Parkinson's disease on:
[Wikipedia]
[Google]
[Amazon]
Parkinson's disease (PD), or simply Parkinson's, is a long-term degenerative disorder of the
 The most recognizable symptoms are movement (motor) related, and include tremor,
The most recognizable symptoms are movement (motor) related, and include tremor,
 Research indicates that PD is the product of a complex interaction of genetic and
Research indicates that PD is the product of a complex interaction of genetic and
 The main
The main  Five major pathways in the brain connect other brain areas with the basal ganglia. These are known as the
Five major pathways in the brain connect other brain areas with the basal ganglia. These are known as the  A particular conceptual model of the motor circuit and its alteration with PD has been of great influence since 1980, although some limitations have been pointed out which have led to modifications. In this model, the basal ganglia normally exert a constant inhibitory influence on a wide range of motor systems, preventing them from becoming active at inappropriate times. When a decision is made to perform a particular action, inhibition is reduced for the required motor system, thereby releasing it for activation. Dopamine acts to facilitate this release of inhibition, so high levels of dopamine function tend to promote motor activity, while low levels of dopamine function, such as occur in PD, demand greater exertions of effort for any given movement. Thus, the net effect of dopamine depletion is to produce hypokinesia, an overall reduction in motor output. Drugs that are used to treat PD, conversely, may produce excessive dopamine activity, allowing motor systems to be activated at inappropriate times and thereby producing
A particular conceptual model of the motor circuit and its alteration with PD has been of great influence since 1980, although some limitations have been pointed out which have led to modifications. In this model, the basal ganglia normally exert a constant inhibitory influence on a wide range of motor systems, preventing them from becoming active at inappropriate times. When a decision is made to perform a particular action, inhibition is reduced for the required motor system, thereby releasing it for activation. Dopamine acts to facilitate this release of inhibition, so high levels of dopamine function tend to promote motor activity, while low levels of dopamine function, such as occur in PD, demand greater exertions of effort for any given movement. Thus, the net effect of dopamine depletion is to produce hypokinesia, an overall reduction in motor output. Drugs that are used to treat PD, conversely, may produce excessive dopamine activity, allowing motor systems to be activated at inappropriate times and thereby producing
 Parkinson-plus syndrome – Multiple diseases can be considered part of the Parkinson's plus group, including corticobasal syndrome, multiple system atrophy,
Parkinson-plus syndrome – Multiple diseases can be considered part of the Parkinson's plus group, including corticobasal syndrome, multiple system atrophy,
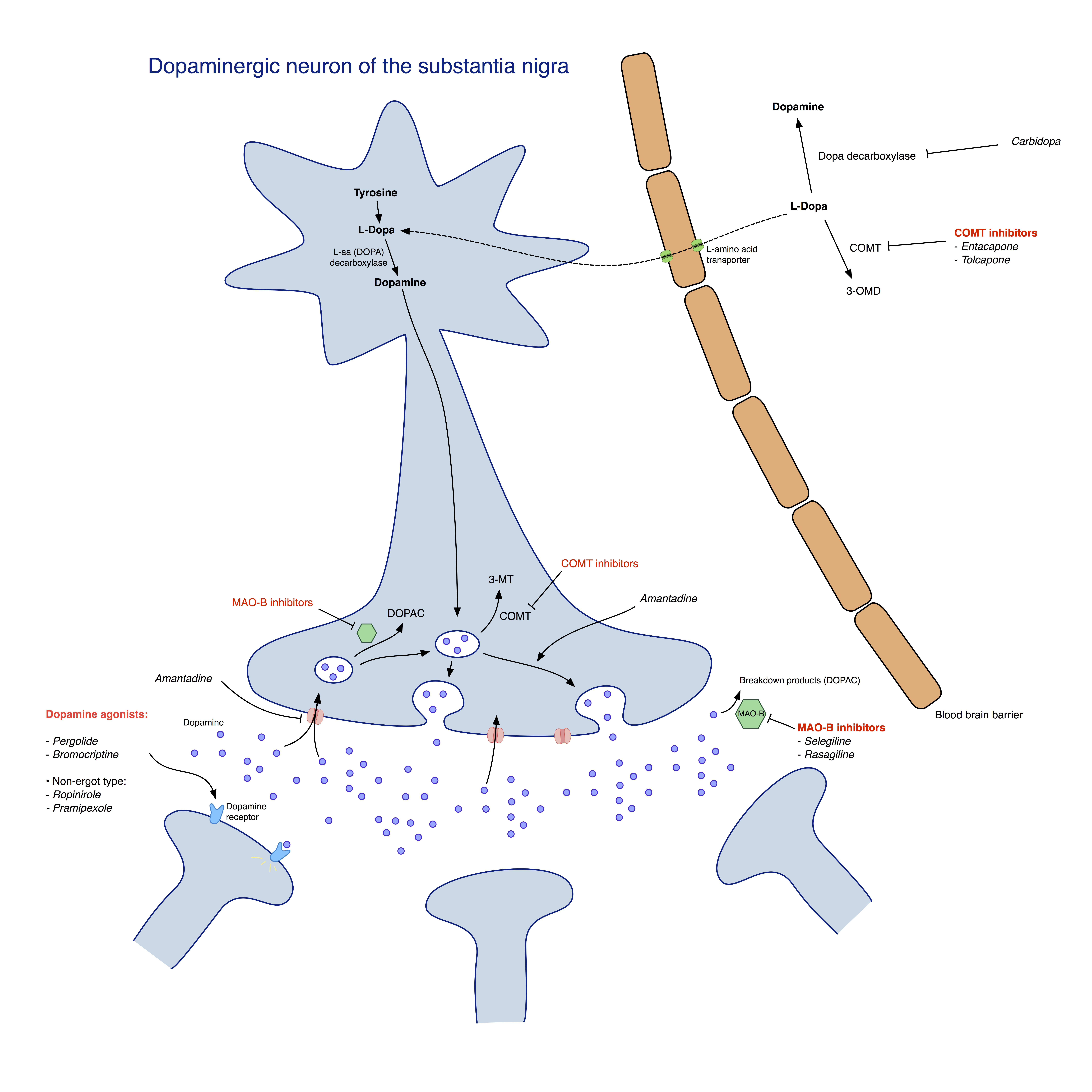 No cure for Parkinson's disease is known. Medications, surgery, and physical treatment may provide relief, improve the quality of a person's life, and are much more effective than treatments available for other neurological disorders such as Alzheimer's disease, motor neuron disease, and Parkinson-plus syndromes. The main families of drugs useful for treating motor symptoms are levodopa always combined with a dopa decarboxylase inhibitor and sometimes also with a
No cure for Parkinson's disease is known. Medications, surgery, and physical treatment may provide relief, improve the quality of a person's life, and are much more effective than treatments available for other neurological disorders such as Alzheimer's disease, motor neuron disease, and Parkinson-plus syndromes. The main families of drugs useful for treating motor symptoms are levodopa always combined with a dopa decarboxylase inhibitor and sometimes also with a
 During the course of PD, affected people can experience a wearing off phenomenon, where they have a recurrence of symptoms after a dose of levodopa, but right before their next dose. Catechol-O-methyltransferase (COMT) is a protein that degrades levodopa before it can cross the blood–brain barrier and these inhibitors allow for more levodopa to cross. They are normally not used in the management of early symptoms, but can be used in conjunction with levodopa/carbidopa when a person is experiencing the wearing off phenomenon with their motor symptoms.
Three COMT inhibitors are available to treat adults with PD and end-of-dose motor fluctuations –
During the course of PD, affected people can experience a wearing off phenomenon, where they have a recurrence of symptoms after a dose of levodopa, but right before their next dose. Catechol-O-methyltransferase (COMT) is a protein that degrades levodopa before it can cross the blood–brain barrier and these inhibitors allow for more levodopa to cross. They are normally not used in the management of early symptoms, but can be used in conjunction with levodopa/carbidopa when a person is experiencing the wearing off phenomenon with their motor symptoms.
Three COMT inhibitors are available to treat adults with PD and end-of-dose motor fluctuations –
 Treating motor symptoms with surgery was once a common practice, but since the discovery of levodopa, the number of operations has declined. Studies in the past few decades have led to great improvements in surgical techniques, so surgery is again being used in people with advanced PD for whom drug therapy is no longer sufficient. Surgery for PD can be divided in two main groups – lesional and
Treating motor symptoms with surgery was once a common practice, but since the discovery of levodopa, the number of operations has declined. Studies in the past few decades have led to great improvements in surgical techniques, so surgery is again being used in people with advanced PD for whom drug therapy is no longer sufficient. Surgery for PD can be divided in two main groups – lesional and
 PD invariably progresses with time. A severity rating method known as the Unified Parkinson's disease rating scale (UPDRS) is the most commonly used metric for a clinical study. A modified version known as the MDS-UPDRS is also sometimes used. An older scaling method known as the Hoehn and Yahr scale (originally published in 1967), and a similar scale known as the Modified Hoehn and Yahr scale, have also been commonly used. The Hoehn and Yahr scale defines five basic stages of progression.
Motor symptoms, if not treated, advance aggressively in the early stages of the disease and more slowly later. Untreated, individuals are expected to lose independent ambulation after an average of eight years and be bedridden after 10 years. However, it is uncommon to find untreated people nowadays. Medication has improved the prognosis of motor symptoms, while at the same time it is a new source of disability, because of the undesired effects of levodopa after years of use. In people taking levodopa, the progression time of symptoms to a stage of high dependency from caregivers may be over 15 years. Predicting what course the disease will take for a given individual is difficult. Age is the best predictor of disease progression. The rate of motor decline is greater in those with less impairment at the time of diagnosis, while cognitive impairment is more frequent in those who are over 70 years of age at symptom onset.
Since current therapies improve motor symptoms, disability at present is mainly related to nonmotor features of the disease. Nevertheless, the relationship between disease progression and disability is not linear. Disability is initially related to motor symptoms. As the disease advances, disability is more related to motor symptoms that do not respond adequately to medication, such as swallowing/speech difficulties, and gait/balance problems; and also to levodopa-induced complications, which appear in up to 50% of individuals after 5 years of levodopa usage. Finally, after ten years most people with the disease have autonomic disturbances, sleep problems, mood alterations and cognitive decline. All of these symptoms, especially cognitive decline, greatly increase disability.
The life expectancy of people with PD is reduced. Mortality ratios are around twice those of unaffected people. Cognitive decline and dementia, old age at onset, a more advanced disease state, and presence of swallowing problems are all mortality
PD invariably progresses with time. A severity rating method known as the Unified Parkinson's disease rating scale (UPDRS) is the most commonly used metric for a clinical study. A modified version known as the MDS-UPDRS is also sometimes used. An older scaling method known as the Hoehn and Yahr scale (originally published in 1967), and a similar scale known as the Modified Hoehn and Yahr scale, have also been commonly used. The Hoehn and Yahr scale defines five basic stages of progression.
Motor symptoms, if not treated, advance aggressively in the early stages of the disease and more slowly later. Untreated, individuals are expected to lose independent ambulation after an average of eight years and be bedridden after 10 years. However, it is uncommon to find untreated people nowadays. Medication has improved the prognosis of motor symptoms, while at the same time it is a new source of disability, because of the undesired effects of levodopa after years of use. In people taking levodopa, the progression time of symptoms to a stage of high dependency from caregivers may be over 15 years. Predicting what course the disease will take for a given individual is difficult. Age is the best predictor of disease progression. The rate of motor decline is greater in those with less impairment at the time of diagnosis, while cognitive impairment is more frequent in those who are over 70 years of age at symptom onset.
Since current therapies improve motor symptoms, disability at present is mainly related to nonmotor features of the disease. Nevertheless, the relationship between disease progression and disability is not linear. Disability is initially related to motor symptoms. As the disease advances, disability is more related to motor symptoms that do not respond adequately to medication, such as swallowing/speech difficulties, and gait/balance problems; and also to levodopa-induced complications, which appear in up to 50% of individuals after 5 years of levodopa usage. Finally, after ten years most people with the disease have autonomic disturbances, sleep problems, mood alterations and cognitive decline. All of these symptoms, especially cognitive decline, greatly increase disability.
The life expectancy of people with PD is reduced. Mortality ratios are around twice those of unaffected people. Cognitive decline and dementia, old age at onset, a more advanced disease state, and presence of swallowing problems are all mortality
 PD is the second most common neurodegenerative disorder after Alzheimer's disease and affects approximately seven million people globally and one million people in the United States. The proportion in a population at a given time is about 0.3% in industrialized countries. PD is more common in the elderly and rates rise from 1% in those over 60 years of age to 4% of the population over 80. The mean age of onset is around 60 years, although 5–10% of cases, classified as young onset PD, begin between the ages of 20 and 50. Males are more often affected than females at a ratio of around 3:2. PD may be less prevalent in those of African and Asian ancestry, although this finding is disputed. The number of new cases per year of PD is between 8 and 18 per 100,000 person–years.
The age-adjusted rate of Parkinson's disease in Estonia is 28.0/100,000 person years. The Estonian rate has been stable between 2000 and 2019. The incidence of Parkinson's disease has increased in China. It is estimated that China will have nearly half of the Parkinson's disease population in the world in 2030. By 2040 the number of patients is expected to grow to approximately 14 million people; this growth has been referred to as the ''Parkinson's pandemic.''
