Gold on:
[Wikipedia]
[Google]
[Amazon]
Gold is a
 Gold is the most
Gold is the most
 Whereas most metals are gray or silvery white, gold is slightly reddish-yellow. This color is determined by the frequency of plasma oscillations among the metal's valence electrons, in the ultraviolet range for most metals but in the visible range for gold due to relativistic effects affecting the orbitals around gold atoms. Similar effects impart a golden hue to metallic caesium.
Common colored gold alloys include the distinctive eighteen-karat
Whereas most metals are gray or silvery white, gold is slightly reddish-yellow. This color is determined by the frequency of plasma oscillations among the metal's valence electrons, in the ultraviolet range for most metals but in the visible range for gold due to relativistic effects affecting the orbitals around gold atoms. Similar effects impart a golden hue to metallic caesium.
Common colored gold alloys include the distinctive eighteen-karat
 Although gold is the most noble of the
Although gold is the most noble of the 2 Au + 3 F2 -> 2 AuF3
: 2 Au + 3 Cl2 -> 2 AuCl3
: 2 Au + 2 Br2 -> AuBr3 + AuBr
: 2 Au + I2 -> 2 AuI
Gold does not react with sulfur directly, but 2 Au + 6 H2SeO4 -> 00^\circ CAu2(SeO4)3 + 3 H2SeO3 + 3 H2O
: Au + 4HCl + HNO3 -> H uCl4+ NO\uparrow + 2H2O
Gold is similarly unaffected by most bases. It does not react with
 Gold is thought to have been produced in supernova nucleosynthesis, and from the collision of neutron stars, and to have been present in the
Gold is thought to have been produced in supernova nucleosynthesis, and from the collision of neutron stars, and to have been present in the

 The earliest recorded metal employed by humans appears to be gold, which can be found free or " native". Small amounts of natural gold have been found in Spanish caves used during the late Paleolithic period, c. 40,000 BC.
The oldest gold artifacts in the world are from
The earliest recorded metal employed by humans appears to be gold, which can be found free or " native". Small amounts of natural gold have been found in Spanish caves used during the late Paleolithic period, c. 40,000 BC.
The oldest gold artifacts in the world are from  Gold is mentioned in the Amarna letters numbered 19 and 26 from around the 14th century BC.
Gold is mentioned frequently in the Old Testament, starting with Genesis 2:11 (at Havilah), the story of the golden calf, and many parts of the temple including the Menorah and the golden altar. In the
Gold is mentioned in the Amarna letters numbered 19 and 26 from around the 14th century BC.
Gold is mentioned frequently in the Old Testament, starting with Genesis 2:11 (at Havilah), the story of the golden calf, and many parts of the temple including the Menorah and the golden altar. In the  The European exploration of the Americas was fueled in no small part by reports of the gold ornaments displayed in great profusion by Native American peoples, especially in
The European exploration of the Americas was fueled in no small part by reports of the gold ornaments displayed in great profusion by Native American peoples, especially in
Gold leaf MET DP260372.jpg, Minoan jewellery; 2300–2100 BC; various sizes;
 "Gold" is cognate with similar words in many
"Gold" is cognate with similar words in many
Mineral Commodity Report 14 — Gold
Institute of geological and Nuclear sciences Ltd – Retrieved 7 June 2012
 In popular culture gold is a high standard of excellence, often used in awards. Great achievements are frequently rewarded with gold, in the form of gold medals, gold trophies and other decorations. Winners of athletic events and other graded competitions are usually awarded a gold medal. Many awards such as the
In popular culture gold is a high standard of excellence, often used in awards. Great achievements are frequently rewarded with gold, in the form of gold medals, gold trophies and other decorations. Winners of athletic events and other graded competitions are usually awarded a gold medal. Many awards such as the
 According to the
According to the

 Since the 1880s, South Africa has been the source of a large proportion of the world's gold supply, and about 22% of the gold presently accounted is from
Since the 1880s, South Africa has been the source of a large proportion of the world's gold supply, and about 22% of the gold presently accounted is from  In South America, the controversial project Pascua Lama aims at exploitation of rich fields in the high mountains of
In South America, the controversial project Pascua Lama aims at exploitation of rich fields in the high mountains of  During the 19th century,
During the 19th century,
, ''The New York Times'', 24 October 2005 Gold ore dumps are the source of many heavy elements such as cadmium, lead, zinc, copper,
 Gold has been widely used throughout the world as
Gold has been widely used throughout the world as
 , gold is valued at around $42 per gram ($1,300 per troy ounce).
Like other precious metals, gold is measured by troy weight and by grams. The proportion of gold in the alloy is measured by ''
, gold is valued at around $42 per gram ($1,300 per troy ounce).
Like other precious metals, gold is measured by troy weight and by grams. The proportion of gold in the alloy is measured by ''

 Because of the softness of pure (24k) gold, it is usually
Because of the softness of pure (24k) gold, it is usually
World Gold Council Nickel is toxic, and its release from nickel white gold is controlled by legislation in Europe. Palladium-gold alloys are more expensive than those using nickel. High-karat white gold alloys are more resistant to corrosion than are either pure silver or

 * Gold produces a deep, intense red color when used as a coloring agent in
* Gold produces a deep, intense red color when used as a coloring agent in
 * Bulk leach extractable gold, for sampling ores
*
* Bulk leach extractable gold, for sampling ores
*
online
* Bernstein, Peter L. ''The Power of Gold: The History of an Obsession'' (2000
online
* Brands, H.W. ''The Age of Gold: The California Gold Rush and the New American Dream'' (2003
excerpt
* Buranelli, Vincent. ''Gold : an illustrated history'' (1979
online
wide-ranging popular history * Cassel, Gustav. "The restoration of the gold standard." ''Economica'' 9 (1923): 171–185
online
* Eichengreen, Barry. ''Golden Fetters: The Gold Standard and the Great Depression, 1919–1939'' (Oxford UP, 1992). * Ferguson, Niall. ''The Ascent of Money - Financial History of the World'' (2009
online
* Hart, Matthew
Gold: The Race for the World's Most Seductive Metal
''Gold : the race for the world's most seductive metal"], New York: Simon & Schuster, 2013. '' * Johnson, Harry G. "The gold rush of 1968 in retrospect and prospect." ''American Economic Review'' 59.2 (1969): 344–348
online
* Kwarteng, Kwasi. ''War and Gold: A Five-Hundred-Year History of Empires, Adventures, and Debt'' (2014
online
* Vilar, Pierre. '' A History of Gold and Money, 1450 to 1920'' (1960)
online
* Vilches, Elvira. ''New World Gold: Cultural Anxiety and Monetary Disorder in Early Modern Spain'' (2010).
