalveolar, and the fluid it s ...
, which are associated with the grooming and maintenance of fur. The apparent absence of these glands in non-mammaliaformes may suggest that fur did not originate until that point in synapsid evolution. It is possible that fur and associated features of true warm-bloodedness did not appear until some synapsids became extremely small and nocturnal, necessitating a higher metabolism. The oldest examples of nocturnality in synapsids is believed to have been in species that lived more than 300 million years ago.
However, Permian
The Permian ( ) is a geologic period and stratigraphic system which spans 47 million years from the end of the Carboniferous Period million years ago (Mya), to the beginning of the Triassic Period 251.9 Mya. It is the last period of the Paleo ...
coprolite
A coprolite (also known as a coprolith) is fossilized feces. Coprolites are classified as trace fossils as opposed to body fossils, as they give evidence for the animal's behaviour (in this case, diet) rather than morphology. The name is ...
s from Russia showcase that at least some synapsids did already have fur in this epoch. These are the oldest impressions of hair on synapsids.
Mammary glands
Early synapsids, as far back as their known evolutionary debut in the Late Carboniferous period, may have laid parchment-shelled (leathery) eggs, which lacked a calcified layer, as most modern reptiles andmonotreme
Monotremes () are prototherian mammals of the order Monotremata. They are one of the three groups of living mammals, along with placentals ( Eutheria), and marsupials (Metatheria). Monotremes are typified by structural differences in their brai ...
s do. This may also explain why there is no fossil evidence for synapsid eggs to date. Because they were vulnerable to desiccation, secretions from apocrine-like glands may have helped keep the eggs moist.
According to Oftedal, early synapsids may have buried the eggs into moisture laden soil, hydrating them with contact with the moist skin, or may have carried them in a moist pouch, similar to that of monotremes (echidna
Echidnas (), sometimes known as spiny anteaters, are quill-covered monotremes (egg-laying mammals) belonging to the family Tachyglossidae . The four extant species of echidnas and the platypus are the only living mammals that lay eggs and the ...
s carry their eggs and offspring via a temporary pouch), though this would limit the mobility of the parent. The latter may have been the primitive form of egg care in synapsids rather than simply burying the eggs, and the constraint on the parent's mobility would have been solved by having the eggs "parked" in nests during foraging or other activities and periodically be hydrated, allowing higher clutch sizes than could fit inside a pouch (or pouches) at once, and large eggs, which would be cumbersome to carry in a pouch, would be easier to care for. The basis of Oftedal's speculation is the fact that many species of anurans can carry eggs or tadpoles attached to the skin, or embedded within cutaneous "pouches" and how most salamander
Salamanders are a group of amphibians typically characterized by their lizard-like appearance, with slender bodies, blunt snouts, short limbs projecting at right angles to the body, and the presence of a tail in both larvae and adults. All t ...
s curl around their eggs to keep them moist, both groups also having glandular skin.
The glands involved in this mechanism would later evolve into true mammary glands with multiple modes of secretion in association with hair follicles. Comparative analyses of the evolutionary origin of milk constituents support a scenario in which the secretions from these glands evolved into a complex, nutrient-rich milk long before true mammals arose (with some of the constituents possibly predating the split between the synapsid and sauropsid
Sauropsida ("lizard faces") is a clade of amniotes, broadly equivalent to the class Reptilia. Sauropsida is the sister taxon to Synapsida, the other clade of amniotes which includes mammals as its only modern representatives. Although early syn ...
lines). Cynodont
The cynodonts () (clade Cynodontia) are a clade of eutheriodont therapsids that first appeared in the Late Permian (approximately 260 mya), and extensively diversified after the Permian–Triassic extinction event. Cynodonts had a wide varie ...
s were almost certainly able to produce this, which allowed a progressive decline of yolk mass and thus egg size, resulting in increasingly altricial
In biology, altricial species are those in which the young are underdeveloped at the time of birth, but with the aid of their parents mature after birth. Precocial species are those in which the young are relatively mature and mobile from the mome ...
hatchlings as milk became the primary source of nutrition, which is all evidenced by the small body size, the presence of epipubic bones, and limited tooth replacement in advanced cynodonts, as well as in mammaliaforms.
Patagia
Aerial locomotion first began in non-mammalianharamiyida
Haramiyida ("thief" from Arabic الحرامية (al ḥarāmiyah), "thief, bandit") is a possibly polyphyletic order of mammaliaform cynodonts or mammals of controversial taxonomic affinites. Their teeth, which are by far the most common remai ...
n cynodonts, with ''Arboroharamiya
''Arboroharamiya'' ("tree thief" from Latin arbor, "tree" + Arabic الحرامية (al ḥarāmiyah), "thief, '' Haramiya''") is an extinct genus of early mammal (or possibly a non-mammalian mammaliaform) from the Middle Jurassic Tiaojishan Form ...