PD is the second most common neurodegenerative disorder after Alzheimer's disease and affects approximately seven million people globally and one million people in the United States. The proportion in a population at a given time is about 0.3% in industrialized countries. PD is more common in the elderly and rates rise from 1% in those over 60 years of age to 4% of the population over 80. The mean age of onset is around 60 years, although 5–10% of cases, classified as young onset PD, begin between the ages of 20 and 50. Males are more often affected than females at a ratio of around 3:2. PD may be less prevalent in those of African and Asian ancestry, although this finding is disputed. The number of new cases per year of PD is between 8 and 18 per 100,000 person–years.
The age-adjusted rate of Parkinson's disease in Estonia is 28.0/100,000 person years. The Estonian rate has been stable between 2000 and 2019. The incidence of Parkinson's disease has increased in China. It is estimated that China will have nearly half of the Parkinson's disease population in the world in 2030. By 2040 the number of patients is expected to grow to approximately 14 million people; this growth has been referred to as the ''Parkinson's pandemic.''
 Several early sources, including an Egyptian papyrus, an Ayurvedic medical treatise, the Bible, and
Several early sources, including an Egyptian papyrus, an Ayurvedic medical treatise, the Bible, and
 The costs of PD to society are high, but precise calculations are difficult due to methodological issues in research and differences between countries. The largest share of direct cost comes from inpatient care and nursing homes, while the share coming from medication is substantially lower. Indirect costs are high, due to reduced productivity and the burden on caregivers. In addition to economic costs, PD reduces quality of life of those with the disease and their caregivers.
A study based on 2017 data estimated the US economic PD burden at $51.9 billion, including direct medical costs of $25.4 billion and $26.5 billion in indirect and non-medical costs. The Medicare program bears the largest share of medical costs, as most PD patients are over age 65. The projected total economic burden surpasses $79 billion by 2037. These findings highlight the need for interventions to reduce PD incidence, delay disease progression, and alleviate symptom burden that may reduce the future economic burden of PD.
The costs of PD to society are high, but precise calculations are difficult due to methodological issues in research and differences between countries. The largest share of direct cost comes from inpatient care and nursing homes, while the share coming from medication is substantially lower. Indirect costs are high, due to reduced productivity and the burden on caregivers. In addition to economic costs, PD reduces quality of life of those with the disease and their caregivers.
A study based on 2017 data estimated the US economic PD burden at $51.9 billion, including direct medical costs of $25.4 billion and $26.5 billion in indirect and non-medical costs. The Medicare program bears the largest share of medical costs, as most PD patients are over age 65. The projected total economic burden surpasses $79 billion by 2037. These findings highlight the need for interventions to reduce PD incidence, delay disease progression, and alleviate symptom burden that may reduce the future economic burden of PD.
 Actor Michael J. Fox has PD and has greatly increased the public awareness of the disease. After diagnosis, Fox embraced his Parkinson's in television roles, sometimes acting without medication, to further illustrate the effects of the condition. He has written four autobiographies in which his fight against the disease plays a major role, and appeared before the
Actor Michael J. Fox has PD and has greatly increased the public awareness of the disease. After diagnosis, Fox embraced his Parkinson's in television roles, sometimes acting without medication, to further illustrate the effects of the condition. He has written four autobiographies in which his fight against the disease plays a major role, and appeared before the
Parkinson's disease at Mayo Clinic
*
Parkinson's disease at NHS
* ttps://web.archive.org/web/20061109041052/http://www.wpda.org/ World Parkinson Disease Association
PDGENE – Database for Parkinson's Disease genetic association studies
{{Authority control Aging-associated diseases Ailments of unknown cause Geriatrics Neurodegenerative disorders Cytoskeletal defects Wikipedia medicine articles ready to translate Wikipedia neurology articles ready to translate
central nervous system
The central nervous system (CNS) is the part of the nervous system consisting primarily of the brain and spinal cord. The CNS is so named because the brain integrates the received information and coordinates and influences the activity of all p ...
that mainly affects the motor system
The motor system is the set of central and peripheral structures in the nervous system that support motor functions, i.e. movement. Peripheral structures may include skeletal muscles and neural connections with muscle tissues. Central structur ...
. The symptoms usually emerge slowly, and as the disease worsens, non-motor symptoms become more common. The most obvious early symptoms are tremor, rigidity, slowness of movement, and difficulty with walking. Cognitive and behavioral problems may also occur with depression, anxiety, and apathy occurring in many people with PD. Parkinson's disease dementia becomes common in the advanced stages of the disease. Those with Parkinson's can also have problems with their sleep and sensory systems. The motor symptoms of the disease result from the death of cells
Cell death is the event of a biological cell ceasing to carry out its functions. This may be the result of the natural process of old cells dying and being replaced by new ones, as in programmed cell death, or may result from factors such as di ...
in the substantia nigra, a region of the midbrain, leading to a dopamine deficit. The cause of this cell death is poorly understood, but involves the build-up of misfolded proteins into Lewy bodies in the neuron
A neuron, neurone, or nerve cell is an electrically excitable cell that communicates with other cells via specialized connections called synapses. The neuron is the main component of nervous tissue in all animals except sponges and placozoa. ...
s. Collectively, the main motor symptoms are also known as parkinsonism or a parkinsonian syndrome.
The cause of PD is unknown, but a combination of genetic factors, and environmental factors are believed to play a role. Those with an affected family member are at an increased risk of getting the disease, with certain genes known to be inheritable risk factors. Environmental risk factors of note are exposure to pesticides, and prior head injuries. Coffee
Coffee is a drink prepared from roasted coffee beans. Darkly colored, bitter, and slightly acidic, coffee has a stimulating effect on humans, primarily due to its caffeine content. It is the most popular hot drink in the world.
Seeds of ...
drinkers, tea
Tea is an aromatic beverage prepared by pouring hot or boiling water over cured or fresh leaves of ''Camellia sinensis'', an evergreen shrub native to East Asia which probably originated in the borderlands of southwestern China and north ...
drinkers, and tobacco smokers are at a reduced risk.
Diagnosis of typical cases is mainly based on symptoms, with motor symptoms being the chief complaint. Tests such as neuroimaging ( magnetic resonance imaging or imaging to look at dopamine neuronal dysfunction known as DaT scan) can be used to help rule out other diseases. Parkinson's disease typically occurs in people over the age of 60, of whom about one percent are affected. Males are more often affected than females at a ratio of around 3:2. When it is seen in people before the age of 50, it is called early-onset PD. By 2015, PD affected 6.2 million people and resulted in about 117,400 deaths globally. The number of people with PD older than fifty is expected to double by 2030. The average life expectancy following diagnosis is between 7 and 15 years.
No cure for PD is known; treatment aims to reduce the effects of the symptoms. Initial treatment is typically with the medications levodopa (L-DOPA
-DOPA, also known as levodopa and -3,4-dihydroxyphenylalanine, is an amino acid that is made and used as part of the normal biology of some plants and animals, including humans. Humans, as well as a portion of the other animals that utilize -DOPA ...
), MAO-B inhibitors, or dopamine agonist
A dopamine agonist (DA) is a compound that activates dopamine receptors. There are two families of dopamine receptors, D2-like and D1-like, and they are all G protein-coupled receptors. D1- and D5-receptors belong to the D1-like family and the ...
s. As the disease progresses, these medications become less effective, while at the same time producing a side effect
In medicine, a side effect is an effect, whether therapeutic or adverse, that is secondary to the one intended; although the term is predominantly employed to describe adverse effects, it can also apply to beneficial, but unintended, consequence ...
marked by involuntary muscle movements. At that time, medications may be used in combination and doses may be increased. Diet and certain forms of rehabilitation have shown some effectiveness at improving symptoms. Surgery to place microelectrode
A microelectrode is an electrode used in electrophysiology either for recording neural signals or for the electrical stimulation of nervous tissue (they were first developed by Ida Hyde in 1921). Pulled glass pipettes with tip diameters of 0. ...
s for deep brain stimulation
Deep brain stimulation (DBS) is a neurosurgical procedure involving the placement of a medical device called a neurostimulator, which sends electrical impulses, through implanted electrodes, to specific targets in the brain (the brain nucleus ...
has been used to reduce motor symptoms in severe cases where drugs are ineffective. Evidence for treatments for the nonmovement-related symptoms of PD, such as sleep disturbances and emotional problems, is less strong.
The disease is named after English doctor James Parkinson, who published the first detailed description in ''An Essay on the Shaking Palsy'', in 1817. Public awareness campaigns include World Parkinson's Day (on the birthday of James Parkinson, 11 April) and the use of a red tulip
Tulips (''Tulipa'') are a genus of spring-blooming perennial herbaceous bulbiferous geophytes (having bulbs as storage organs). The flowers are usually large, showy and brightly coloured, generally red, pink, yellow, or white (usually in warm ...
as the symbol of the disease. People with PD who have increased the public's awareness of the condition include boxer Muhammad Ali, comedian Billy Connolly
Sir William Connolly (born 24 November 1942) is a Scottish actor, retired comedian, artist, writer, musician, and presenter. He is sometimes known, especially in his homeland, by the Scots nickname the Big Yin ("the Big One"). Known for his ...
, actor Michael J. Fox, Olympic cyclist Davis Phinney
Davis Phinney (born July 10, 1959) is a retired professional road bicycle racer from the United States. He won 328 races in the 1980s and 1990s, a record for an American, including two Tour de France stages. He has worked in media since retiring ...
, and actor Alan Alda
Alan Alda (; born Alphonso Joseph D'Abruzzo; January 28, 1936) is an American actor, screenwriter, and director. A six-time Emmy Award and Golden Globe Award winner, he is best known for playing Captain Benjamin "Hawkeye" Pierce in the war come ...
.
Classification
Parkinson's disease is the most common form of parkinsonism and is sometimes called ''idiopathic parkinsonism'', meaning that it has no identifiable cause. Parkinson's disease is a neurodegenerative disease classed as a synucleinopathy, and more specifically as an alpha-synucleinopathy (αsynucleinopathy) due to the accumulation of a misfolded proteinalpha-synuclein
Alpha-synuclein is a protein that, in humans, is encoded by the ''SNCA'' gene. Alpha-synuclein is a neuronal protein that regulates synaptic vesicle trafficking and subsequent neurotransmitter release.
It is abundant in the brain, while smaller a ...
in the brain
A brain is an organ that serves as the center of the nervous system in all vertebrate and most invertebrate animals. It is located in the head, usually close to the sensory organs for senses such as vision. It is the most complex organ in a ve ...
, and its spread throughout the brain.
There are other Parkinson-plus syndromes that can have similar movement symptoms, but have a variety of associated symptoms. Some of these are also synucleinopathies. Lewy body dementia involves motor symptoms with early onset of cognitive dysfunction and hallucinations, with these often (though not necessarily) preceding the motor symptoms. Alternatively, multiple systems atrophy or MSA usually has early onset of autonomic dysfunction (such as orthostasis), and may have autonomic predominance, cerebellar symptom predominance, or Parkinsonian predominance.
Other Parkinson-plus syndromes involve tau
Tau (uppercase Τ, lowercase τ, or \boldsymbol\tau; el, ταυ ) is the 19th letter of the Greek alphabet, representing the voiceless dental or alveolar plosive . In the system of Greek numerals, it has a value of 300.
The name in English ...
, rather than alpha-synuclein. These include progressive supranuclear palsy
Progressive supranuclear palsy (PSP) is a late-onset degenerative disease involving the gradual deterioration and death of specific volumes of the brain. The condition leads to symptoms including loss of balance, slowing of movement, difficulty ...
(PSP) and corticobasal syndrome (CBS). PSP predominantly involves rigidity, early falls, bulbar symptoms, and vertical gaze restriction; it can also be associated with frontotemporal dementia symptoms. CBS involves asymmetric parkinsonism, dystonia, alien limb, and myoclonic jerking. These unique presentation timelines and associated symptoms can help develop these similar movement disorders from idiopathic Parkinson disease.
Signs and symptoms
 The most recognizable symptoms are movement (motor) related, and include tremor,
The most recognizable symptoms are movement (motor) related, and include tremor, bradykinesia
Hypokinesia is one of the classifications of movement disorders, and refers to decreased bodily movement. Hypokinesia is characterized by a partial or complete loss of muscle movement due to a disruption in the basal ganglia. Hypokinesia is a sym ...
, rigidity, and shuffling/stooped gait. Non-motor symptoms, including autonomic dysfunction ( dysautonomia), neuropsychiatric
Neuropsychiatry or Organic Psychiatry is a branch of medicine that deals with psychiatry as it relates to neurology, in an effort to understand and attribute behavior to the interaction of neurobiology and social psychology factors. Within neurop ...
problems (mood, cognition, behavior or thought alterations), and sensory (especially altered sense of smell) and sleep difficulties may be present as well. Patients may have nonmotor symptoms that precede the onset of motor symptoms by several years, such as constipation, anosmia, and REM behavior disorder. Generally, symptoms such as dementia, psychosis, orthostasis, and more severe falls do not occur until later.