Chemistry in its element podcast
(MP3) from the
Gold
www.rsc.org
at ''
''Getting Gold'' 1898 book
www.lateralscience.co.uk * , www.epa.gov
Gold element information
- rsc.org {{Authority control Chemical elements Transition metals Noble metals Precious metals Cubic minerals Minerals in space group 225 Dental materials Electrical conductors Native element minerals E-number additives Symbols of Alaska Symbols of California Chemical elements with face-centered cubic structure
chemical element
A chemical element is a species of atoms that have a given number of protons in their nuclei, including the pure substance consisting only of that species. Unlike chemical compounds, chemical elements cannot be broken down into simpler sub ...
with the symbol Au (from la, aurum) and atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of every ...
79. This makes it one of the higher atomic number elements that occur naturally. It is a bright, slightly orange-yellow, dense, soft, malleable
Ductility is a mechanical property commonly described as a material's amenability to drawing (e.g. into wire). In materials science, ductility is defined by the degree to which a material can sustain plastic deformation under tensile stres ...
, and ductile
Ductility is a mechanical property commonly described as a material's amenability to drawing (e.g. into wire). In materials science, ductility is defined by the degree to which a material can sustain plastic deformation under tensile stres ...
metal
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
in a pure form. Chemically, gold is a transition metal
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. They are the elements that ca ...
and a group 11 element
Group 11, by modern IUPAC numbering, is a group of chemical elements in the periodic table, consisting of copper (Cu), silver (Ag), and gold (Au), and roentgenium (Rg), although no chemical experiments have yet been carried out to confirm tha ...
. It is one of the least reactive chemical elements and is solid under standard conditions
Standard temperature and pressure (STP) are standard sets of conditions for experimental measurements to be established to allow comparisons to be made between different sets of data. The most used standards are those of the International Union ...
. Gold often occurs in free elemental ( native state), as nuggets or grains, in rocks, veins, and alluvial deposit
Alluvium (from Latin ''alluvius'', from ''alluere'' 'to wash against') is loose clay, silt, sand, or gravel that has been deposited by running water in a stream bed, on a floodplain, in an alluvial fan or beach, or in similar settings. Alluv ...
s. It occurs in a solid solution
A solid solution, a term popularly used for metals, is a homogenous mixture of two different kinds of atoms in solid state and have a single crystal structure. Many examples can be found in metallurgy, geology, and solid-state chemistry. The wor ...
series with the native element silver (as electrum), naturally alloy
An alloy is a mixture of chemical elements of which at least one is a metal. Unlike chemical compounds with metallic bases, an alloy will retain all the properties of a metal in the resulting material, such as electrical conductivity, ductilit ...
ed with other metals like copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
and palladium, and mineral inclusions such as within pyrite. Less commonly, it occurs in minerals as gold compounds, often with tellurium ( gold tellurides).
Gold is resistant to most acids, though it does dissolve in aqua regia (a mixture of nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
and hydrochloric acid
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid
Acid strength is the tendency of an acid, symbol ...
), forming a soluble tetrachloroaurate anion. Gold is insoluble in nitric acid alone, which dissolves silver and base metals, a property long used to refine gold and confirm the presence of gold in metallic substances, giving rise to the term ' acid test'. Gold dissolves in alkaline solutions of cyanide, which are used in mining
Mining is the extraction of valuable minerals or other geological materials from the Earth, usually from an ore body, lode, vein, seam, reef, or placer deposit. The exploitation of these deposits for raw material is based on the economic ...
and electroplating. Gold also dissolves in mercury, forming amalgam alloys, and as the gold acts simply as a solute, this is not a chemical reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the pos ...
.
A relatively rare element, gold is a precious metal that has been used for coinage
Coinage may refer to:
* Coins, standardized as currency
* Neologism, coinage of a new word
* '' COINage'', numismatics magazine
* Tin coinage, a tax on refined tin
* Protologism
''Protologism'' is a term coined in 2003 by the American literary ...
, jewelry, and other arts
The arts are a very wide range of human practices of creative expression, storytelling and cultural participation. They encompass multiple diverse and plural modes of thinking, doing and being, in an extremely broad range of media. Both ...
throughout recorded history
Recorded history or written history describes the historical events that have been recorded in a written form or other documented communication which are subsequently evaluated by historians using the historical method. For broader world his ...
. In the past, a gold standard was often implemented as a monetary policy
Monetary policy is the policy adopted by the monetary authority of a nation to control either the interest rate payable for very short-term borrowing (borrowing by banks from each other to meet their short-term needs) or the money supply, often a ...
. Gold coins ceased to be minted as a circulating currency in the 1930s, and the world gold standard was abandoned for a fiat currency system after the Nixon shock measures of 1971.
In 2020, the world's largest gold producer was China, followed by Russia and Australia. A total of around 201,296 tonne
The tonne ( or ; symbol: t) is a unit of mass equal to 1000 kilograms. It is a non-SI unit accepted for use with SI. It is also referred to as a metric ton to distinguish it from the non-metric units of the short ton ( United State ...
s of gold exists above ground, . This is equal to a cube with each side measuring roughly . The world consumption of new gold produced is about 50% in jewelry, 40% in investments and 10% in industry
Industry may refer to:
Economics
* Industry (economics), a generally categorized branch of economic activity
* Industry (manufacturing), a specific branch of economic activity, typically in factories with machinery
* The wider industrial sector ...
. Gold's high malleability, ductility, resistance to corrosion and most other chemical reactions, and conductivity of electricity have led to its continued use in corrosion-resistant electrical connectors in all types of computerized devices (its chief industrial use). Gold is also used in infrared shielding, production of colored glass
Glass coloring and color marking may be obtained in several ways.
# by the addition of ''coloring ions'',Bernard H. W. S. De Jong, Ruud G. C. Beerkens, Peter A. van Nijnatten: "Glass", in: "Ullmann's Encyclopedia of Industrial Chemistry"; Wiley-VC ...
, gold leafing, and tooth restoration
Dental restoration, dental fillings, or simply fillings are treatments used to restore the function, integrity, and morphology of missing tooth structure resulting from caries or external trauma as well as to the replacement of such structure sup ...
. Certain gold salts are still used as anti-inflammatories in medicine.