'', '' Xianshou'', '' Maiopatagium'' and '' Vilevolodon'' bearing exquisitely preserved, fur-covered wing membranes that stretch across the limbs and tail. Their fingers are elongated, similar to those of bats and colugos and likely sharing similar roles both as wing supports and to hang on tree branches.
Within true mammals, aerial locomotion first occurs in volaticotherian eutriconodonts. A fossil '' Volaticotherium'' has an exquisitely preserved furry patagium
The patagium (plural: patagia) is a membranous body part that assists an animal in obtaining lift when gliding or flight. The structure is found in extant and extinct groups of flying and gliding animals including bats, birds, some dromaeos ...
with delicate wrinkles and that is very extensive, "sandwiching" the poorly preserved hands and feet and extending to the base of the tail. '' Argentoconodon'', a close relative, shares a similar femur adapted for flight stresses, indicating a similar lifestyle.
Theria
Theria (; Greek: , wild beast) is a subclass of mammals amongst the Theriiformes. Theria includes the eutherians (including the placental mammals) and the metatherians (including the marsupials) but excludes the egg-laying monotremes.
...
n mammals would only achieve powered flight and gliding long after these early aeronauts became extinct, with the earliest-known gliding metatheria
Metatheria is a mammalian clade that includes all mammals more closely related to marsupials than to placentals. First proposed by Thomas Henry Huxley in 1880, it is a more inclusive group than the marsupials; it contains all marsupials as w ...
ns and bats evolving in the Paleocene
The Paleocene, ( ) or Palaeocene, is a geological epoch that lasted from about 66 to 56 million years ago (mya). It is the first epoch of the Paleogene Period in the modern Cenozoic Era. The name is a combination of the Ancient Greek ''pala ...
.
Metabolism
Recently, it has been found that endothermy was developed as early as '' Ophiacodon'' in the late Carboniferous. The presence of fibrolamellar, a specialised type of bone that can grow quickly while maintaining a stable structure, shows that Ophiacodon would have used its high internal body temperature to fuel a fast growth comparable to modern endotherms.Evolutionary history
 '' Asaphestera'', '' Archaeothyris'' and ''
'' Asaphestera'', '' Archaeothyris'' and ''Clepsydrops
''Clepsydrops'' is an extinct genus of primitive synapsids from the early Late Carboniferous that was related to ''Archaeothyris''. The name means 'hour-glass appearance' (Greek ''klepsydra'' = "hourglass" + Greek ''ops'' = "eye, face, appearan ...
'', the earliest-known synapsids, lived in the Pennsylvanian subperiod (323–299 mya) of the Carboniferous
The Carboniferous ( ) is a geologic period and system of the Paleozoic that spans 60 million years from the end of the Devonian Period million years ago ( Mya), to the beginning of the Permian Period, million years ago. The name ''Carboniferou ...
period and were one of many types of primitive synapsids that are now informally grouped together as stem mammals or sometimes as protomammals (previously known as pelycosaur
Pelycosaur ( ) is an older term for basal or primitive Late Paleozoic synapsids, excluding the therapsids and their descendants. Previously, the term ''mammal-like reptile'' had been used, and pelycosaur was considered an order, but this is ...
s). The early synapsids spread and diversified, becoming the largest terrestrial animals in the latest Carboniferous and Early Permian 01 or '01 may refer to:
* The year 2001, or any year ending with 01
* The month of January
* 1 (number)
Music
* 01'' (Richard Müller album), 2001
* ''01'' (Son of Dave album), 2000
* ''01'' (Urban Zakapa album), 2011
* ''O1'' (Hiroyuki Sawan ...
periods, ranging up to in length. They were sprawling, bulky, possibly cold-blooded, and had small brains. Some, such as ''Dimetrodon'', had large sails that might have helped raise their body temperature. A few relict groups lasted into the later Permian but, by the middle of the Late Permian, all had either died off or evolved into their successors, the therapsids.