Motor
Four motor symptoms are considered as cardinal signs in PD: tremor, slowness of movement (bradykinesia), rigidity, and postural instability. The most common presenting sign is a coarse, slow tremor of the hand at rest, which disappears during voluntary movement of the affected arm and in the deeper stages of sleep. It typically appears in only one hand, eventually affecting both hands as the disease progresses. Frequency of PD tremor is between 4 and 6hertz
The hertz (symbol: Hz) is the unit of frequency in the International System of Units (SI), equivalent to one event (or cycle) per second. The hertz is an SI derived unit whose expression in terms of SI base units is s−1, meaning that o ...
(cycles per second). A feature of tremor is pill-rolling, the tendency of the index finger and thumb to touch and perform together with a circular movement. The term derives from the similarity between the movement of people with PD and the early pharmaceutical technique of manually making pills.
Bradykinesia
Hypokinesia is one of the classifications of movement disorders, and refers to decreased bodily movement. Hypokinesia is characterized by a partial or complete loss of muscle movement due to a disruption in the basal ganglia. Hypokinesia is a sym ...
is found in every case of PD, and is due to disturbances in motor planning of movement initiation, and associated with difficulties along the whole course of the movement process, from planning to initiation to execution of a movement. Performance of sequential and simultaneous movement is impaired. Bradykinesia is the most handicapping symptom of Parkinson's disease, leading to difficulties with everyday tasks such as dressing, feeding, and bathing. It leads to particular difficulty in carrying out two independent motor activities at the same time, and can be made worse by emotional stress or concurrent illnesses. Paradoxically, people with PD can often ride a bicycle or climb stairs more easily than walk on the level. While most physicians may readily notice bradykinesia, formal assessment requires persons to do repetitive movements with their fingers and feet.
Rigidity is stiffness and resistance to limb movement caused by increased muscle tone, an excessive and continuous contraction of muscles. In parkinsonism, the rigidity can be uniform, known as ''lead-pipe rigidity'', or ratcheted, known as ''cogwheel rigidity''. The combination of tremor and increased tone is considered to be at the origin of cogwheel rigidity. Rigidity may be associated with joint pain; such pain being a frequent initial manifestation of the disease. In early stages of PD, rigidity is often asymmetrical and tends to affect the neck and shoulder muscles prior to the muscles of the face and extremities. With the progression of the disease, rigidity typically affects the whole body and reduces the ability to move.
Postural instability
A balance disorder is a disturbance that causes an individual to feel unsteady, for example when standing or walking. It may be accompanied by feelings of giddiness, or wooziness, or having a sensation of movement, spinning, or floating. Balance ...
is typical in the later stages of the disease, leading to impaired balance and frequent falls, and secondarily to bone fractures, loss of confidence, and reduced mobility. Instability is often absent in the initial stages, especially in younger people, especially prior to the development of bilateral symptoms. Up to 40% of people diagnosed with PD may experience falls, and around 10% may have falls weekly, with the number of falls being related to the severity of PD.
Other recognized motor signs and symptoms include gait and posture disturbances such as festination (rapid shuffling steps and a forward-flexed posture when walking with no flexed arm swing). Other common signs include freezing of gait (brief arrests when the feet seem to get stuck to the floor, especially on turning or changing direction), a slurred, monotonous, quiet voice, mask-like facial expression, and handwriting that gets smaller and smaller.
Cognitive
PD causesneuropsychiatric
Neuropsychiatry or Organic Psychiatry is a branch of medicine that deals with psychiatry as it relates to neurology, in an effort to understand and attribute behavior to the interaction of neurobiology and social psychology factors. Within neurop ...
disturbances ranging from mild to severe. They include disorders of cognition, mood, behavior, and thought. Cognitive disturbances can occur in the early stages or sometimes prior to diagnosis, and increase in prevalence with duration of the disease. The most common cognitive deficit is executive dysfunction
In psychology and neuroscience, executive dysfunction, or executive function deficit, is a disruption to the efficacy of the executive functions, which is a group of cognitive processes that regulate, control, and manage other cognitive processes ...
, which can include problems with planning, cognitive flexibility, abstract thinking, rule acquisition, inhibiting inappropriate actions, initiating appropriate actions, working memory, and control of attention. Other cognitive difficulties include slowed cognitive processing speed, impaired recall, and impaired perception and estimation of time. Nevertheless, improvement appears when recall is aided by cues. Visuospatial difficulties are also part of the disease, seen for example when the individual is asked to perform tests of facial recognition and perception of the orientation of drawn lines.
A person with PD has two to six times the risk of dementia compared to the general population. Up to 78% of people with PD have Parkinson's disease dementia. The prevalence of dementia increases with age, and to a lesser degree, duration of the disease. Dementia is associated with a reduced quality of life in people with PD and their caregivers, increased mortality, and a higher probability of needing nursing home care.
Psychosis
Psychosis can be considered a symptom with a prevalence at its widest range from 26 to 83%.Hallucination
A hallucination is a perception in the absence of an external stimulus that has the qualities of a real perception. Hallucinations are vivid, substantial, and are perceived to be located in external objective space. Hallucination is a combinati ...
s or delusions occur in about 50% of people with PD over the course of the illness, and may herald the emergence of dementia. These range from minor hallucinations – sense of passage (something quickly passing beside the person) or sense of presence (the perception of something/someone standing just to the side or behind the person) – to full blown vivid, formed visual hallucinations and paranoid ideation. Auditory hallucinations are uncommon in PD, and are rarely described as voices. Psychosis is now believed to be an integral part of the disease. A psychosis with delusions and associated delirium is a recognized complication of anti-Parkinson drug treatment and may also be caused by urinary-tract infections (as frequently occurs in the fragile elderly), but drugs and infection are not the only factors, and underlying brain pathology or changes in neurotransmitters or their receptors (e.g., acetylcholine, serotonin) are also thought to play a role in psychosis in PD.
Neuropsychiatric
Behavior and mood alterations are more common in PD without cognitive impairment than in the general population and are usually present in PD with dementia. The most frequent mood difficulties are depression, apathy, and anxiety. Depression has been estimated to appear in 20 to 35% of people with PD, and can appear at any stage of the disease. It can manifest with symptoms that are common to the disease process (fatigue, insomnia, and difficulty with concentration), which makes diagnosis difficult. The imbalance and changes in dopamine, serotonin, andnoradrenergic
Norepinephrine (NE), also called noradrenaline (NA) or noradrenalin, is an organic chemical in the catecholamine family that functions in the brain and body as both a hormone and neurotransmitter. The name "noradrenaline" (from Latin '' ad'', ...
hormones are known to be a primary cause of depression in PD-affected people. Another cause is the functional impairment that is caused by the disease. Symptoms of depression can include loss of interest, sadness, guilt, feelings of helplessness/hopelessness/guilt, and suicidal ideation. Suicidal ideation in PD-affected people is higher than in the general population, but suicidal attempts themselves are lower than in people with depression without PD. Risk factors for depression in PD can include disease onset under age 50, being a woman, previous history of depression, severe motor symptoms, and others.
Anxiety has been estimated to have a prevalence in PD-affected people usually around 30–40% (60% has been found). Anxiety can often be found during off periods (times when medication is not working as well as it did before). PD-affected people experience panic attacks more frequently compared to the general population. Both anxiety and depression have been found to be associated with decreased quality of life. Symptoms can range from mild and episodic to chronic with potential causes being abnormal gamma-aminobutyric acid levels and embarrassment or fear about symptoms or disease. Risk factors for anxiety in PD are disease onset under age 50, women, and off periods.
Apathy and anhedonia can be defined as a loss of motivation and an impaired ability to experience pleasure, respectively. They are symptoms classically associated with depression, but they differ in PD-affected people in treatment and mechanism and do not always occur with depression. Apathy presents in around 16.5–40%. Symptoms of apathy include reduced initiative/interests in new activities or the world around them, emotional indifference, and loss of affection or concern for others. Apathy is associated with deficits in cognitive functions including executive and verbal memory. Anhedonia occurs in 5-75% of people with PD, depending on the study population assessed, and has a significant overlap with apathy.
Impulse-control disorders, including pathological gambling, compulsive sexual behavior, binge eating, compulsive shopping, and reckless generosity, can be caused by medication, particularly orally active dopamine agonists. The dopamine dysregulation syndrome – with wanting of medication leading to overuse – is a rare complication of levodopa use.
Punding, in which complicated, repetitive, aimless, stereotyped behaviors occur for many hours, is another disturbance caused by anti-Parkinson medication.
Gastrointestinal
Gastrointestinal issues in Parkinson's disease include constipation, impaired stomach emptying (gastric dysmotility), and excessive production of saliva can be severe enough to cause discomfort or endanger health. Other upper gastrointestinal symptoms include swallowing impairment ( Oropharyngeal dysphagia) and small intestinal bacterial overgrowth. Invidividuals with Parkinson's have alpha-synuclein deposits in the digestive tract as well as the brain. Constipation is one of the symptoms associated with an increased risk of PD, and may precede diagnosis by several years.Other
Sleep disorder
A sleep disorder, or somnipathy, is a medical disorder of an individual's sleep patterns. Some sleep disorders are severe enough to interfere with normal physical, mental, social and emotional functioning. Polysomnography and actigraphy are tests ...
s are a feature of the disease and can be worsened by medications. Symptoms can manifest as daytime drowsiness
Somnolence (alternatively sleepiness or drowsiness) is a state of strong desire for sleep, or sleeping for unusually long periods (compare hypersomnia). It has distinct meanings and causes. It can refer to the usual state preceding falling asleep ...
(including sudden sleep attacks resembling narcolepsy
Narcolepsy is a long-term neurological disorder that involves a decreased ability to regulate sleep–wake cycles. Symptoms often include periods of excessive daytime sleepiness and brief involuntary sleep episodes. About 70% of those affect ...
), disturbances in Rapid eye movement sleep
Rapid eye movement sleep (REM sleep or REMS) is a unique phase of sleep in mammals and birds, characterized by random rapid movement of the eyes, accompanied by low muscle tone throughout the body, and the propensity of the sleeper to dream ...
, or insomnia
Insomnia, also known as sleeplessness, is a sleep disorder in which people have trouble sleeping. They may have difficulty falling asleep, or staying asleep as long as desired. Insomnia is typically followed by daytime sleepiness, low energy, ...
. REM behavior disorder, in which people act out dreams, sometimes injuring themselves or their bed partner, may begin many years before the development of motor or cognitive features of PD or dementia with Lewy bodies
Dementia with Lewy bodies (DLB) is a type of dementia characterized by changes in sleep, behavior, cognition, movement, and regulation of automatic bodily functions. Memory loss is not always an early symptom. The disease worsens over time an ...
.
Alterations in the autonomic nervous system can lead to orthostatic hypotension (low blood pressure upon standing), oily skin, excessive sweating, urinary incontinence
Urinary incontinence (UI), also known as involuntary urination, is any uncontrolled leakage of urine. It is a common and distressing problem, which may have a large impact on quality of life. It has been identified as an important issue in geri ...
, and altered sexual function.
Changes in perception may include an impaired sense of smell, disturbed vision, pain, and paresthesia (tingling and numbness). All of these symptoms can occur years before diagnosis of the disease.
Causes
Many risk factors have been proposed, sometimes in relation to theories concerning possible mechanisms of the disease; however, none has been proven conclusively. The most frequently replicated relationships are an increased risk in those exposed to pesticides, and a reduced risk in smokers. A possible link exists between PD and ''Helicobacter pylori
''Helicobacter pylori'', previously known as ''Campylobacter pylori'', is a gram-negative, microaerophilic, spiral (helical) bacterium usually found in the stomach. Its helical shape (from which the genus name, helicobacter, derives) is thoug ...
'' infection that can prevent the absorption of some drugs, including levodopa.
Genetic
 Research indicates that PD is the product of a complex interaction of genetic and
Research indicates that PD is the product of a complex interaction of genetic and environmental factor
An environmental factor, ecological factor or eco factor is any factor, abiotic or biotic, that influences living organisms. Abiotic factors include ambient temperature, amount of sunlight, and pH of the water soil in which an organism lives. Bi ...
s. Around 15% of individuals with PD have a first-degree relative
A first-degree relative (FDR) is a person's parent (father or mother), full sibling (brother or sister) or child. It constitutes a category of family members that largely overlaps with the term nuclear family, but without spouses.
If the persons ...
who has the disease, and 5–10% of people with PD are known to have forms of the disease that occur because of a mutation in one of several specific genes. Harboring one of these gene mutations may not lead to the disease; susceptibility factors put the individual at an increased risk, often in combination with other risk factors, which also affect age of onset, severity and progression. At least 11 autosomal dominant and 9 autosomal recessive gene mutations have been implicated in the development of PD. The autosomal dominant genes include ''SNCA'','' PARK3'', ''UCHL1'', '' LRRK2'', ''GIGYF2'', ''HTRA2'', ''EIF4G1'', '' TMEM230'', '' CHCHD2'', '' RIC3'', and ''VPS35
Vacuolar protein sorting ortholog 35 (VPS35) is a protein involved in autophagy and is implicated in neurodegenerative diseases, such as Parkinson's disease (PD) and Alzheimer's disease (AD). VPS35 is part of a complex called the retromer, which ...