Characteristics

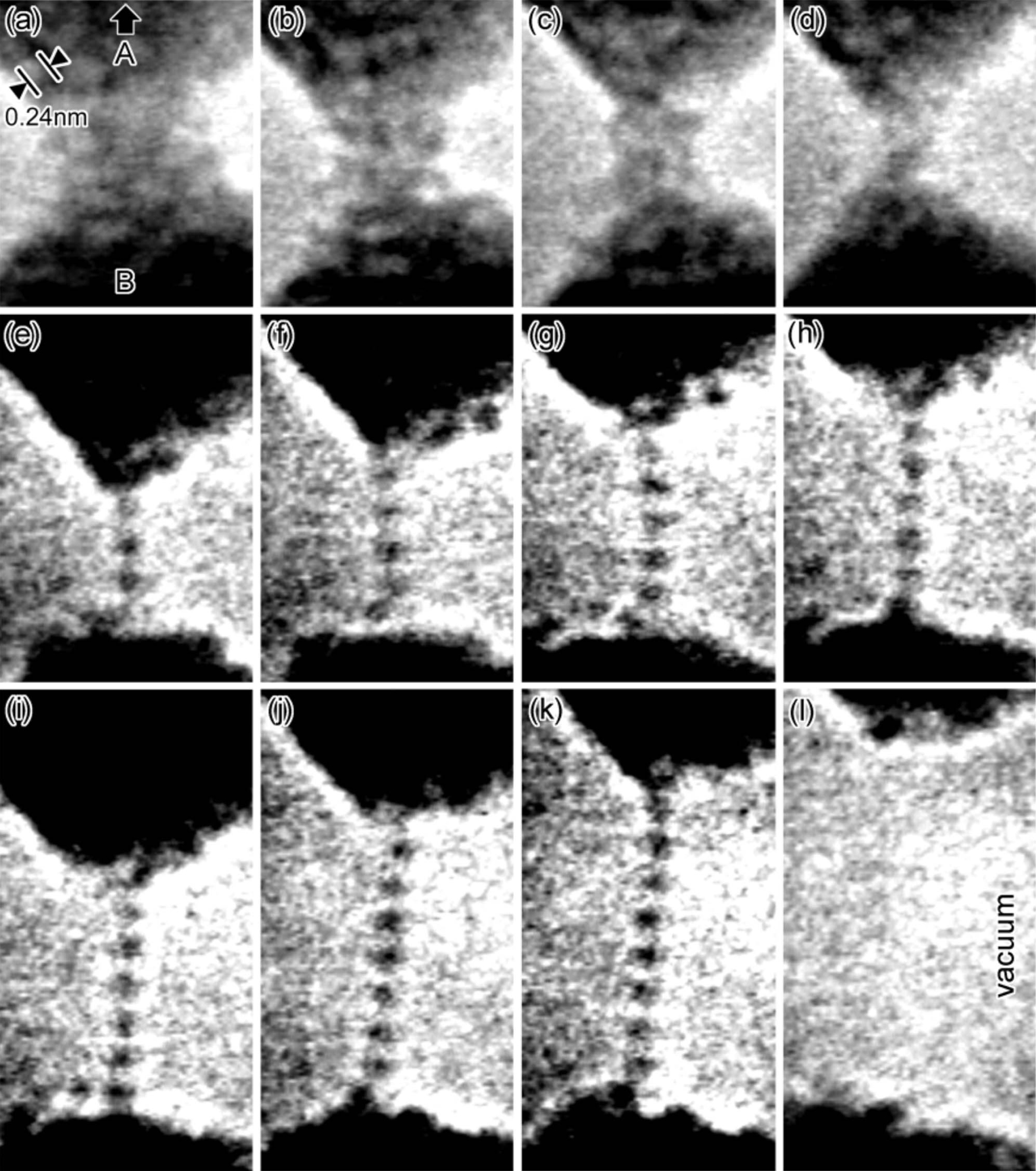
 Gold is the most
Gold is the most malleable
Ductility is a mechanical property commonly described as a material's amenability to drawing (e.g. into wire). In materials science, ductility is defined by the degree to which a material can sustain plastic deformation under tensile stres ...
of all metals. It can be drawn into a wire of single-atom width, and then stretched considerably before it breaks. Such nanowires distort via formation, reorientation and migration of dislocations and crystal twins without noticeable hardening. A single gram of gold can be beaten into a sheet of , and an avoirdupois ounce into . Gold leaf can be beaten thin enough to become semi-transparent. The transmitted light appears greenish-blue, because gold strongly reflects yellow and red. Such semi-transparent sheets also strongly reflect infrared light, making them useful as infrared (radiant heat) shields in visors of heat-resistant suits, and in sun-visors for spacesuits. Gold is a good conductor of heat and electricity
Electricity is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as describ ...
.
Gold has a density of 19.3 g/cm3, almost identical to that of tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isol ...
at 19.25 g/cm3; as such, tungsten has been used in counterfeiting of gold bars, such as by plating a tungsten bar with gold, or taking an existing gold bar, drilling holes, and replacing the removed gold with tungsten rods. By comparison, the density of lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
is 11.34 g/cm3, and that of the densest element, osmium, is .
Color
rose gold
Pure gold is slightly reddish yellow in color, but colored gold in various other colors can be produced by alloying gold with other elements.
Colored golds can be classified in three groups:
* Alloys with silver and copper in various proportion ...
created by the addition of copper. Alloys containing palladium or nickel
Nickel is a chemical element with symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive but large pieces are slow ...
are also important in commercial jewelry as these produce white gold alloys. Fourteen-karat gold-copper alloy is nearly identical in color to certain bronze alloys, and both may be used to produce police and other badge
A badge is a device or accessory, often containing the insignia of an organization, which is presented or displayed to indicate some feat of service, a special accomplishment, a symbol of authority granted by taking an oath (e.g., police and ...
s. Fourteen- and eighteen-karat gold alloys with silver alone appear greenish-yellow and are referred to as green gold. Blue gold can be made by alloying with iron
Iron () is a chemical element with Symbol (chemistry), symbol Fe (from la, Wikt:ferrum, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 element, group 8 of the periodic table. It is, Abundanc ...
, and purple gold can be made by alloying with aluminium
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. I ...
. Less commonly, addition of manganese
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy use ...
, indium
Indium is a chemical element with the symbol In and atomic number 49. Indium is the softest metal that is not an alkali metal. It is a silvery-white metal that resembles tin in appearance. It is a post-transition metal that makes up 0.21 parts ...
, and other elements can produce more unusual colors of gold for various applications.
Colloidal gold, used by electron-microscopists, is red if the particles are small; larger particles of colloidal gold are blue.
Isotopes
Gold has only one stableisotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers (mass numb ...
, , which is also its only naturally occurring isotope, so gold is both a mononuclidic and monoisotopic element
A monoisotopic element is an element which has only a single stable isotope (nuclide). There are only 26 elements that have this property. A list is given in a following section.
Stability is experimentally defined for chemical elements, as ther ...