 The therapsids, a more advanced group of synapsids, appeared during the
The therapsids, a more advanced group of synapsids, appeared during the Middle Permian
The Guadalupian is the second and middle series/ epoch of the Permian. The Guadalupian was preceded by the Cisuralian and followed by the Lopingian. It is named after the Guadalupe Mountains of New Mexico and Texas, and dates between 272.95 ± ...
and included the largest terrestrial animals in the Middle and Late Permian
Late may refer to:
* LATE, an acronym which could stand for:
** Limbic-predominant age-related TDP-43 encephalopathy, a proposed form of dementia
** Local-authority trading enterprise, a New Zealand business law
** Local average treatment effect, ...
. They included herbivores and carnivores, ranging from small animals the size of a rat (e.g.: '' Robertia''), to large, bulky herbivores a ton or more in weight (e.g.: '' Moschops''). After flourishing for many millions of years, these successful animals were all but wiped out by the Permian–Triassic mass extinction about 250 mya, the largest known extinction
Extinction is the termination of a kind of organism or of a group of kinds (taxon), usually a species. The moment of extinction is generally considered to be the death of the Endling, last individual of the species, although the Functional ext ...
in Earth's history, possibly related to the Siberian Traps volcanic event.

 Only a few therapsids went on to be successful in the new early
Only a few therapsids went on to be successful in the new early Triassic
The Triassic ( ) is a geologic period and system which spans 50.6 million years from the end of the Permian Period 251.902 million years ago ( Mya), to the beginning of the Jurassic Period 201.36 Mya. The Triassic is the first and shortest per ...
landscape; they include '' Lystrosaurus'' and '' Cynognathus'', the latter of which appeared later in the early Triassic. However, they were accompanied by the early archosaur
Archosauria () is a clade of diapsids, with birds and crocodilians as the only living representatives. Archosaurs are broadly classified as reptiles, in the cladistic sense of the term which includes birds. Extinct archosaurs include non-avia ...
s (soon to give rise to the dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the evolution of dinosaurs is t ...
s). Some of these archosaurs, such as '' Euparkeria'', were small and lightly built, while others, such as ''Erythrosuchus
''Erythrosuchus'' (from el, ἐρυθρός , 'red' and el, σοῦχος , 'crocodile') is an extinct genus of archosauriform reptile from the Triassic of South Africa. Remains have been found from the Cynognathus Assemblage Zone of the Beauf ...
'', were as big as or bigger than the largest therapsids.
After the Permian extinction, the synapsids did not count more than three surviving clades. The first comprised the therocephalia
Therocephalia is an extinct suborder of eutheriodont therapsids (mammals and their close relatives) from the Permian and Triassic. The therocephalians ("beast-heads") are named after their large skulls, which, along with the structure of thei ...
ns, which only lasted the first 20 million years of the Triassic period. The second were specialised, beaked herbivores known as dicynodont
Dicynodontia is an extinct clade of anomodonts, an extinct type of non-mammalian therapsid. Dicynodonts were herbivorous animals with a pair of tusks, hence their name, which means 'two dog tooth'. Members of the group possessed a horny, typic ...
s (such as the Kannemeyeriidae), which contained some members that reached large size (up to a tonne or more). And finally there were the increasingly mammal-like carnivorous, herbivorous, and insectivorous cynodonts, including the eucynodonts
Eucynodontia ("true dog teeth") is a clade of cynodont therapsids including mammals and most non-mammalian cynodonts. The oldest eucynodonts are known from the Early Triassic and possibly Late Permian. Eucynodontia includes two major subgroups, ...
from the Olenekian age, an early representative of which was ''Cynognathus''.
 Unlike the dicynodonts, which were large, the cynodonts became progressively smaller and more mammal-like as the Triassic progressed, though some forms like '' Trucidocynodon'' remained large. The first mammaliaforms evolved from the cynodonts during the early
Unlike the dicynodonts, which were large, the cynodonts became progressively smaller and more mammal-like as the Triassic progressed, though some forms like '' Trucidocynodon'' remained large. The first mammaliaforms evolved from the cynodonts during the early Norian
The Norian is a division of the Triassic Period. It has the rank of an age ( geochronology) or stage (chronostratigraphy). It lasted from ~227 to million years ago. It was preceded by the Carnian and succeeded by the Rhaetian.
Stratigraphic ...
age of the Late Triassic, about 225 mya.
During the evolutionary succession from early therapsid to cynodont to eucynodont to mammal, the main lower jaw bone, the dentary, replaced the adjacent bones. Thus, the lower jaw gradually became just one large bone, with several of the smaller jaw bones migrating into the inner ear
The inner ear (internal ear, auris interna) is the innermost part of the vertebrate ear. In vertebrates, the inner ear is mainly responsible for sound detection and balance. In mammals, it consists of the bony labyrinth, a hollow cavity in th ...
and allowing sophisticated hearing.