''. Autosomal recessive genes include ''PRKN
Parkin is a 465-amino acid residue (chemistry), residue E3 ubiquitin ligase, a protein that in humans and mice is encoded by the ''PARK2'' gene. Parkin plays a critical role in ubiquitination – the process whereby molecules are covalently labell ...
, PINK1, PARK7, ATP13A2, PLA2G6, FBXO7, DNAJC6
Putative tyrosine-protein phosphatase auxilin is an enzyme that in humans is encoded by the ''DNAJC6'' gene.
Function
DNAJC6 belongs to the evolutionarily conserved DNAJ/HSP40 family of proteins, which regulate molecular chaperone activity b ...
, SYNJ1,'' and '' VPS13C''. Some genes are X-linked
Sex linked describes the sex-specific patterns of inheritance and presentation when a gene mutation (allele) is present on a sex chromosome (allosome) rather than a non-sex chromosome (autosome). In humans, these are termed X-linked recessive, ...
or have unknown inheritance pattern; those include '' PARK10, PARK12'', and '' PARK16''. A 22q11 Q11 may refer to:
* Q11 (New York City bus)
* , a survey ship of the Argentine Navy
* DZOE-TV
DZOE-TV, Channel 11 (analog) and Channel 20 (digital), is the flagship television property of ZOE Broadcasting Network. As part of a blocktime ag ...
deletion is also known to be associated with PD. An autosomal dominant form has been associated with mutations in the'' LRP10
Low-density lipoprotein receptor-related protein 10 is a protein that in humans is encoded by the ''LRP10'' gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." ...
'' gene.
About 5% of people with PD have mutations in the '' GBA1'' gene. These mutations are present in less than 1% of the unaffected population. The risk of developing PD is increased 20–30 fold if these mutations are present. PD associated with these mutations has the same clinical features, but an earlier age of onset and a more rapid cognitive and motor decline. This gene encodes glucocerebrosidase
β-Glucocerebrosidase (also called acid β-glucosidase, D-glucosyl-N-acylsphingosine glucohydrolase, or GCase) is an enzyme with glucosylceramidase activity () that is needed to cleave, by hydrolysis, the beta-glycosidic linkage of the chemical ...
. Low levels of this enzyme cause Gaucher's disease.
''SNCA'' gene mutations are important in PD because the protein this gene encodes, alpha-synuclein
Alpha-synuclein is a protein that, in humans, is encoded by the ''SNCA'' gene. Alpha-synuclein is a neuronal protein that regulates synaptic vesicle trafficking and subsequent neurotransmitter release.
It is abundant in the brain, while smaller a ...
, is the main component of the Lewy bodies that accumulate in the brains of people with PD. Alpha-synuclein activates ataxia telangiectasia mutated, a major DNA damage-repair signaling kinase. In addition, alpha-synuclein activates the non-homologous end joining DNA repair pathway. The aggregation of alpha-synuclein in Lewy bodies appears to be a link between reduced DNA repair and brain-cell death in PD.
Mutations in some genes, including ''SNCA'', ''LRRK2'', and ''GBA'', have been found to be risk factors for sporadic (nonfamilial) PD. Mutations in the gene ''LRRK2'' are the most common known cause of familial and sporadic PD, accounting for around 5% of individuals with a family history of the disease and 3% of sporadic cases. A mutation in ''GBA'' presents the greatest genetic risk of developing Parkinsons disease.
Several Parkinson-related genes are involved in the function of lysosomes, organelles that digest cellular waste products. Some cases of PD may be caused by lysosomal disorders that reduce the ability of cells to break down alpha-synuclein.
Non-genetic
Exposure to pesticides and a history of head injury have each been linked with PD, but the risks are modest. Never drinking caffeinated beverages is also associated with small increases in risk of developing PD. Some toxins can cause parkinsonism, includingmanganese
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy use ...
and carbon disulfide
Carbon disulfide (also spelled as carbon disulphide) is a neurotoxic, colorless, volatile liquid with the formula and structure . The compound is used frequently as a building block in organic chemistry as well as an industrial and chemical n ...
.
Medical drugs implicated in cases of parkinsonism. Drug-induced parkinsonism is normally reversible by stopping the offending agent, such as phenothiazines (chlorpromazine, promazine, etc.); butyrophenone
Butyrophenone is an organic compound with the formula C6H5C(O)C3H7. It is a colorless liquid.
The butyrophenone structure—a ketone flanked by a phenyl ring and a butyl chain—forms the basis for many other chemicals containing various sub ...
s (haloperidol, benperidol, etc.); metoclopramide
Metoclopramide is a medication used for stomach and esophageal problems. It is commonly used to treat and prevent nausea and vomiting, to help with emptying of the stomach in people with delayed stomach emptying, and to help with gastroesoph ...
and Tetrabenazine
Tetrabenazine is a drug for the symptomatic treatment of hyperkinetic movement disorders. It is sold under the brand names Nitoman and Xenazine among others. On August 15, 2008, the U.S. Food and Drug Administration approved the use of tetrabena ...
. 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) is a drug known for causing irreversible parkinsonism that is commonly used in animal-model research.
Low concentrations of urate in the blood is associated with an increased risk of PD.
Other identifiable causes of parkinsonism include infections and metabolic derangement. Several neurodegenerative disorders
A neurodegenerative disease is caused by the progressive loss of structure or function of neurons, in the process known as neurodegeneration. Such neuronal damage may ultimately involve cell death. Neurodegenerative diseases include amyotrophic ...
also may present with parkinsonism, and are sometimes referred to as ''atypical parkinsonism'' or ''parkinson plus'' syndromes (illnesses with parkinsonism plus some other features distinguishing them from PD). They include multiple system atrophy, progressive supranuclear palsy
Progressive supranuclear palsy (PSP) is a late-onset degenerative disease involving the gradual deterioration and death of specific volumes of the brain. The condition leads to symptoms including loss of balance, slowing of movement, difficulty ...
, corticobasal degeneration, and dementia with Lewy bodies. Dementia with Lewy bodies is another synucleinopathy and it has close pathological similarities with PD, especially with the subset of PD cases with dementia known as Parkinson's disease dementia. The relationship between PD and DLB is complex and incompletely understood. They may represent parts of a continuum, with variable distinguishing clinical and pathological features, or they may prove to be separate diseases.
Vascular parkinsonism is the phenomenon of the presence of Parkinson's disease symptoms combined with findings of vascular events (such as a cerebral stroke). The damaging of the dopaminergic pathways
Dopaminergic pathways (dopamine pathways, dopaminergic projections) in the human brain are involved in both physiological and behavioral processes including movement, cognition, executive functions, reward, motivation, and neuroendocrine control. ...
is similar in cause for both vascular parkinsonism and idiopathic PD, so they can present with many of the same symptoms. Differentiation can be made with careful bedside examination, history evaluation, and imaging.
Pathophysiology
 The main
The main pathological
Pathology is the study of the causes and effects of disease or injury. The word ''pathology'' also refers to the study of disease in general, incorporating a wide range of biology research fields and medical practices. However, when used in th ...
characteristics of PD are cell death in the brain's basal ganglia (affecting up to 70% of the dopamine-secreting neurons in the substantia nigra pars compacta
The pars compacta (SNpc) is a portion of the ''substantia nigra'', located in the midbrain. It is formed by dopaminergic neurons and located medial to the pars reticulata. Parkinson's disease is characterized by the death of dopaminergic neuron ...
by the end of life). In Parkinson's disease, alpha-synuclein becomes misfolded and clump together with other alpha-synuclein. Cells are unable to remove these clumps, and the alpha-synuclein becomes cytotoxic, damaging the cells. These clumps can be seen in neurons under a microscope and are called Lewy bodies. Loss of neurons is accompanied by the death of astrocyte
Astrocytes (from Ancient Greek , , "star" + , , "cavity", "cell"), also known collectively as astroglia, are characteristic star-shaped glial cells in the brain and spinal cord. They perform many functions, including biochemical control of e ...
s (star-shaped glial cells) and a significant increase in the number of microglia (another type of glial cell) in the substantia nigra. Braak staging is a way to explain the progression of the parts of the brain affected by PD. According to this staging, PD starts in the medulla and the olfactory bulb before moving to the substantia nigra pars compacta and the rest of the midbrain/basal forebrain. Movement symptom onset is associated when the disease begins to affect the substantia nigra pars compacta.
 Five major pathways in the brain connect other brain areas with the basal ganglia. These are known as the
Five major pathways in the brain connect other brain areas with the basal ganglia. These are known as the motor
An engine or motor is a machine designed to convert one or more forms of energy into mechanical energy.
Available energy sources include potential energy (e.g. energy of the Earth's gravitational field as exploited in hydroelectric power g ...
, oculomotor
The oculomotor nerve, also known as the third cranial nerve, cranial nerve III, or simply CN III, is a cranial nerve that enters the orbit through the superior orbital fissure and innervates extraocular muscles that enable most movements of ...
, associative, limbic
The limbic system, also known as the paleomammalian cortex, is a set of brain structures located on both sides of the thalamus, immediately beneath the medial temporal lobe of the cerebrum primarily in the forebrain.Schacter, Daniel L. 2012. ''Ps ...
, and orbitofrontal circuits, with names indicating the main projection area of each circuit. All of them are affected in PD, and their disruption explains many of the symptoms of the disease, since these circuits are involved in a wide variety of functions, including movement, attention and learning. Scientifically, the motor circuit has been examined the most intensively.
dyskinesia
Dyskinesia refers to a category of movement disorders that are characterized by involuntary muscle movements, including movements similar to tics or chorea and diminished voluntary movements. Dyskinesia can be anything from a slight tremor of ...
s.
Brain cell death
Brain cells could be lost by several proposed mechanisms. One mechanism consists of an abnormal accumulation of the protein alpha-synuclein bound toubiquitin
Ubiquitin is a small (8.6 kDa) regulatory protein found in most tissues of eukaryotic organisms, i.e., it is found ''ubiquitously''. It was discovered in 1975 by Gideon Goldstein and further characterized throughout the late 1970s and 1980s. Fo ...
in the damaged cells. This insoluble protein accumulates inside neurons forming inclusions called Lewy bodies. According to the Braak staging, a classification of the disease based on pathological findings proposed by Heiko Braak, Lewy bodies first appear in the olfactory bulb, medulla oblongata, and pontine tegmentum
Pontine may refer to:
* Having to do with the pons, a structure located in the brain stem (from ''pons'', "bridge")
* Pontine Marshes, a region of Italy near Rome
* Pontine Islands
The Pontine Islands (, also ; it, Isole Ponziane ) are an ar ...
; individuals at this stage may be asymptomatic or may have early nonmotor symptoms (such as loss of sense of smell, or some sleep or automatic dysfunction). As the disease progresses, Lewy bodies develop in the substantia nigra, areas of the midbrain and basal forebrain
In the anatomy of the brain of vertebrates, the forebrain or prosencephalon is the rostral (forward-most) portion of the brain. The forebrain (prosencephalon), the midbrain (mesencephalon), and hindbrain (rhombencephalon) are the three primary ...
, and finally, the neocortex. These brain sites are the main places of neuronal degeneration in PD, but Lewy bodies may not cause cell death and they may be protective (with the abnormal protein sequestered or walled off). Other forms of alpha-synuclein (e.g., oligomers) that are not aggregated in Lewy bodies and Lewy neurites may actually be the toxic forms of the protein. In people with dementia, a generalized presence of Lewy bodies is common in cortical areas. Neurofibrillary tangles and senile plaques
Amyloid plaques (also known as neuritic plaques, amyloid beta plaques or senile plaques) are extracellular deposits of the amyloid beta (Aβ) protein mainly in the grey matter of the brain. Degenerative neuronal elements and an abundance of micr ...
, characteristic of Alzheimer's disease, are not common unless the person is demented.
Other cell-death mechanisms include proteasomal and lysosomal systems dysfunction and reduced mitochondrial activity. Iron accumulation in the substantia nigra is typically observed in conjunction with the protein inclusions. It may be related to oxidative stress
Oxidative stress reflects an imbalance between the systemic manifestation of reactive oxygen species and a biological system's ability to readily detoxify the reactive intermediates or to repair the resulting damage. Disturbances in the normal ...
, protein aggregation, and neuronal death, but the mechanisms are not fully understood.