. Thirty-six radioisotopes
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess nuclear energy, making it unstable. This excess energy can be used in one of three ways: emitted from the nucleus as gamma radiation; transferr ...
have been synthesized, ranging in atomic mass from 169 to 205. The most stable of these is with a half-life of 186.1 days. The least stable is , which decays by proton emission
Proton emission (also known as proton radioactivity) is a rare type of radioactive decay in which a proton is ejected from a nucleus. Proton emission can occur from high-lying excited states in a nucleus following a beta decay, in which case t ...
with a half-life of 30 µs. Most of gold's radioisotopes with atomic masses below 197 decay by some combination of proton emission
Proton emission (also known as proton radioactivity) is a rare type of radioactive decay in which a proton is ejected from a nucleus. Proton emission can occur from high-lying excited states in a nucleus following a beta decay, in which case t ...
, α decay, and β+ decay. The exceptions are , which decays by electron capture, and , which decays most often by electron capture (93%) with a minor β− decay path (7%). All of gold's radioisotopes with atomic masses above 197 decay by β− decay.
At least 32 nuclear isomer
A nuclear isomer is a metastable state of an atomic nucleus, in which one or more nucleons (protons or neutrons) occupy higher energy levels than in the ground state of the same nucleus. "Metastable" describes nuclei whose excited states have ...
s have also been characterized, ranging in atomic mass from 170 to 200. Within that range, only , , , , and do not have isomers. Gold's most stable isomer is with a half-life of 2.27 days. Gold's least stable isomer is with a half-life of only 7 ns. has three decay paths: β+ decay, isomeric transition, and alpha decay. No other isomer or isotope of gold has three decay paths.
Synthesis
The possible production of gold from a more common element, such aslead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
, has long been a subject of human inquiry, and the ancient and medieval discipline of alchemy often focused on it; however, the transmutation of the chemical elements did not become possible until the understanding of nuclear physics
Nuclear physics is the field of physics that studies atomic nuclei and their constituents and interactions, in addition to the study of other forms of nuclear matter.
Nuclear physics should not be confused with atomic physics, which studies the ...
in the 20th century. The first synthesis of gold was conducted by Japanese physicist Hantaro Nagaoka
was a Japanese physicist and a pioneer of Japanese physics during the Meiji period.
Life
Nagaoka was born in Nagasaki, Japan on August 19, 1865 and educated at the University of Tokyo. After graduating with a degree in physics in 1887, Naga ...
, who synthesized gold from mercury in 1924 by neutron bombardment. An American team, working without knowledge of Nagaoka's prior study, conducted the same experiment in 1941, achieving the same result and showing that the isotopes of gold produced by it were all radioactive. In 1980, Glenn Seaborg
Glenn Theodore Seaborg (; April 19, 1912February 25, 1999) was an American chemist whose involvement in the synthesis, discovery and investigation of ten transuranium elements earned him a share of the 1951 Nobel Prize in Chemistry. His work i ...
transmuted several thousand atoms of bismuth into gold at the Lawrence Berkeley Laboratory. Gold can be manufactured in a nuclear reactor, but doing so is highly impractical and would cost far more than the value of the gold that is produced.
Chemistry
 Although gold is the most noble of the
Although gold is the most noble of the noble metal
A noble metal is ordinarily regarded as a metallic chemical element that is generally resistant to corrosion and is usually found in nature in its raw form. Gold, platinum, and the other platinum group metals ( ruthenium, rhodium, palladium, o ...
s, it still forms many diverse compounds. The oxidation state of gold in its compounds ranges from −1 to +5, but Au(I) and Au(III) dominate its chemistry. Au(I), referred to as the aurous ion, is the most common oxidation state with soft ligand
In coordination chemistry, a ligand is an ion or molecule ( functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's elec ...
s such as thioether
In organic chemistry, an organic sulfide (British English sulphide) or thioether is an organosulfur functional group with the connectivity as shown on right. Like many other sulfur-containing compounds, volatile sulfides have foul odors. A su ...
s, thiolate
In organic chemistry, a thiol (; ), or thiol derivative, is any organosulfur compound of the form , where R represents an alkyl or other organic substituent. The functional group itself is referred to as either a thiol group or a sulfhydryl grou ...
s, and organophosphines. Au(I) compounds are typically linear. A good example is , which is the soluble form of gold encountered in mining. The binary gold halides, such as AuCl, form zigzag polymeric chains, again featuring linear coordination at Au. Most drugs based on gold are Au(I) derivatives.
Au(III) (referred to as the auric) is a common oxidation state, and is illustrated by gold(III) chloride
Gold(III) chloride, traditionally called auric chloride, is a compound of gold and chlorine with the molecular formula . The "III" in the name indicates that the gold has an oxidation state of +3, typical for many gold compounds. Gold(III) c ...
, . The gold atom centers in Au(III) complexes, like other d8 compounds, are typically square planar, with chemical bond
A chemical bond is a lasting attraction between atoms or ions that enables the formation of molecules and crystals. The bond may result from the electrostatic force between oppositely charged ions as in ionic bonds, or through the sharing of ...
s that have both covalent and ionic character. Gold(I,III) chloride is also known, an example of a mixed-valence complex
Mixed valence complexes contain an element which is present in more than one oxidation state. Well-known mixed valence compounds include the Creutz–Taube complex, Prussian blue, and molybdenum blue. Many solids are mixed-valency including ...
.
Gold does not react with oxygen at any temperature and, up to 100 °C, is resistant to attack from ozone.
:
:
Some free halogens react with gold. Gold is strongly attacked by fluorine at dull-red heat to form gold(III) fluoride
Gold(III) fluoride, , is an orange solid that sublimes at 300 °C. It is a powerful fluorinating agent.
Preparation
AuF3 can be prepared by reacting AuCl3 with F2 or BrF3.
Structure
The crystal structure of AuF3 consists of spirals ...
. Powdered gold reacts with chlorine at 180 °C to form gold(III) chloride
Gold(III) chloride, traditionally called auric chloride, is a compound of gold and chlorine with the molecular formula . The "III" in the name indicates that the gold has an oxidation state of +3, typical for many gold compounds. Gold(III) c ...
. Gold reacts with bromine at 140 °C to form gold(III) bromide , but reacts only very slowly with iodine to form gold(I) iodide AuI.
: gold(III) sulfide
Gold(III) sulfide or auric sulfide is an inorganic compound with the formula .Auric sulfide has been described as a black and amorphous. Little evidence has been published supporting the existence of macroscopic quantities of this material.
Clai ...
can be made by passing hydrogen sulfide through a dilute solution of gold(III) chloride or chlorauric acid.