 Whether through climate change, vegetation change, ecological competition, or a combination of factors, most of the remaining large cynodonts (belonging to the Traversodontidae) and dicynodonts (of the family Kannemeyeriidae) had disappeared by the
Whether through climate change, vegetation change, ecological competition, or a combination of factors, most of the remaining large cynodonts (belonging to the Traversodontidae) and dicynodonts (of the family Kannemeyeriidae) had disappeared by the Rhaetian
The Rhaetian is the latest age of the Triassic Period (in geochronology) or the uppermost stage of the Triassic System (in chronostratigraphy). It was preceded by the Norian and succeeded by the Hettangian (the lowermost stage or earliest a ...
age, even before the Triassic–Jurassic extinction event that killed off most of the large non-dinosaurian archosaurs. The remaining Mesozoic synapsids were small, ranging from the size of a shrew to the badger-like mammal '' Repenomamus''.
 During the Jurassic and Cretaceous, the remaining non-mammalian cynodonts were small, such as ''
During the Jurassic and Cretaceous, the remaining non-mammalian cynodonts were small, such as ''Tritylodon
''Tritylodon'' (Greek for 3 cusped tooth) is an extinct genus of tritylodonts, one of the most advanced group of cynodont therapsids. They lived in the Early Jurassic and possibly Late Triassic periods along with dinosaurs. They also shared man ...
''. No cynodont grew larger than a cat. Most Jurassic and Cretaceous cynodonts were herbivorous
A herbivore is an animal anatomically and physiologically adapted to eating plant material, for example foliage or marine algae, for the main component of its diet. As a result of their plant diet, herbivorous animals typically have mouthpar ...
, though some were carnivorous
A carnivore , or meat-eater (Latin, ''caro'', genitive ''carnis'', meaning meat or "flesh" and ''vorare'' meaning "to devour"), is an animal or plant whose food and energy requirements derive from animal tissues (mainly muscle, fat and other ...
. The family Tritheledontidae, which first appeared near the end of the Triassic, was carnivorous and persisted well into the Middle Jurassic
The Middle Jurassic is the second Epoch (geology), epoch of the Jurassic Period (geology), Period. It lasted from about 174.1 to 163.5 million years ago. Fossils of land-dwelling animals, such as dinosaurs, from the Middle Jurassic are relatively ...
. The other, Tritylodontidae
Tritylodontidae ("three-knob teeth", named after the shape of their cheek teeth) is an extinct family of small to medium-sized, highly specialized mammal-like cynodonts, bearing several mammalian traits like erect limbs, endothermy and details ...
, first appeared at the same time as the tritheledonts, but was herbivorous. This group became extinct at the end of the Early Cretaceous epoch. Dicynodonts are thought to have become extinct near the end of the Triassic period, but there is evidence this group survived. New fossil finds have been found in the Cretaceous rocks of Gondwana
Gondwana () was a large landmass, often referred to as a supercontinent, that formed during the late Neoproterozoic (about 550 million years ago) and began to break up during the Jurassic period (about 180 million years ago). The final sta ...
.
Today, the 5,500 species of living synapsids, known as the mammal
Mammals () are a group of vertebrate animals constituting the class Mammalia (), characterized by the presence of mammary glands which in females produce milk for feeding (nursing) their young, a neocortex (a region of the brain), fur ...
s, include both aquatic (whales
Whales are a widely distributed and diverse group of fully aquatic placental marine mammals. As an informal and colloquial grouping, they correspond to large members of the infraorder Cetacea, i.e. all cetaceans apart from dolphins and ...
) and flying ( bats) species, and the largest animal ever known to have existed (the blue whale
The blue whale (''Balaenoptera musculus'') is a marine mammal and a baleen whale. Reaching a maximum confirmed length of and weighing up to , it is the largest animal known to have ever existed. The blue whale's long and slender body can ...
). Humans are synapsids, as well. Most mammals are viviparous
Among animals, viviparity is development of the embryo inside the body of the parent. This is opposed to oviparity which is a reproductive mode in which females lay developing eggs that complete their development and hatch externally from the ...
and give birth to live young rather than laying eggs with the exception being the monotreme
Monotremes () are prototherian mammals of the order Monotremata. They are one of the three groups of living mammals, along with placentals ( Eutheria), and marsupials (Metatheria). Monotremes are typified by structural differences in their brai ...
s.