The neuroimmune connection
The neuroimmune interaction is heavily implicated in PD pathology. PD andautoimmune disorders
An autoimmune disease is a condition arising from an abnormal immune response to a functioning body part. At least 80 types of autoimmune diseases have been identified, with some evidence suggesting that there may be more than 100 types. Nearly a ...
share several genetic variations and molecular pathways. Some autoimmune diseases may even increase one's risk of developing PD, up to 33% in one study. Autoimmune diseases linked to protein expression profiles of monocytes and CD4+ T cells are also linked to PD. There is some evidence that Herpes virus infections can trigger autoimmune reactions to alpha-synuclein, perhaps through molecular mimicry of viral proteins. Alpha-synuclein, and its aggregate form Lewy bodies, can also bind to microglia. Microglia can proliferate and be over-activated by alpha-synuclein binding to MHC receptors on inflammasomes, leading to a release of proinflammatory cytokines like IL-1β, IFNγ, and TNFα. Activated microglia also influence the activation of astrocytes, converting their neuroprotective phenotype to a neurotoxic one. Astrocytes in healthy brains serve to protect neuronal connections. In PD patients, astrocytes cannot protect the dopaminergic connections in the striatum. Microglia also present antigens via MHC-I and MHC-II to T cells. CD4+ T cells, activated by this process, are able to cross the blood brain barrier (BBB) and release more proinflammatory cytokines, like interferon-γ (IFNγ), TNFα, and IL-1β. Mast cell degranulation and subsequent proinflammatory cytokine release is also implicated in BBB breakdown in PD. Another immune cell implicated in PD are peripheral monocytes and have been found in the substantia nigra of PD patients. These monocytes can lead to more dopaminergic connection breakdown. In addition, monocytes isolated from PD patients express higher levels of the PD-associated protein, LRRK2, compared to non-PD individuals via vasodilation
Vasodilation is the widening of blood vessels. It results from relaxation of smooth muscle cells within the vessel walls, in particular in the large veins, large arteries, and smaller arterioles. The process is the opposite of vasoconstrictio ...
. In addition, high levels of pro-inflammatory cytokines, such as IL-6, can lead to the production of C-reactive protein
C-reactive protein (CRP) is an annular (ring-shaped) pentameric protein found in blood plasma, whose circulating concentrations rise in response to inflammation. It is an acute-phase protein of hepatic origin that increases following interleukin- ...
by the liver, another protein commonly found in PD patients, that can lead to an increase in peripheral inflammation. Peripheral inflammation can also affect the gut-brain axis, an area of the body highly implicated in PD. PD patients often have altered gut microbiota and colon problems years before motor issues arise. Alpha-synuclein is created in the gut and may migrate via the vagus nerve to the brainstem and then to the substantia nigra. Furthermore, the bacteria ''Proteus mirabilis'' has been associated with higher levels of alpha-synuclein and an increase of motor symptoms in PD patients. Further elucidation of the causal role of alpha-synuclein, the role of inflammation, the gut-brain axis, as well as an understanding of the individual differences in immune stress responses is needed to better understand the pathological development of PD.
Diagnosis
A physician initially assesses for PD with a careful medical history and neurological examination. Focus is put on confirming motor symptoms (bradykinesia, rest tremor, etc.) and supporting tests with clinical diagnostic criteria. The finding of Lewy bodies in the midbrain on autopsy is usually considered final proof that the person had PD. The clinical course of the illness over time may reveal it is not PD, requiring that the clinical presentation be periodically reviewed to confirm the accuracy of the diagnosis. Multiple causes can occur for parkinsonism or diseases that look similar. Stroke, certain medications, and toxins can cause "secondary parkinsonism" and need to be assessed during visit. Parkinson-plus syndromes, such asprogressive supranuclear palsy
Progressive supranuclear palsy (PSP) is a late-onset degenerative disease involving the gradual deterioration and death of specific volumes of the brain. The condition leads to symptoms including loss of balance, slowing of movement, difficulty ...
and multiple system atrophy, must also be considered and ruled out appropriately due to different treatment and disease progression (anti-Parkinson's medications are typically less effective at controlling symptoms in Parkinson-plus syndromes). Faster progression rates, early cognitive dysfunction or postural instability, minimal tremor, or symmetry at onset may indicate a Parkinson-plus disease rather than PD itself.
Medical organizations have created diagnostic criteria
Medical diagnosis (abbreviated Dx, Dx, or Ds) is the process of determining which disease or condition explains a person's symptoms and signs. It is most often referred to as diagnosis with the medical context being implicit. The information req ...
to ease and standardize the diagnostic process, especially in the early stages of the disease. The most widely known criteria come from the UK Queen Square Brain Bank for Neurological Disorders and the U.S. National Institute of Neurological Disorders and Stroke. The Queen Square Brain Bank criteria require slowness of movement (bradykinesia) plus either rigidity, resting tremor, or postural instability. Other possible causes of these symptoms need to be ruled out. Finally, three or more of the following supportive features are required during onset or evolution: unilateral onset, tremor at rest, progression in time, asymmetry of motor symptoms, response to levodopa for at least five years, the clinical course of at least ten years and appearance of dyskinesia
Dyskinesia refers to a category of movement disorders that are characterized by involuntary muscle movements, including movements similar to tics or chorea and diminished voluntary movements. Dyskinesia can be anything from a slight tremor of ...
s induced by the intake of excessive levodopa. Assessment of sudomotor function through electrochemical skin conductance can be helpful in diagnosing dysautonomia.
When PD diagnoses are checked by autopsy, movement disorders experts are found on average to be 79.6% accurate at initial assessment and 83.9% accurate after they have refined their diagnoses at follow-up examinations. When clinical diagnoses performed mainly by nonexperts are checked by autopsy, the average accuracy is 73.8%. Overall, 80.6% of PD diagnoses are accurate, and 82.7% of diagnoses using the Brain Bank criteria are accurate.
Imaging
Computed tomography (CT) scans of people with PD usually appear normal. Magnetic resonance imaging has become more accurate in diagnosis of the disease over time, specifically through iron-sensitive T2* andsusceptibility weighted imaging
Susceptibility weighted imaging (SWI), originally called BOLD venographic imaging, is an MRI sequence that is exquisitely sensitive to venous blood, hemorrhage and iron storage. SWI uses a fully flow compensated, long echo, gradient recalled echo ...
sequences at a magnetic field strength of at least 3T, both of which can demonstrate absence of the characteristic 'swallow tail' imaging pattern in the dorsolateral substantia nigra. In a meta-analysis, absence of this pattern was highly sensitive and specific
Specific may refer to:
* Specificity (disambiguation)
* Specific, a cure or therapy for a specific illness
Law
* Specific deterrence, focussed on an individual
* Specific finding, intermediate verdict used by a jury in determining the fina ...
for the disease. A meta-analysis found that neuromelanin-MRI can discriminate individuals with Parkinson's from healthy subjects. Diffusion MRI
Diffusion-weighted magnetic resonance imaging (DWI or DW-MRI) is the use of specific MRI sequences as well as software that generates images from the resulting data that uses the diffusion of water molecules to generate contrast in MR images. It ...
has shown potential in distinguishing between PD and Parkinson-plus syndromes, as well as between PD motor subtypes, though its diagnostic value is still under investigation. CT and MRI are also used to rule out other diseases that can be secondary causes of parkinsonism, most commonly encephalitis and chronic ischemic insults, as well as less frequent entities such as basal ganglia tumors and hydrocephalus.
The metabolic activity of dopamine transporter
The dopamine transporter (also dopamine active transporter, DAT, SLC6A3) is a membrane-spanning protein that pumps the neurotransmitter dopamine out of the synaptic cleft back into cytosol. In the cytosol, other transporters sequester the dop ...
s in the basal ganglia can be directly measured with positron emission tomography and single-photon emission computed tomography scans, with the DaTSCAN being a common proprietary version of this study. It has shown high agreement with clinical diagnoses of PD. Reduced dopamine-related activity in the basal ganglia can help exclude drug-induced Parkinsonism. This finding is not entirely specific, however, and can be seen with both PD and Parkinson-plus disorders. In the United States, DaTSCANs are only FDA approved to distinguish PD or Parkinsonian syndromes from essential tremor
Essential tremor (ET), also called benign tremor, familial tremor, and idiopathic tremor, is a medical condition characterized by involuntary rhythmic contractions and relaxations ( oscillations or twitching movements) of certain muscle groups in ...
.
Iodine-123-meta-iodobenzylguanidine myocardial scintigraphy can help find denervation of the muscles around the heart, which can support a PD diagnosis.
Differential diagnosis
Secondary parkinsonism – The multiple causes of parkinsonism can be differentiated between with careful history, physical examination, and appropriate imaging. This topic is further discussed in the causes section here. Parkinson-plus syndrome – Multiple diseases can be considered part of the Parkinson's plus group, including corticobasal syndrome, multiple system atrophy,
Parkinson-plus syndrome – Multiple diseases can be considered part of the Parkinson's plus group, including corticobasal syndrome, multiple system atrophy, progressive supranuclear palsy
Progressive supranuclear palsy (PSP) is a late-onset degenerative disease involving the gradual deterioration and death of specific volumes of the brain. The condition leads to symptoms including loss of balance, slowing of movement, difficulty ...
, and dementia with lewy bodies
Dementia with Lewy bodies (DLB) is a type of dementia characterized by changes in sleep, behavior, cognition, movement, and regulation of automatic bodily functions. Memory loss is not always an early symptom. The disease worsens over time an ...
. Differential diagnosis can be narrowed down with careful history and physical (especially focused on onset of specific symptoms), progression of the disease, and response to treatment. Some key features between them:
* Corticobasal syndrome – levodopa-resistance, myoclonus
Myoclonus is a brief, involuntary, irregular (lacking rhythm) twitching of a muscle or a group of muscles, different from clonus, which is rhythmic or regular. Myoclonus (myo "muscle", clonic "jerk") describes a medical sign and, generally, is ...
, dystonia, corticosensory loss, apraxia
Apraxia is a motor disorder caused by damage to the brain (specifically the posterior parietal cortex or corpus callosum), which causes difficulty with motor planning to perform tasks or movements. The nature of the damage determines the disorde ...
, and non-fluent aphasia
Expressive aphasia, also known as Broca's aphasia, is a type of aphasia characterized by partial loss of the ability to produce language ( spoken, manual, or written), although comprehension generally remains intact. A person with expressive aphas ...
* Multiple system atrophy – levodopa resistance, rapidly progressive, autonomic failure, stridor, present Babinski sign
The plantar reflex is a reflex elicited when the sole of the foot is stimulated with a blunt instrument. The reflex can take one of two forms. In healthy adults, the plantar reflex causes a downward response of the hallux (flexion). An upward re ...
, cerebellar ataxia, and specific MRI findings
* Progressive supranuclear palsy
Progressive supranuclear palsy (PSP) is a late-onset degenerative disease involving the gradual deterioration and death of specific volumes of the brain. The condition leads to symptoms including loss of balance, slowing of movement, difficulty ...
– levodopa resistance, restrictive vertical gaze, specific MRI findings, and early and different postural difficulties
* Dementia with Lewy bodies
Dementia with Lewy bodies (DLB) is a type of dementia characterized by changes in sleep, behavior, cognition, movement, and regulation of automatic bodily functions. Memory loss is not always an early symptom. The disease worsens over time an ...
– levodopa resistance, cognitive predominance before motor symptoms, and fluctuating cognitive symptoms, (visual hallucinations are very common in this disease, but PD patients also have them)
* Essential tremor
Essential tremor (ET), also called benign tremor, familial tremor, and idiopathic tremor, is a medical condition characterized by involuntary rhythmic contractions and relaxations ( oscillations or twitching movements) of certain muscle groups in ...
– This can at first look like parkinsonism, but has key differentiators. In essential tremor, the tremor gets worse with action (whereas in PD, it gets better), a lack of other symptoms is common in PD, and normal DatSCAN is seen.
Other conditions that can have similar presentations to PD include:
* Arthritis
* Creutzfeldt–Jakob disease
Creutzfeldt–Jakob disease (CJD), also known as subacute spongiform encephalopathy or neurocognitive disorder due to prion disease, is an invariably fatal degenerative brain disorder. Early symptoms include memory problems, behavioral changes ...
* Dystonia
* Depression
* Fragile X-associated tremor/ataxia syndrome
* Frontotemporal dementia and parkinsonism linked to chromosome 17
* Huntington's disease
* Idiopathic basal ganglia calcification
* Neurodegeneration with brain iron accumulation
* Normal-pressure hydrocephalus
* Obsessional slowness
* Psychogenic parkinsonism
* Wilson's disease
Wilson's disease is a genetic disorder in which excess copper builds up in the body. Symptoms are typically related to the brain and liver. Liver-related symptoms include vomiting, weakness, fluid build up in the abdomen, swelling of the legs, ...
Prevention
Exercise in middle age may reduce the risk of PD later in life.Caffeine
Caffeine is a central nervous system (CNS) stimulant of the methylxanthine class. It is mainly used recreationally as a cognitive enhancer, increasing alertness and attentional performance. Caffeine acts by blocking binding of adenosine to ...
also appears protective with a greater decrease in risk occurring with a larger intake of caffeinated beverages such as coffee.
Antioxidants, such as vitamins C and E, have been proposed to protect against the disease, but results of studies have been contradictory and no positive effect has been shown. The results regarding fat and fatty acids have been contradictory, with various studies reporting protective, risk-increasing, or no effects. There have been preliminary indications that the use of nonsteroidal anti-inflammatory drugs (NSAIDs) and calcium channel blockers may be protective. A 2010 meta-analysis
A meta-analysis is a statistical analysis that combines the results of multiple scientific studies. Meta-analyses can be performed when there are multiple scientific studies addressing the same question, with each individual study reporting me ...
found that NSAIDs (apart from aspirin), have been associated with at least a 15% (higher in long-term and regular users) reduction in the incidence of the development of PD. There is a growing body of evidence linking this neuroprotective effect of NSAIDs in PD but as of 2019 meta-analyses have failed to confirm this link. However, multiple studies have demonstrated a link between the use of ibuprofen and a decreased risk of Parkinson's development.