Gold readily dissolves in mercury at room temperature to form an amalgam, and forms alloy
An alloy is a mixture of chemical elements of which at least one is a metal. Unlike chemical compounds with metallic bases, an alloy will retain all the properties of a metal in the resulting material, such as electrical conductivity, ductilit ...
s with many other metals at higher temperatures. These alloys can be produced to modify the hardness and other metallurgical properties, to control melting point
The melting point (or, rarely, liquefaction point) of a substance is the temperature at which it changes state from solid to liquid. At the melting point the solid and liquid phase exist in equilibrium. The melting point of a substance depen ...
or to create exotic colors.
Gold is unaffected by most acids. It does not react with hydrofluoric, hydrochloric
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid. It is a component of the gastric acid in the digestiv ...
, hydrobromic, hydriodic, sulfuric, or nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
. It does react with selenic acid, and is dissolved by aqua regia, a 1:3 mixture of nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
and hydrochloric acid
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid
Acid strength is the tendency of an acid, symbol ...
. Nitric acid oxidizes the metal to +3 ions, but only in minute amounts, typically undetectable in the pure acid because of the chemical equilibrium of the reaction. However, the ions are removed from the equilibrium by hydrochloric acid, forming ions, or chloroauric acid, thereby enabling further oxidation.
: aqueous
An aqueous solution is a solution in which the solvent is water. It is mostly shown in chemical equations by appending (aq) to the relevant chemical formula. For example, a solution of table salt, or sodium chloride (NaCl), in water would be re ...
, solid, or molten sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable ...
or potassium hydroxide
Potassium hydroxide is an inorganic compound with the formula K OH, and is commonly called caustic potash.
Along with sodium hydroxide (NaOH), KOH is a prototypical strong base. It has many industrial and niche applications, most of which exp ...
. It does however, react with sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable ...
or potassium cyanide under alkaline conditions when oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as ...
is present to form soluble complexes.
Common oxidation states of gold include +1 (gold(I) or aurous compounds) and +3 (gold(III) or auric compounds). Gold ions in solution are readily reduced and precipitated as metal by adding any other metal as the reducing agent. The added metal is oxidized and dissolves, allowing the gold to be displaced from solution and be recovered as a solid precipitate.
Rare oxidation states
Less common oxidation states of gold include −1, +2, and +5. The −1 oxidation state occurs in aurides, compounds containing the anion. Caesium auride (CsAu), for example, crystallizes in the caesium chloride motif; rubidium, potassium, and tetramethylammonium aurides are also known. Gold has the highest electron affinity of any metal, at 222.8 kJ/mol, making a stable species, analogous to the halides. Gold also has a –1 oxidation state in covalent complexes with thegroup 4 Group 4 may refer to:
*Group 4 element, chemical element classification
*Group 4 (racing), classification for cars in auto racing and rallying
* G4S, formerly Group 4 Securicor, a prominent British security company
*IB Group 4 subjects
The Group 4 ...
transition metals, such as in titanium tetraauride and the analogous zirconium and hafnium compounds. These chemicals are expected to form gold-bridged dimers in a manner similar to titanium(IV) hydride.
Gold(II) compounds are usually diamagnetic
Diamagnetic materials are repelled by a magnetic field; an applied magnetic field creates an induced magnetic field in them in the opposite direction, causing a repulsive force. In contrast, paramagnetic and ferromagnetic materials are attracted ...
with Au–Au bonds such as The evaporation of a solution of in concentrated produces red crystals of gold(II) sulfate, . Originally thought to be a mixed-valence compound, it has been shown to contain cations, analogous to the better-known mercury(I) ion, . A gold(II) complex, the tetraxenonogold(II) cation, which contains xenon as a ligand, occurs in .
Gold pentafluoride, along with its derivative anion, , and its difluorine complex, gold heptafluoride, is the sole example of gold(V), the highest verified oxidation state.
Some gold compounds exhibit '' aurophilic bonding'', which describes the tendency of gold ions to interact at distances that are too long to be a conventional Au–Au bond but shorter than van der Waals bonding. The interaction is estimated to be comparable in strength to that of a hydrogen bond.
Well-defined cluster compounds are numerous. In some cases, gold has a fractional oxidation state. A representative example is the octahedral species .
Origin
Gold production in the universe
 Gold is thought to have been produced in supernova nucleosynthesis, and from the collision of neutron stars, and to have been present in the
Gold is thought to have been produced in supernova nucleosynthesis, and from the collision of neutron stars, and to have been present in the dust
Dust is made of fine particles of solid matter. On Earth, it generally consists of particles in the atmosphere that come from various sources such as soil lifted by wind (an aeolian process), volcanic eruptions, and pollution. Dust in ho ...
from which the Solar System
The Solar System Capitalization of the name varies. The International Astronomical Union, the authoritative body regarding astronomical nomenclature, specifies capitalizing the names of all individual astronomical objects but uses mixed "Solar ...
formed.
Traditionally, gold in the universe is thought to have formed by the r-process (rapid neutron capture) in supernova nucleosynthesis, but more recently it has been suggested that gold and other elements heavier than iron
Iron () is a chemical element with Symbol (chemistry), symbol Fe (from la, Wikt:ferrum, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 element, group 8 of the periodic table. It is, Abundanc ...
may also be produced in quantity by the r-process in the collision
In physics, a collision is any event in which two or more bodies exert forces on each other in a relatively short time. Although the most common use of the word ''collision'' refers to incidents in which two or more objects collide with great fo ...
of neutron stars. In both cases, satellite spectrometers at first only indirectly detected the resulting gold. However, in August 2017, the spectroscopic signatures of heavy elements, including gold, were observed by electromagnetic observatories in the GW170817 neutron star merger event, after gravitational wave detectors confirmed the event as a neutron star merger. Current astrophysical models suggest that this single neutron star merger event generated between 3 and 13 Earth masses of gold. This amount, along with estimations of the rate of occurrence of these neutron star merger events, suggests that such mergers may produce enough gold to account for most of the abundance of this element in the universe.
Asteroid origin theories
Because the Earth was molten when it was formed, almost all of the gold present in the early Earth probably sank into the planetary core. Therefore, most of the gold that is in the Earth's crust and mantle has in one model thought to have been delivered to Earth later, byasteroid impact
An impact event is a collision between astronomical objects causing measurable effects. Impact events have physical consequences and have been found to regularly occur in planetary systems, though the most frequent involve asteroids, comets or me ...
s during the Late Heavy Bombardment
The Late Heavy Bombardment (LHB), or lunar cataclysm, is a hypothesized event thought to have occurred approximately 4.1 to 3.8 billion years (Ga) ago, at a time corresponding to the Neohadean and Eoarchean eras on Earth. According to the hypot ...
, about 4 billion years ago.