Triassic and Jurassic ancestors of living mammals, along with their close relatives, had high metabolic rates. This meant consuming food (generally thought to be insects) in much greater quantity. To facilitate rapid digestion
Digestion is the breakdown of large insoluble food molecules into small water-soluble food molecules so that they can be absorbed into the watery blood plasma. In certain organisms, these smaller substances are absorbed through the small intest ...
, these synapsids evolved mastication
Chewing or mastication is the process by which food is crushed and ground by teeth. It is the first step of digestion, and it increases the surface area of foods to allow a more efficient break down by enzymes. During the mastication process, ...
(chewing) and specialized teeth that aided chewing. Limbs also evolved to move under the body instead of to the side, allowing them to breathe more efficiently during locomotion.
This helped make it possible to support their higher metabolic demands.
Relationships
Below is acladogram
A cladogram (from Greek ''clados'' "branch" and ''gramma'' "character") is a diagram used in cladistics to show relations among organisms. A cladogram is not, however, an evolutionary tree because it does not show how ancestors are related to ...
of the most commonly accepted phylogeny
A phylogenetic tree (also phylogeny or evolutionary tree Felsenstein J. (2004). ''Inferring Phylogenies'' Sinauer Associates: Sunderland, MA.) is a branching diagram or a tree showing the evolutionary relationships among various biological s ...
of synapsids, showing a long stem lineage including Mammalia and successively more basal clades such as Theriodontia, Therapsida and Sphenacodontia:
Most uncertainty in the phylogeny of synapsids lies among the earliest members of the group, including forms traditionally placed within Pelycosauria. As one of the earliest phylogenetic analyses, Brinkman & Eberth (1983) placed the family Varanopidae
Varanopidae is an extinct family of amniotes that resembled monitor lizards and may have filled a similar niche, hence the name. Typically, they are considered synapsids that evolved from an '' Archaeothyris''-like synapsid in the Late Carbonife ...
with Caseasauria
Caseasauria is one of the two main clades of early synapsids, the other being the Eupelycosauria. Caseasaurs are currently known only from the Late Carboniferous and the Permian, and include two superficially different families, the small insec ...
as the most basal offshoot of the synapsid lineage. Reisz (1986) removed Varanopidae from Caseasauria, placing it in a more derived position on the stem. While most analyses find Caseasauria to be the most basal synapsid clade, Benson's analysis (2012) placed a clade containing Ophiacodontidae and Varanopidae as the most basal synapsids, with Caseasauria occupying a more derived position. Benson attributed this revised phylogeny to the inclusion of postcranial characteristics, or features of the skeleton other than the skull, in his analysis. When only cranial or skull features were included, Caseasauria remained the most basal synapsid clade. Below is a cladogram
A cladogram (from Greek ''clados'' "branch" and ''gramma'' "character") is a diagram used in cladistics to show relations among organisms. A cladogram is not, however, an evolutionary tree because it does not show how ancestors are related to ...
modified from Benson's analysis (2012):
However, more recent examination of the phylogeny of basal synapsids, incorporating newly described basal caseids and eothyridids, returned Caseasauria to its position as the sister to all other synapsids. Brocklehurst et al. (2016) demonstrated that many of the postcranial characters used by Benson (2012) to unite Caseasauria with Sphenacodontidae and Edaphosauridae were absent in the newly discovered postcranial material of eothyridids, and were therefore acquired convergently.
See also
*Anapsid
An anapsid is an amniote whose skull lacks one or more skull openings (fenestra, or fossae) near the temples. Traditionally, the Anapsida are the most primitive subclass of amniotes, the ancestral stock from which Synapsida and Diapsida evolv ...
* Diapsid
Diapsids ("two arches") are a clade of sauropsids, distinguished from more primitive eureptiles by the presence of two holes, known as temporal fenestrae, in each side of their skulls. The group first appeared about three hundred million years a ...
* Euryapsida
* Lists of synapsids
These lists of synapsids collectively include every genus that has ever been included in the clade Synapsida- the mammals and their evolutionary precursors. The lists includes accepted genera along with those now considered invalid, doubtful (''nom ...
* Mammal classification
* List of prehistoric mammals
* Timeline of the evolutionary history of life
* Vertebrate paleontology
Vertebrate paleontology is the subfield of paleontology that seeks to discover, through the study of fossilized remains, the behavior, reproduction and appearance of extinct animals with vertebrae or a notochord. It also tries to connect, by us ...
Notes
References
Further reading
*External links
Synapsida - Pelycosauria
- at Palaeos
- includes description of important transitional genera in the evolutionary sequence linking primitive synapsids to mammals {{Authority control Tetrapod unranked clades Extant Pennsylvanian first appearances Taxa named by Henry Fairfield Osborn