Management
 No cure for Parkinson's disease is known. Medications, surgery, and physical treatment may provide relief, improve the quality of a person's life, and are much more effective than treatments available for other neurological disorders such as Alzheimer's disease, motor neuron disease, and Parkinson-plus syndromes. The main families of drugs useful for treating motor symptoms are levodopa always combined with a dopa decarboxylase inhibitor and sometimes also with a
No cure for Parkinson's disease is known. Medications, surgery, and physical treatment may provide relief, improve the quality of a person's life, and are much more effective than treatments available for other neurological disorders such as Alzheimer's disease, motor neuron disease, and Parkinson-plus syndromes. The main families of drugs useful for treating motor symptoms are levodopa always combined with a dopa decarboxylase inhibitor and sometimes also with a COMT inhibitor
A catechol-''O''-methyltransferase (COMT) inhibitor is a drug that inhibits the enzyme catechol-''O''-methyltransferase. This enzyme methylates catecholamines such as dopamine, norepinephrine and epinephrine. It also methylates levodopa. COMT in ...
, dopamine agonist
A dopamine agonist (DA) is a compound that activates dopamine receptors. There are two families of dopamine receptors, D2-like and D1-like, and they are all G protein-coupled receptors. D1- and D5-receptors belong to the D1-like family and the ...
s, and MAO-B inhibitors. The stage of the disease and the age at disease onset determine which group is most useful.
Braak staging of PD uses six stages that can identify early, middle, and late stages. The initial stage in which some disability has already developed and requires pharmacological treatment is followed by later stages associated with the development of complications related to levodopa usage, and a third stage when symptoms unrelated to dopamine deficiency or levodopa treatment may predominate.
Treatment in the first stage aims for an optimal trade-off
A trade-off (or tradeoff) is a situational decision that involves diminishing or losing one quality, quantity, or property of a set or design in return for gains in other aspects. In simple terms, a tradeoff is where one thing increases, and anot ...
between symptom control and treatment side effects. The start of levodopa treatment may be postponed by initially using other medications, such as MAO-B inhibitors and dopamine agonists, instead, in the hope of delaying the onset of complications due to levodopa use. Levodopa is still the most effective treatment for the motor symptoms of PD, though, and should not be delayed in people when their quality of life is impaired. Levodopa-related dyskinesias correlate more strongly with duration and severity of the disease than duration of levodopa treatment, so delaying this therapy may not provide much longer dyskinesia-free time than early use.
In later stages, the aim is to reduce PD symptoms, while controlling fluctuations in the effect of the medication. Sudden withdrawals from medication or its overuse must be managed. When oral medications are not enough to control symptoms, surgery, (deep brain stimulation or more recently high-intensity focused ultrasound
High-intensity focused ultrasound (HIFU) is a non-invasive therapeutic technique that uses non-ionizing ultrasonic waves to heat or ablate tissue. HIFU can be used to increase the flow of blood or lymph or to destroy tissue, such as tumors, vi ...
), subcutaneous waking-day apomorphine
Apomorphine, sold under the brand name Apokyn among others, is a type of aporphine having activity as a non- selective dopamine agonist which activates both D2-like and, to a much lesser extent, D1-like receptors. It also acts as an antag ...
infusion, and enteral
Enteral administration is food or drug administration via the human gastrointestinal tract. This contrasts with parenteral nutrition or drug administration (Greek ''para'', "besides" + ''enteros''), which occurs from routes outside the GI tract, ...
dopa pumps may be useful. Late-stage PD presents many challenges requiring a variety of treatments, including those for psychiatric symptoms particularly depression, orthostatic hypotension, bladder dysfunction, and erectile dysfunction. In the final stages of the disease, palliative care is provided to improve a person's quality of life.
A 2020 Cochrane review found no certain evidence that cognitive training is beneficial for people with Parkinson's disease, dementia or mild cognitive impairment. The findings are based on low certainty evidence of seven studies.
Medications
Levodopa
The motor symptoms of PD are the result of reduced dopamine production in the brain's basal ganglia. Dopamine does not cross the blood–brain barrier, so it cannot be taken as a medicine to boost the brain's depleted levels of dopamine, but a precursor of dopamine, levodopa, can pass through to the brain, where it is readily converted to dopamine, and administration of levodopa temporarily diminishes the motor symptoms of PD. Levodopa has been the most widely used PD treatment for over 40 years. Only 5–10% of levodopa crosses the blood–brain barrier. Much of the remainder is metabolized to dopamine elsewhere in the body, causing a variety of side effects, including nausea, vomiting, and orthostatic hypotension.Carbidopa
Carbidopa (Lodosyn) is a drug given to people with Parkinson's disease in order to inhibit peripheral metabolism of levodopa. This property is significant in that it allows a greater proportion of administered levodopa to cross the blood–br ...
and benserazide
Benserazide (also called Serazide or Ro 4-4602) is a peripherally acting aromatic L-amino acid decarboxylase or DOPA decarboxylase inhibitor, which is unable to cross the blood–brain barrier.
It is on the World Health Organization's List of ...
are dopa decarboxylase inhibitors that do not cross the blood–brain barrier and inhibit the conversion of levodopa to dopamine outside the brain, reducing side effects and improving the availability of levodopa for passage into the brain. One of these drugs is usually taken along with levodopa, often combined with levodopa in the same pill.
Levodopa use leads in the long term to the development of complications, such as involuntary movements (dyskinesias) and fluctuations in the effectiveness of the medication. When fluctuations occur, a person can cycle through phases with good response to medication and reduced PD symptoms (on state), and phases with poor response to medication and significant PD symptoms (off state). Using lower doses of levodopa may reduce the risk and severity of these levodopa-induced complications. A former strategy to reduce levodopa-related dyskinesia and fluctuations was to withdraw levodopa medication for some time. This is now discouraged, since it can bring on dangerous side effects such as neuroleptic malignant syndrome
Neuroleptic malignant syndrome (NMS) is a rare but life-threatening reaction that can occur in response to neuroleptic or antipsychotic medication. Symptoms include high fever, confusion, rigid muscles, variable blood pressure, sweating, and fa ...
. Most people with PD eventually need levodopa and later develop levodopa-induced fluctuations and dyskinesias.
Controlled-release (CR) versions of levodopa are available. Older CR levodopa preparations have poor and unreliable absorption and bioavailability
In pharmacology, bioavailability is a subcategory of absorption and is the fraction (%) of an administered drug that reaches the systemic circulation.
By definition, when a medication is administered intravenously, its bioavailability is 100%. Ho ...
and have not demonstrated improved control of PD motor symptoms or a reduction in levodopa-related complications when compared to immediate-release preparations. A newer extended-release levodopa preparation does seem to be more effective in reducing fluctuations, but in many people, problems persist. Intestinal infusions of levodopa (Duodopa) can result in striking improvements in fluctuations compared to oral levodopa when the fluctuations are due to insufficient uptake caused by gastroparesis
Gastroparesis (gastro- from Ancient Greek γαστήρ – gaster, "stomach"; and -paresis, πάρεσις – "partial paralysis"), also called delayed gastric emptying, is a medical disorder consisting of weak muscular contractions (peristalsis) ...
.
Inbrija is an inhaled form of carbidopa-levodopa used when oral medications are not effective.
COMT inhibitors
 During the course of PD, affected people can experience a wearing off phenomenon, where they have a recurrence of symptoms after a dose of levodopa, but right before their next dose. Catechol-O-methyltransferase (COMT) is a protein that degrades levodopa before it can cross the blood–brain barrier and these inhibitors allow for more levodopa to cross. They are normally not used in the management of early symptoms, but can be used in conjunction with levodopa/carbidopa when a person is experiencing the wearing off phenomenon with their motor symptoms.
Three COMT inhibitors are available to treat adults with PD and end-of-dose motor fluctuations –
During the course of PD, affected people can experience a wearing off phenomenon, where they have a recurrence of symptoms after a dose of levodopa, but right before their next dose. Catechol-O-methyltransferase (COMT) is a protein that degrades levodopa before it can cross the blood–brain barrier and these inhibitors allow for more levodopa to cross. They are normally not used in the management of early symptoms, but can be used in conjunction with levodopa/carbidopa when a person is experiencing the wearing off phenomenon with their motor symptoms.
Three COMT inhibitors are available to treat adults with PD and end-of-dose motor fluctuations – opicapone
Opicapone, sold under the brand name Ongentys, is a medication which is administered together with levodopa in people with Parkinson's disease. Opicapone is a catechol-O-methyltransferase (COMT) inhibitor.
The most common side effects are dys ...
, entacapone
Entacapone, sold under the brand name Comtan among others, is a medication commonly used in combination with other medications for the treatment of Parkinson's disease. Entacapone together with levodopa and carbidopa allows levodopa to have a lo ...
, and tolcapone
Tolcapone, sold under the brand name Tasmar, is a medication used to treat Parkinson's disease (PD). It is a selective, potent and reversible nitrocatechol-type inhibitor of the enzyme catechol-''O''-methyltransferase (COMT). It has demonstrat ...
. Tolcapone has been available for several years, but its usefulness is limited by possible liver damage complications, so requires liver-function monitoring. Entacapone and opicapone have not been shown to cause significant alterations to liver function. Licensed preparations of entacapone contain entacapone alone or in combination with carbidopa and levodopa. Opicapone is a once-daily COMT inhibitor.
Dopamine agonists
Severaldopamine agonist
A dopamine agonist (DA) is a compound that activates dopamine receptors. There are two families of dopamine receptors, D2-like and D1-like, and they are all G protein-coupled receptors. D1- and D5-receptors belong to the D1-like family and the ...
s that bind to dopamine receptors in the brain have similar effects to levodopa. These were initially used as a complementary therapy to levodopa for individuals experiencing levodopa complications (on-off fluctuations and dyskinesias); they are now mainly used on their own as first therapy for the motor symptoms of PD with the aim of delaying the initiation of levodopa therapy, thus delaying the onset of levodopa's complications. Dopamine agonists include bromocriptine
Bromocriptine, originally marketed as Parlodel and subsequently under many brand names, is an ergoline derivative and dopamine agonist that is used in the treatment of pituitary tumors, Parkinson's disease, hyperprolactinaemia, neuroleptic ...
, pergolide
Pergolide, sold under the brand name Permax and Prascend (veterinary) among others, is an ergoline-based dopamine receptor agonist used in some countries for the treatment of Parkinson's disease. Parkinson's disease is associated with reduced do ...
, pramipexole, ropinirole
Ropinirole, sold under the brand name Requip among others, is a medication used to treat Parkinson's disease (PD) and restless legs syndrome (RLS). In PD the dose needs to be adjusted to the effect and treatment should not be suddenly stopped. ...
, piribedil
Piribedil (trade names Pronoran, Trivastal Retard, Trastal, Trivastan, Clarium and others) is an antiparkinsonian agent and piperazine derivative which acts as a D2 and D3 receptor agonist. It also has α2-adrenergic antagonist properties.
...
, cabergoline
Cabergoline, sold under the brand name Dostinex among others, is a dopaminergic medication used in the treatment of high prolactin levels, prolactinomas, Parkinson's disease, and for other indications. It is taken by mouth.
Cabergoline is an e ...
, apomorphine
Apomorphine, sold under the brand name Apokyn among others, is a type of aporphine having activity as a non- selective dopamine agonist which activates both D2-like and, to a much lesser extent, D1-like receptors. It also acts as an antag ...
, and lisuride.
Though dopamine agonists are less effective than levodopa at controlling PD motor symptoms, they are usually effective enough to manage these symptoms in the first years of treatment. Dyskinesias due to dopamine agonists are rare in younger people who have PD, but along with other complications, become more common with older age at onset. Thus, dopamine agonists are the preferred initial treatment for younger-onset PD, and levodopa is preferred for older-onset PD.
Dopamine agonists produce significant, although usually mild, side effects, including drowsiness, hallucinations, insomnia, nausea, and constipation. Sometimes, side effects appear even at a minimal clinically effective dose, leading the physician to search for a different drug. Agonists have been related to impulse-control disorders (such as compulsive sexual activity, eating, gambling, and shopping) even more strongly than levodopa. They tend to be more expensive than levodopa.
Apomorphine
Apomorphine, sold under the brand name Apokyn among others, is a type of aporphine having activity as a non- selective dopamine agonist which activates both D2-like and, to a much lesser extent, D1-like receptors. It also acts as an antag ...
, a dopamine agonist, may be used to reduce off periods and dyskinesia in late PD. It is administered only by intermittent injections or continuous subcutaneous infusions. Since secondary effects such as confusion and hallucinations are common, individuals receiving apomorphine treatment should be closely monitored. Two dopamine agonists administered through skin patches ( lisuride and rotigotine
Rotigotine, sold under the brand name Neupro among others, is a dopamine agonist of the non-ergoline class of medications indicated for the treatment of Parkinson's disease and restless legs syndrome. It is formulated as a once-daily transdermal ...
) are useful for people in the initial stages and possibly to control off states in those in advanced states.
MAO-B inhibitors
MAO-B inhibitors ( safinamide, selegiline andrasagiline
Rasagiline (Azilect, Azipron) is an irreversible inhibitor of monoamine oxidase-B used as a monotherapy to treat symptoms in early Parkinson's disease or as an adjunct therapy in more advanced cases.