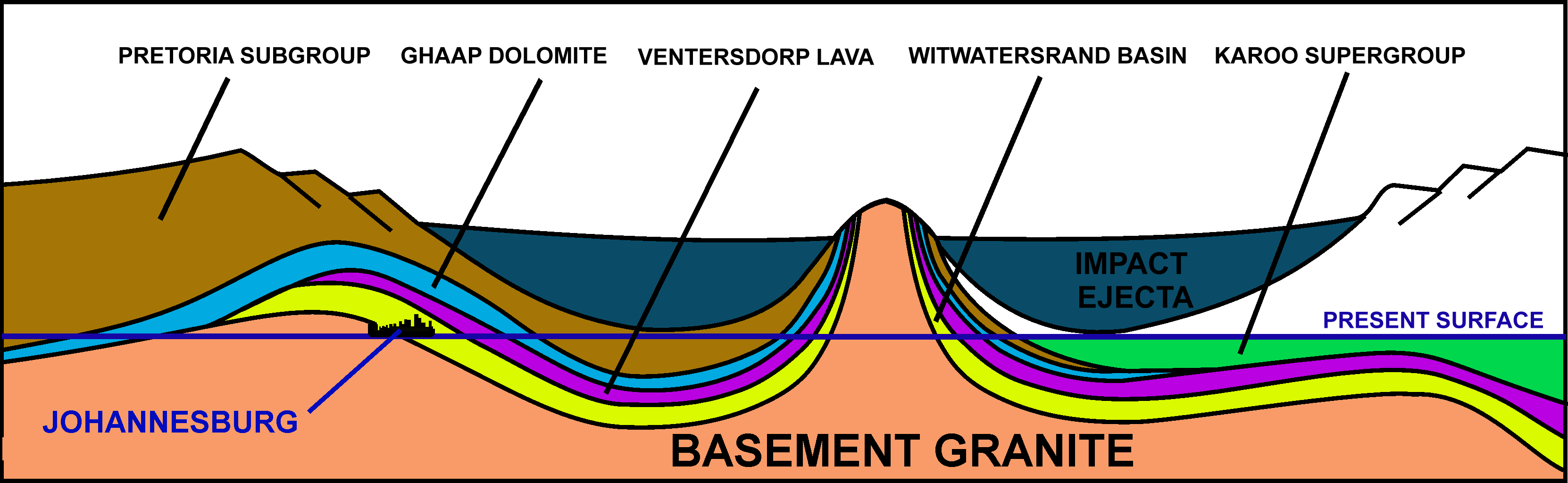
Gold which is reachable by humans has, in one case, been associated with a particular asteroid impact. The asteroid that formed Vredefort impact structure
The Vredefort impact structure is the largest verified impact structure on Earth. The crater, which has since been eroded away, was around across when it was formed. The remaining structure, comprising the deformed underlying bedrock, is loca ...
2.020 billion years ago is often credited with seeding the Witwatersrand basin in South Africa
South Africa, officially the Republic of South Africa (RSA), is the Southern Africa, southernmost country in Africa. It is bounded to the south by of coastline that stretch along the Atlantic Ocean, South Atlantic and Indian Oceans; to the ...
with the richest gold deposits on earth. However, this scenario is now questioned. The gold-bearing Witwatersrand rocks were laid down between 700 and 950 million years before the Vredefort impact.McCarthy, T., Rubridge, B. (2005). ''The Story of Earth and Life''. Struik Publishers, Cape Town. pp. 89–90, 102–107, 134–136. Norman, N., Whitfield, G. (2006) ''Geological Journeys''. Struik Publishers, Cape Town. pp. 38–49, 60–61. These gold-bearing rocks had furthermore been covered by a thick layer of Ventersdorp lavas and the Transvaal Supergroup of rocks before the meteor struck, and thus the gold did not actually arrive in the asteroid/meteorite. What the Vredefort impact achieved, however, was to distort the Witwatersrand basin in such a way that the gold-bearing rocks were brought to the present erosion surface
In geology and geomorphology, an erosion surface is a surface of rock (geology), rock or regolith that was formed by erosion and not by construction (e.g. lava flows, sediment deposition) nor fault (geology), fault displacement. Erosional surfaces ...
in Johannesburg
Johannesburg ( , , ; Zulu and xh, eGoli ), colloquially known as Jozi, Joburg, or "The City of Gold", is the largest city in South Africa, classified as a megacity, and is one of the 100 largest urban areas in the world. According to Dem ...
, on the Witwatersrand, just inside the rim of the original diameter crater caused by the meteor strike. The discovery of the deposit in 1886 launched the Witwatersrand Gold Rush
The Witwatersrand Gold Rush was a gold rush in 1886 that led to the establishment of Johannesburg, South Africa. It was a part of the Mineral Revolution.
Origins
In the modern day province of Mpumalanga, gold miners in the alluvial mines of ...
. Some 22% of all the gold that is ascertained to exist today on Earth has been extracted from these Witwatersrand rocks.
Mantle return theories
Notwithstanding the impact above, much of the rest of the gold on Earth is thought to have been incorporated into the planet since its very beginning, as planetesimals formed the planet's mantle, early in Earth's creation. In 2017, an international group of scientists, established that gold "came to the Earth's surface from the deepest regions of our planet", the mantle, evidenced by their findings at Deseado Massif in the Argentinian Patagonia.Occurrence
On Earth, gold is found inore
Ore is natural rock or sediment that contains one or more valuable minerals, typically containing metals, that can be mined, treated and sold at a profit.Encyclopædia Britannica. "Ore". Encyclopædia Britannica Online. Retrieved 7 Apr ...
s in rock formed from the Precambrian time onward. It most often occurs as a native metal, typically in a metal solid solution
A solid solution, a term popularly used for metals, is a homogenous mixture of two different kinds of atoms in solid state and have a single crystal structure. Many examples can be found in metallurgy, geology, and solid-state chemistry. The wor ...
with silver (i.e. as a gold/silver alloy
An alloy is a mixture of chemical elements of which at least one is a metal. Unlike chemical compounds with metallic bases, an alloy will retain all the properties of a metal in the resulting material, such as electrical conductivity, ductilit ...
). Such alloys usually have a silver content of 8–10%. Electrum is elemental gold with more than 20% silver, and is commonly known as white gold. Electrum's color runs from golden-silvery to silvery, dependent upon the silver content. The more silver, the lower the specific gravity.
Native gold occurs as very small to microscopic particles embedded in rock, often together with quartz
Quartz is a hard, crystalline mineral composed of silica ( silicon dioxide). The atoms are linked in a continuous framework of SiO4 silicon-oxygen tetrahedra, with each oxygen being shared between two tetrahedra, giving an overall chemical ...
or sulfide mineral
The sulfide minerals are a class of minerals containing sulfide (S2−) or disulfide (S22−) as the major anion. Some sulfide minerals are economically important as metal ores. The sulfide class also includes the selenides, the tellurides, th ...
s such as "fool's gold", which is a pyrite. These are called lode
In geology, a lode is a deposit of metalliferous ore that fills or is embedded in a fissure (or crack) in a rock formation or a vein of ore that is deposited or embedded between layers of rock.
The current meaning (ore vein) dates from the 1 ...
deposits. The metal in a native state is also found in the form of free flakes, grains or larger nuggets that have been eroded from rocks and end up in alluvial deposits called placer deposit
In geology, a placer deposit or placer is an accumulation of valuable minerals formed by gravity separation from a specific source rock during sedimentary processes. The name is from the Spanish word ''placer'', meaning "alluvial sand". Placer mi ...
s. Such free gold is always richer at the exposed surface of gold-bearing veins, owing to the oxidation of accompanying minerals followed by weathering; and by washing of the dust into streams and rivers, where it collects and can be welded by water action to form nuggets.
Gold sometimes occurs combined with tellurium as the mineral
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid chemical compound with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed. (2 ...
s calaverite
Calaverite, or gold telluride, is an uncommon telluride of gold, a metallic mineral with the chemical formula AuTe2, with approximately 3% of the gold replaced by silver. It was first discovered in Calaveras County, California in 1861, and was ...
, krennerite
Krennerite is an orthorhombic gold telluride mineral which can contain variable amounts of silver in the structure. The formula is AuTe2, but specimen with gold substituted by up to 24% with silver have been found ( u0.77Ag0.24e2). Both of the c ...
, nagyagite, petzite
The mineral petzite, Ag3 Au Te2, is a soft, steel-gray telluride mineral generally deposited by hydrothermal activity. It forms isometric crystals, and is usually associated with rare tellurium and gold minerals, often with silver, mercury, ...
and sylvanite (see telluride mineral
A telluride mineral is a mineral that has the telluride anion as a main component.
Tellurides are similar to sulfides and are grouped with them in both the Dana and Strunz mineral classification systems.http://webmineral.com/strunz/II.shtml Webm ...
s), and as the rare bismuthide maldonite () and antimonide aurostibite (). Gold also occurs in rare alloys with copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
, lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
, and mercury: the minerals auricupride
Auricupride is a natural alloy that combines copper and gold. Its chemical formula is Cu3Au. The alloy crystallizes in the cubic crystal system in the L12 structure type and occurs as malleable grains or platey masses. It is an opaque yellow w ...
(), novodneprite () and weishanite ().
Recent research suggests that microbes can sometimes play an important role in forming gold deposits, transporting and precipitating gold to form grains and nuggets that collect in alluvial deposits.
Another recent study has claimed water in faults vaporizes during an earthquake, depositing gold. When an earthquake strikes, it moves along a fault. Water often lubricates faults, filling in fractures and jogs. About below the surface, under very high temperatures and pressures, the water carries high concentrations of carbon dioxide, silica, and gold. During an earthquake, the fault jog suddenly opens wider. The water inside the void instantly vaporizes, flashing to steam and forcing silica, which forms the mineral quartz, and gold out of the fluids and onto nearby surfaces.
Seawater
The world'socean
The ocean (also the sea or the world ocean) is the body of salt water that covers approximately 70.8% of the surface of Earth and contains 97% of Earth's water. An ocean can also refer to any of the large bodies of water into which the wo ...
s contain gold. Measured concentrations of gold in the Atlantic and Northeast Pacific are 50–150 femtomol/L or 10–30 parts per quadrillion
Two naming scales for large numbers have been used in English and other European languages since the early modern era: the long and short scales. Most English variants use the short scale today, but the long scale remains dominant in many non-E ...
(about 10–30 g/km3). In general, gold concentrations for south Atlantic and central Pacific samples are the same (~50 femtomol/L) but less certain. Mediterranean deep waters contain slightly higher concentrations of gold (100–150 femtomol/L) attributed to wind-blown dust and/or rivers. At 10 parts per quadrillion the Earth's oceans would hold 15,000 tonnes of gold. These figures are three orders of magnitude less than reported in the literature prior to 1988, indicating contamination problems with the earlier data.
A number of people have claimed to be able to economically recover gold from sea water, but they were either mistaken or acted in an intentional deception. Prescott Jernegan ran a gold-from-seawater swindle in the United States
The United States of America (U.S.A. or USA), commonly known as the United States (U.S. or US) or America, is a country primarily located in North America. It consists of 50 states, a federal district, five major unincorporated territori ...
in the 1890s, as did an English fraudster in the early 1900s. Fritz Haber
Fritz Haber (; 9 December 186829 January 1934) was a German chemist who received the Nobel Prize in Chemistry in 1918 for his invention of the Haber–Bosch process, a method used in industry to synthesize ammonia from nitrogen gas and hydroge ...
did research on the extraction of gold from sea water in an effort to help pay Germany
Germany,, officially the Federal Republic of Germany, is a country in Central Europe. It is the second most populous country in Europe after Russia, and the most populous member state of the European Union. Germany is situated betwe ...
's reparations following World War I
World War I (28 July 1914 11 November 1918), often abbreviated as WWI, was one of the deadliest global conflicts in history. Belligerents included much of Europe, the Russian Empire, the United States, and the Ottoman Empire, with fightin ...
. Based on the published values of 2 to 64 ppb of gold in seawater a commercially successful extraction seemed possible. After analysis of 4,000 water samples yielding an average of 0.004 ppb it became clear that extraction would not be possible and he ended the project.
History

 The earliest recorded metal employed by humans appears to be gold, which can be found free or " native". Small amounts of natural gold have been found in Spanish caves used during the late Paleolithic period, c. 40,000 BC.
The oldest gold artifacts in the world are from
The earliest recorded metal employed by humans appears to be gold, which can be found free or " native". Small amounts of natural gold have been found in Spanish caves used during the late Paleolithic period, c. 40,000 BC.
The oldest gold artifacts in the world are from Bulgaria
Bulgaria (; bg, България, Bǎlgariya), officially the Republic of Bulgaria,, ) is a country in Southeast Europe. It is situated on the eastern flank of the Balkans, and is bordered by Romania to the north, Serbia and North Macedo ...
and are dating back to the 5th millennium BC (4,600 BC to 4,200 BC), such as those found in the Varna Necropolis near Lake Varna and the Black Sea
The Black Sea is a marginal mediterranean sea of the Atlantic Ocean lying between Europe and Asia, east of the Balkans, south of the East European Plain, west of the Caucasus, and north of Anatolia. It is bounded by Bulgaria, Georgia, Rom ...
coast, thought to be the earliest "well-dated" finding of gold artifacts in history. Several prehistoric Bulgarian finds are considered no less old – the golden treasures of Hotnitsa, Durankulak, artifacts from the Kurgan settlement of Yunatsite near Pazardzhik
Pazardzhik ( bg, Пазарджик ) is a city situated along the banks of the Maritsa river, southern Bulgaria. It is the capital of Pazardzhik Province and centre for the homonymous Pazardzhik Municipality.