The racemic form of the drug was invented b ...
) increase the amount of dopamine in the basal ganglia by inhibiting the activity of monoamine oxidase B
Monoamine oxidase B, also known as MAOB, is an enzyme that in humans is encoded by the ''MAOB'' gene.
The protein encoded by this gene belongs to the flavin monoamine oxidase family. It is an enzyme located in the outer mitochondrial membrane. I ...
, an enzyme that breaks down dopamine. They have been found to help alleviate motor symptoms when used as monotherapy (on their own); when used in conjunction with levodopa, they reduce the time spent in the off phase. Selegiline has been shown to delay the need for levodopa commencement, suggesting that it might be neuroprotective and slow the progression of the disease (but this has not been proven). An initial study indicated that selegiline in combination with levodopa increased the risk of death, but this has been refuted.
Common side effects are nausea, dizziness, insomnia, sleepiness, and (in selegiline and rasagiline) orthostatic hypotension. Along with dopamine, MAO-Bs are known to increase serotonin, so care must be taken when used with certain antidepressants due to a potentially dangerous condition known as serotonin syndrome
Serotonin syndrome (SS) is a group of symptoms that may occur with the use of certain serotonergic medications or drugs. The degree of symptoms can range from mild to severe, including a potentiality of death. Symptoms in mild cases include high ...
.
Other drugs
Other drugs such as amantadine andanticholinergic
Anticholinergics (anticholinergic agents) are substances that block the action of the neurotransmitter called acetylcholine (ACh) at synapses in the central and peripheral nervous system.
These agents inhibit the parasympathetic nervous sys ...
s may be useful as treatment of motor symptoms, but the evidence supporting them lacks quality, so they are not first-choice treatments. In addition to motor symptoms, PD is accompanied by a diverse range of symptoms. Several drugs have been used to treat some of these problems. Examples are the use of quetiapine
Quetiapine, sold under the brand name Seroquel among others, is an atypical antipsychotic medication used for the treatment of schizophrenia, bipolar disorder, and major depressive disorder. Despite being widely used as a sleep aid due to its ...
for psychosis, cholinesterase inhibitors for dementia, and modafinil for excessive daytime sleepiness. In 2016, pimavanserin
Pimavanserin (ACP-103; BVF-036), sold under the brand name Nuplazid, is an atypical antipsychotic which is approved for the treatment of Parkinson's disease psychosis and is also being studied for the treatment of Alzheimer’s disease psychos ...
was approved for the management of PD psychosis. Doxepin and rasagline may reduce physical fatigue in PD.
Surgery
 Treating motor symptoms with surgery was once a common practice, but since the discovery of levodopa, the number of operations has declined. Studies in the past few decades have led to great improvements in surgical techniques, so surgery is again being used in people with advanced PD for whom drug therapy is no longer sufficient. Surgery for PD can be divided in two main groups – lesional and
Treating motor symptoms with surgery was once a common practice, but since the discovery of levodopa, the number of operations has declined. Studies in the past few decades have led to great improvements in surgical techniques, so surgery is again being used in people with advanced PD for whom drug therapy is no longer sufficient. Surgery for PD can be divided in two main groups – lesional and deep brain stimulation
Deep brain stimulation (DBS) is a neurosurgical procedure involving the placement of a medical device called a neurostimulator, which sends electrical impulses, through implanted electrodes, to specific targets in the brain (the brain nucleus ...
(DBS). Target areas for DBS or lesions include the thalamus, globus pallidus, or subthalamic nucleus. DBS involves the implantation of a medical device called a neurostimulator, which sends electrical impulses to specific parts of the brain. DBS is recommended for people who have PD with motor fluctuations and tremor inadequately controlled by medication, or to those who are intolerant to medication, as long as they do not have severe neuropsychiatric problems. Other, less common surgical therapies involve intentional formation of lesions to suppress overactivity of specific subcortical areas. For example, pallidotomy
Pallidotomy is a neurosurgical procedure. It is used to treat Parkinson's disease and some other conditions, often as an alternative to deep brain stimulation. It involves placing a tiny electrical probe in the globus pallidus, one of the basal g ...
involves surgical destruction of the globus pallidus to control dyskinesia.
Four areas of the brain have been treated with neural stimulators in PD. These are the globus pallidus interna, thalamus, subthalamic nucleus, and pedunculopontine nucleus. DBS of the globus pallidus interna improves motor function, while DBS of the thalamic DBS improves tremor, but has little effect on bradykinesia or rigidity. DBS of the subthalamic nucleus is usually avoided if a history of depression or neurocognitive impairment is present. DBS of the subthalamic nucleus is associated with a reduction in medication. Pedunculopontine nucleus DBS remains experimental at present. Generally, DBS is associated with 30–60% improvement in motor score evaluations.
Rehabilitation
Exercise programs are recommended in people with PD. Some evidence shows that speech or mobility problems can improve with rehabilitation, although studies are scarce and of low quality. Regular physical exercise with or without physical therapy can be beneficial to maintain and improve mobility, flexibility, strength, gait speed, and quality of life. When an exercise program is performed under the supervision of a physiotherapist, more improvements occur in motor symptoms, mental and emotional functions, daily living activities, and quality of life compared to a self-supervised exercise program at home. Clinical exercises may be an effective intervention targeting overall well-being of individuals with Parkinson's. Improvement in motor function and depression may happen. In improving flexibility and range of motion for people experiencing rigidity, generalized relaxation techniques such as gentle rocking have been found to decrease excessive muscle tension. Other effective techniques to promote relaxation include slow rotational movements of the extremities and trunk, rhythmic initiation, diaphragmatic breathing, and meditation techniques. As for gait and addressing the challenges associated with the disease such as hypokinesia, shuffling, and decreased arm swing, physiotherapists have a variety of strategies to improve functional mobility and safety. Areas of interest concerning gait during rehabilitation programs focus on improving gait speed, the base of support, stride length, and trunk and arm-swing movement. Strategies include using assistive equipment (pole walking and treadmill walking), verbal cueing (manual, visual, and auditory), exercises (marching and PNF patterns), and altering environments (surfaces, inputs, open vs. closed). Strengthening exercises have shown improvements in strength and motor function for people with primary muscular weakness and weakness related to inactivity with mild to moderate PD, but reports show a significant interaction between strength and the time the medications were taken. Therefore, people with PD should perform exercises 45 minutes to one hour after medications when they are at their best. Also, due to the forward flexed posture, and respiratory dysfunctions in advanced PD, deep diaphragmatic breathing exercises are beneficial in improving chest-wall mobility and vital capacity. Exercise may improve constipation. If exercise reduces physical fatigue in PD remains unclear.Strength training
Strength training or resistance training involves the performance of physical exercises that are designed to improve strength and endurance. It is often associated with the lifting of weights. It can also incorporate a variety of training te ...
exercise has been shown to increase manual dexterity in PD patients after exercising with manual putty. This positively affects everyday life when gripping for PD patients.
One of the most widely practiced treatments for speech disorders associated with PD is the Lee Silverman voice treatment (LSVT). Speech therapy and specifically LSVT may improve speech. Occupational therapy (OT) aims to promote health and quality of life by helping people with the disease to participate in as many of their daily living activities as possible. Few studies have been conducted on the effectiveness of OT, and their quality is poor, although with some indication that it may improve motor skills and quality of life for the duration of the therapy.
Palliative care
Palliative care is specialized medical care for people with serious illnesses, including Parkinson's. The goal of this speciality is to improve quality of life for both the person with PD and the family by providing relief from the symptoms, pain, and stress of illnesses. As Parkinson's is not a curable disease, all treatments are focused on slowing decline and improving quality of life, and are therefore palliative in nature. Palliative care should be involved earlier, rather than later, in the disease course. Palliative care specialists can help with physical symptoms, emotional factors such as loss of function and jobs, depression, fear, and existential concerns. Along with offering emotional support to both the affected person and family, palliative care serves an important role in addressing goals of care. People with PD may have many difficult decisions to make as the disease progresses, such as wishes forfeeding tube
Eating (also known as consuming) is the ingestion of food, typically to provide a heterotrophic organism with energy and to allow for growth. Animals and other heterotrophs must eat in order to survive — carnivores eat other animals, herbi ...
, noninvasive ventilator or tracheostomy, wishes for or against cardiopulmonary resuscitation
Cardiopulmonary resuscitation (CPR) is an emergency procedure consisting of chest compressions often combined with artificial ventilation in an effort to manually preserve intact brain function until further measures are taken to restore spon ...
, and when to use hospice care. Palliative-care team members can help answer questions and guide people with PD on these complex and emotional topics to help them make the best decision based on their own values.
Muscles and nerves that control the digestive process may be affected by PD, resulting in constipation and gastroparesis
Gastroparesis (gastro- from Ancient Greek γαστήρ – gaster, "stomach"; and -paresis, πάρεσις – "partial paralysis"), also called delayed gastric emptying, is a medical disorder consisting of weak muscular contractions (peristalsis) ...
(food remaining in the stomach for a longer period than normal). A balanced diet, based on periodical nutritional assessments, is recommended, and should be designed to avoid weight loss or gain and minimize the consequences of gastrointestinal dysfunction. As the disease advances, swallowing difficulties (dysphagia
Dysphagia is difficulty in swallowing. Although classified under "symptoms and signs" in ICD-10, in some contexts it is classified as a condition in its own right.
It may be a sensation that suggests difficulty in the passage of solids or liq ...
) may appear. In such cases, using thickening agents for liquid intake and an upright posture when eating may be useful; both measures reduce the risk of choking. Gastrostomy to deliver food directly into the stomach is possible in severe cases.
Levodopa and proteins use the same transportation system in the intestine and the blood–brain barrier, thereby competing for access. Taking them together results in reduced effectiveness of the drug. Therefore, when levodopa is introduced, excessive protein consumption is discouraged, and a well-balanced Mediterranean diet
The Mediterranean diet is a diet inspired by the eating habits of people who live near the Mediterranean Sea. When initially formulated in the 1960s, it drew on the cuisines of Greece, Italy, France and Spain. In decades since, it has also incor ...
is recommended. In advanced stages, additional intake of low-protein products such as bread or pasta is recommended for similar reasons. To minimize interaction with proteins, levodopa should be taken 30 minutes before meals. At the same time, regimens for PD restrict proteins during breakfast and lunch, allowing protein intake in the evening.
Prognosis
risk factor
In epidemiology, a risk factor or determinant is a variable associated with an increased risk of disease or infection.
Due to a lack of harmonization across disciplines, determinant, in its more widely accepted scientific meaning, is often ...
s. A disease pattern mainly characterized by tremor as opposed to rigidity, though, predicts an improved survival. Death from aspiration pneumonia
Aspiration pneumonia is a type of lung infection that is due to a relatively large amount of material from the stomach or mouth entering the lungs. Signs and symptoms often include fever and cough of relatively rapid onset. Complications may inc ...
is twice as common in individuals with PD as in the healthy population.
In 2016, PD resulted in about 211,000 deaths globally, an increase of 161% since 1990. The overall death rate increased by 19% to 1.81 per 100,000 people during that time.
Epidemiology
History
 Several early sources, including an Egyptian papyrus, an Ayurvedic medical treatise, the Bible, and
Several early sources, including an Egyptian papyrus, an Ayurvedic medical treatise, the Bible, and Galen
Aelius Galenus or Claudius Galenus ( el, Κλαύδιος Γαληνός; September 129 – c. AD 216), often Anglicized as Galen () or Galen of Pergamon, was a Greek physician, surgeon and philosopher in the Roman Empire. Considered to be one ...
's writings, describe symptoms resembling those of PD.. The article mistakenly refers to Job 34:19 instead of Job 33:19. After Galen there are no references unambiguously related to PD until the 17th century. In the 17th and 18th centuries, several authors wrote about elements of the disease, including Sylvius, Gaubius, Hunter and Chomel.
In 1817, an English doctor, James Parkinson, published his essay reporting six cases of paralysis agitans. ''An Essay on the Shaking Palsy'' described the characteristic resting tremor, abnormal posture and gait, paralysis and diminished muscle strength, and the way that the disease progresses over time. Early neurologists who made further additions to the knowledge of the disease include Trousseau, Gowers, Kinnier Wilson and Erb
Erb, ERB or ErB may refer to:
People
* Edgar Rice Burroughs (1875–1950), American author
* Erb of Gwent (–), king of Gwent and Glywysing
Surname
* Abraham Erb (1772–1830), American-born Canadian settler
* Albrecht Erb (1628–1714), Aus ...
, and most notably Jean-Martin Charcot, whose studies between 1868 and 1881 were a landmark in the understanding of the disease. Among other advances, he made the distinction between rigidity, weakness and bradykinesia. He also championed the renaming of the disease in honor of James Parkinson.
In 1912, Frederic Lewy described microscopic particles in affected brains, later named Lewy bodies. In 1919, Konstantin Tretiakoff
Konstantin Nikolaevitch Tretiakoff (; December 26, 1892 – 1958) was a Russian neuropathologist. He was born in Fergana, Uzbekistan, as a son of military physician, who was member of Pierre Bonvalot's first Pamir expedition. He studied medicine ...
reported that the substantia nigra was the main cerebral structure affected, but this finding was not widely accepted until it was confirmed by further studies published by Rolf Hassler in 1938. The underlying biochemical changes in the brain were identified in the 1950s, due largely to the work of Arvid Carlsson on the neurotransmitter dopamine and Oleh Hornykiewicz
Oleh Hornykiewicz (17 November 1926 - 26 May 2020) was an Austrian biochemist.
Life
Oleh Hornykiewicz was born in 1926 in Sykhiw (a district of Lviv), then in Poland (now Ukraine). In 1951, he received his M.D. degree from the University of Vie ...
on its role on PD. In 1997, alpha-synuclein was found to be the main component of Lewy bodies by Spillantini, Trojanowski, Goedert and others.
Anticholinergics and surgery (lesioning of the corticospinal pathway or some of the basal ganglia structures) were the only treatments until the arrival of levodopa, which reduced their use dramatically. Levodopa was first synthesized in 1911 by Casimir Funk
Kazimierz Funk (; February 23, 1884 – November 19, 1967), commonly anglicized as Casimir Funk, was a Polish-American biochemist generally credited with being among the first to formulate (in 1912) the concept of vitamins, which he called "vit ...
, but it received little attention until the mid 20th century. It entered clinical practice in 1967 and brought about a revolution in the management of PD. By the late 1980s deep brain stimulation
Deep brain stimulation (DBS) is a neurosurgical procedure involving the placement of a medical device called a neurostimulator, which sends electrical impulses, through implanted electrodes, to specific targets in the brain (the brain nucleus ...
introduced by Alim Louis Benabid and colleagues at Grenoble
lat, Gratianopolis
, commune status = Prefecture and commune
, image = Panorama grenoble.png
, image size =
, caption = From upper left: Panorama of the city, Grenoble’s cable cars, place Saint- ...
, France, emerged as a possible treatment.
Society and culture
Cost
 The costs of PD to society are high, but precise calculations are difficult due to methodological issues in research and differences between countries. The largest share of direct cost comes from inpatient care and nursing homes, while the share coming from medication is substantially lower. Indirect costs are high, due to reduced productivity and the burden on caregivers. In addition to economic costs, PD reduces quality of life of those with the disease and their caregivers.
A study based on 2017 data estimated the US economic PD burden at $51.9 billion, including direct medical costs of $25.4 billion and $26.5 billion in indirect and non-medical costs. The Medicare program bears the largest share of medical costs, as most PD patients are over age 65. The projected total economic burden surpasses $79 billion by 2037. These findings highlight the need for interventions to reduce PD incidence, delay disease progression, and alleviate symptom burden that may reduce the future economic burden of PD.
The costs of PD to society are high, but precise calculations are difficult due to methodological issues in research and differences between countries. The largest share of direct cost comes from inpatient care and nursing homes, while the share coming from medication is substantially lower. Indirect costs are high, due to reduced productivity and the burden on caregivers. In addition to economic costs, PD reduces quality of life of those with the disease and their caregivers.
A study based on 2017 data estimated the US economic PD burden at $51.9 billion, including direct medical costs of $25.4 billion and $26.5 billion in indirect and non-medical costs. The Medicare program bears the largest share of medical costs, as most PD patients are over age 65. The projected total economic burden surpasses $79 billion by 2037. These findings highlight the need for interventions to reduce PD incidence, delay disease progression, and alleviate symptom burden that may reduce the future economic burden of PD.
Advocacy
The birthday of James Parkinson, 11 April, has been designated as World Parkinson's Day. A red tulip was chosen by international organizations as the symbol of the disease in 2005; it represents the 'James Parkinson' tulip cultivar, registered in 1981 by a Dutch horticulturalist. Advocacy organizations include the National Parkinson Foundation, which has provided more than $180 million in care, research, and support services since 1982, Parkinson's Disease Foundation, which has distributed more than $115 million for research and nearly $50 million for education and advocacy programs since its founding in 1957 by William Black; theAmerican Parkinson Disease Association
The American Parkinson Disease Association (APDA) is a grassroots advocacy organization for sufferers of Parkinson's disease and their families.
Founded in 1961, the APDA aims to "ease the burden" for Americans with Parkinson's disease, as well a ...
, founded in 1961; and the European Parkinson's Disease Association
Parkinson's Europe, formerly the European Parkinson's Disease Association (EPDA), is a non-political, non-religious, and non-profit making organisation concerned with the health and welfare of people living with Parkinson's disease (PD) and thei ...
, founded in 1992.
Notable cases
 Actor Michael J. Fox has PD and has greatly increased the public awareness of the disease. After diagnosis, Fox embraced his Parkinson's in television roles, sometimes acting without medication, to further illustrate the effects of the condition. He has written four autobiographies in which his fight against the disease plays a major role, and appeared before the
Actor Michael J. Fox has PD and has greatly increased the public awareness of the disease. After diagnosis, Fox embraced his Parkinson's in television roles, sometimes acting without medication, to further illustrate the effects of the condition. He has written four autobiographies in which his fight against the disease plays a major role, and appeared before the United States Congress
The United States Congress is the legislature of the federal government of the United States. It is bicameral, composed of a lower body, the House of Representatives, and an upper body, the Senate. It meets in the U.S. Capitol in Washing ...
without medication to illustrate the effects of the disease. The Michael J. Fox Foundation aims to develop a cure for Parkinson's disease. Fox received an honorary doctorate in medicine from Karolinska Institutet
The Karolinska Institute (KI; sv, Karolinska Institutet; sometimes known as the (Royal) Caroline Institute in English) is a research-led medical university in Solna within the Stockholm urban area of Sweden. The Karolinska Institute is consist ...
for his contributions to research in Parkinson's disease.
Professional cyclist and Olympic medalist Davis Phinney
Davis Phinney (born July 10, 1959) is a retired professional road bicycle racer from the United States. He won 328 races in the 1980s and 1990s, a record for an American, including two Tour de France stages. He has worked in media since retiring ...
, who was diagnosed with young-onset Parkinson's at age 40, started the Davis Phinney Foundation
The Davis Phinney Foundation is a Nonprofit organization, non-profit with a mission to help people with Parkinson's disease, Parkinson's live well with the disease. It was founded in 2004 by Davis Phinney, a former professional road bicycle racin ...
in 2004 to support PD research, focusing on quality of life for people with the disease.
Boxer Muhammad Ali showed signs of PD when he was 38, but was not diagnosed until he was 42, and has been called the "world's most famous Parkinson's patient". Whether he had PD or parkinsonism related to boxing is unresolved.
At the time of his suicide in 2014, Robin Williams, the American actor and comedian, had been diagnosed with PD. According to his widow, his autopsy found diffuse Lewy body disease, while the autopsy used the term ''diffuse Lewy body dementia''. Dennis Dickson, a spokesperson for the Lewy Body Dementia Association, clarified the distinction by stating that diffuse Lewy body dementia is more commonly called ''diffuse Lewy body disease'' and refers to the underlying disease process. Ian G. McKeith, professor and researcher of Lewy body dementias, commented that Williams' symptoms and autopsy findings were explained by dementia with Lewy bodies
Dementia with Lewy bodies (DLB) is a type of dementia characterized by changes in sleep, behavior, cognition, movement, and regulation of automatic bodily functions. Memory loss is not always an early symptom. The disease worsens over time an ...
.
Research
As of 2022, no disease-modifying drugs (drugs that target the causes or damage) are approved for Parkinson's, so this is a major focus of Parkinson's research. Active research directions include the search for newanimal model
An animal model (short for animal disease model) is a living, non-human, often genetic-engineered animal used during the research and investigation of human disease, for the purpose of better understanding the disease process without the risk of ha ...
s of the disease and studies of the potential usefulness of gene therapy, stem cell transplants, and neuroprotective
Neuroprotection refers to the relative preservation of neuronal structure and/or function. In the case of an ongoing insult (a neurodegenerative insult) the relative preservation of neuronal integrity implies a reduction in the rate of neuronal los ...
agents. To aid in earlier diagnosis, research criteria for identifying prodromal
In medicine, a prodrome is an early sign or symptom (or set of signs and symptoms) that often indicates the onset of a disease before more diagnostically specific signs and symptoms develop. It is derived from the Greek word ''prodromos'', meaning ...
biomarkers
In biomedical contexts, a biomarker, or biological marker, is a measurable indicator of some biological state or condition. Biomarkers are often measured and evaluated using blood, urine, or soft tissues to examine normal biological processes, pa ...
of the disease have been established.
The role of the gut–brain axis and the gut flora in PD are recognized but the mechanism leading to gastrointestinal symptoms is unclear.
Gene therapy
Gene therapy typically involves the use of a noninfectiousvirus
A virus is a submicroscopic infectious agent that replicates only inside the living cells of an organism. Viruses infect all life forms, from animals and plants to microorganisms, including bacteria and archaea.
Since Dmitri Ivanovsk ...
(i.e., a viral vector such as the adeno-associated virus) to shuttle genetic material into a part of the brain. Several approaches have been tried. These approaches have involved the expression of growth factors to try to prevent damage ( Neurturin – a GDNF-family growth factor), and enzymes such as glutamic acid decarboxylase ( GAD – the enzyme that produces GABA), tyrosine hydroxylase (the enzyme that produces L-DOPA) and catechol-O-methyl transferase
Catechol-''O''-methyltransferase (COMT; ) is one of several enzymes that degrade catecholamines (neurotransmitters such as dopamine, epinephrine, and norepinephrine), catecholestrogens, and various drugs and substances having a catechol struct ...
(COMT – the enzyme that converts L-DOPA to dopamine). There have been no reported safety concerns, but the approaches have largely failed in phase 2 clinical trials. The delivery of GAD showed promise in phase 2 trials in 2011, but whilst effective at improving motor function, was inferior to DBS. Follow-up studies in the same cohort have suggested persistent improvement.
Neuroprotective treatments
A vaccine that primes the human immune system to destroyalpha-synuclein
Alpha-synuclein is a protein that, in humans, is encoded by the ''SNCA'' gene. Alpha-synuclein is a neuronal protein that regulates synaptic vesicle trafficking and subsequent neurotransmitter release.
It is abundant in the brain, while smaller a ...
, PD01A (developed by Austrian company, Affiris), entered clinical trials and a phase 1 report in 2020 suggested safety and tolerability. In 2018, an antibody, PRX002/RG7935, showed preliminary safety evidence in stage I trials supporting continuation to stage II trials.
Cell-based therapies
Since early in the 1980s,fetal
A fetus or foetus (; plural fetuses, feti, foetuses, or foeti) is the unborn offspring that develops from an animal embryo. Following embryonic development the fetal stage of development takes place. In human prenatal development, fetal develo ...
, porcine
The pig (''Sus domesticus''), often called swine, hog, or domestic pig when distinguishing from other members of the genus '' Sus'', is an omnivorous, domesticated, even-toed, hoofed mammal. It is variously considered a subspecies of ''Sus ...
, carotid or retinal tissues have been used in cell transplants, in which dissociated cells are injected into the substantia nigra in the hope that they will incorporate themselves into the brain in a way that replaces the dopamine-producing cells that have been lost. These sources of tissues have been largely replaced by induced pluripotent stem cell
Induced pluripotent stem cells (also known as iPS cells or iPSCs) are a type of pluripotent stem cell that can be generated directly from a somatic cell. The iPSC technology was pioneered by Shinya Yamanaka's lab in Kyoto, Japan, who showed in ...
derived dopaminergic neurons, as this is thought to represent a more feasible source of tissue. Initial evidence showed mesencephalic dopamine-producing cell transplants being beneficial, but double-blind trials
In a blind or blinded experiment, information which may influence the participants of the experiment is withheld until after the experiment is complete. Good blinding can reduce or eliminate experimental biases that arise from a participants' expec ...
to date have not determined a long-term benefit. An additional significant problem was the excess release of dopamine by the transplanted tissue, leading to dyskinesia
Dyskinesia refers to a category of movement disorders that are characterized by involuntary muscle movements, including movements similar to tics or chorea and diminished voluntary movements. Dyskinesia can be anything from a slight tremor of ...
. In 2020, a first in human clinical trial reported the transplantation of induced pluripotent stem cells into the brain of a person with PD.
Pharmaceutical
Ventures have been undertaken to explore antagonists ofadenosine receptor
The adenosine receptors (or P1 receptors) are a class of purinergic G protein-coupled receptors with adenosine as the endogenous ligand. There are four known types of adenosine receptors in humans: A1, A2A, A2B and A3; each is encoded by a di ...
s (specifically A2A) as an avenue for novel drugs for Parkinson's. Of these, istradefylline has emerged as the most successful medication and was approved for medical use in the United States in 2019. It is approved as an add-on treatment to the levodopa/carbidopa regime.
References
External links
* - links to several sites including *Parkinson's disease at Mayo Clinic
*
Parkinson's disease at NHS
* ttps://web.archive.org/web/20061109041052/http://www.wpda.org/ World Parkinson Disease Association
PDGENE – Database for Parkinson's Disease genetic association studies
{{Authority control Aging-associated diseases Ailments of unknown cause Geriatrics Neurodegenerative disorders Cytoskeletal defects Wikipedia medicine articles ready to translate Wikipedia neurology articles ready to translate