The Tatars founded Pazardzhik i ...
, the golden treasure Sakar, as well as beads and gold jewelry found in the Kurgan settlement of Provadia
Provadia ( bg, Провадия ) is a town in northeastern Bulgaria, part of Varna Province, located in a deep karst gorge (Provadia syncline) along the Provadiya River not far from the Bulgarian Black Sea Coast. It is the administrative centre ...
– Solnitsata (“salt pit”). However, Varna gold is most often called the oldest since this treasure is the largest and most diverse.
Gold artifacts probably made their first appearance in Ancient Egypt at the very beginning of the pre-dynastic period, at the end of the fifth millennium BC and the start of the fourth, and smelting was developed during the course of the 4th millennium; gold artifacts appear in the archeology of Lower Mesopotamia during the early 4th millennium. As of 1990, gold artifacts found at the Wadi Qana cave cemetery of the 4th millennium BC
The 4th millennium BC spanned the years 4000 BC to 3001 BC. Some of the major changes in human culture during this time included the beginning of the Bronze Age and the invention of writing, which played a major role in starting recorded history. ...
in West Bank
The West Bank ( ar, الضفة الغربية, translit=aḍ-Ḍiffah al-Ġarbiyyah; he, הגדה המערבית, translit=HaGadah HaMaʽaravit, also referred to by some Israelis as ) is a landlocked territory near the coast of the Mediter ...
were the earliest from the Levant. Gold artifacts such as the golden hats and the Nebra disk appeared in Central Europe from the 2nd millennium BC Bronze Age
The Bronze Age is a historic period, lasting approximately from 3300 BC to 1200 BC, characterized by the use of bronze, the presence of writing in some areas, and other early features of urban civilization. The Bronze Age is the second prin ...
.
The oldest known map of a gold mine was drawn in the 19th Dynasty of Ancient Egypt (1320–1200 BC), whereas the first written reference to gold was recorded in the 12th Dynasty around 1900 BC. Egyptian hieroglyphs from as early as 2600 BC describe gold, which King Tushratta of the Mitanni
Mitanni (; Hittite cuneiform ; ''Mittani'' '), c. 1550–1260 BC, earlier called Ḫabigalbat in old Babylonian texts, c. 1600 BC; Hanigalbat or Hani-Rabbat (''Hanikalbat'', ''Khanigalbat'', cuneiform ') in Assyrian records, or ''Naharin'' in ...
claimed was "more plentiful than dirt" in Egypt. Egypt and especially Nubia had the resources to make them major gold-producing areas for much of history. One of the earliest known maps, known as the Turin Papyrus Map, shows the plan of a gold mine Gold Mine may refer to:
* Gold Mine (board game)
*Gold Mine (Long Beach), an arena
*"Gold Mine", a song by Joyner Lucas from the 2020 album ''ADHD
Attention deficit hyperactivity disorder (ADHD) is a neurodevelopmental disorder characteri ...
in Nubia together with indications of the local geology. The primitive working methods are described by both Strabo and Diodorus Siculus, and included fire-setting
Fire-setting is a method of traditional mining used most commonly from Prehistory, prehistoric times up to the Middle Ages. Fires were set against a rock face to heat the Rock (geology), stone, which was then doused with liquid, causing the sto ...
. Large mines were also present across the Red Sea
The Red Sea ( ar, البحر الأحمر - بحر القلزم, translit=Modern: al-Baḥr al-ʾAḥmar, Medieval: Baḥr al-Qulzum; or ; Coptic: ⲫⲓⲟⲙ ⲛ̀ϩⲁϩ ''Phiom Enhah'' or ⲫⲓⲟⲙ ⲛ̀ϣⲁⲣⲓ ''Phiom ǹšari''; ...
in what is now Saudi Arabia
Saudi Arabia, officially the Kingdom of Saudi Arabia (KSA), is a country in Western Asia. It covers the bulk of the Arabian Peninsula, and has a land area of about , making it the fifth-largest country in Asia, the second-largest in the A ...
.
 Gold is mentioned in the Amarna letters numbered 19 and 26 from around the 14th century BC.
Gold is mentioned frequently in the Old Testament, starting with Genesis 2:11 (at Havilah), the story of the golden calf, and many parts of the temple including the Menorah and the golden altar. In the
Gold is mentioned in the Amarna letters numbered 19 and 26 from around the 14th century BC.
Gold is mentioned frequently in the Old Testament, starting with Genesis 2:11 (at Havilah), the story of the golden calf, and many parts of the temple including the Menorah and the golden altar. In the New Testament
The New Testament grc, Ἡ Καινὴ Διαθήκη, transl. ; la, Novum Testamentum. (NT) is the second division of the Christian biblical canon. It discusses the teachings and person of Jesus, as well as events in first-century Chri ...
, it is included with the gifts of the magi in the first chapters of Matthew. The Book of Revelation
The Book of Revelation is the final book of the New Testament (and consequently the final book of the Christian Bible). Its title is derived from the first word of the Koine Greek text: , meaning "unveiling" or "revelation". The Book of ...
21:21 describes the city of New Jerusalem as having streets "made of pure gold, clear as crystal". Exploitation of gold in the south-east corner of the Black Sea
The Black Sea is a marginal mediterranean sea of the Atlantic Ocean lying between Europe and Asia, east of the Balkans, south of the East European Plain, west of the Caucasus, and north of Anatolia. It is bounded by Bulgaria, Georgia, Rom ...
is said to date from the time of Midas
Midas (; grc-gre, Μίδας) was the name of a king in Phrygia with whom several myths became associated, as well as two later members of the Phrygian royal house.
The most famous King Midas is popularly remembered in Greek mythology for his ...
, and this gold was important in the establishment of what is probably the world's earliest coinage in Lydia around 610 BC. The legend of the golden fleece
In Greek mythology, the Golden Fleece ( el, Χρυσόμαλλον δέρας, ''Chrysómallon déras'') is the fleece of the golden-woolled,, ''Khrusómallos''. winged ram, Chrysomallos, that rescued Phrixus and brought him to Colchis, where ...
dating from eighth century BCE may refer to the use of fleeces to trap gold dust from placer deposit
In geology, a placer deposit or placer is an accumulation of valuable minerals formed by gravity separation from a specific source rock during sedimentary processes. The name is from the Spanish word ''placer'', meaning "alluvial sand". Placer mi ...
s in the ancient world. From the 6th or 5th century BC, the Chu (state) circulated the Ying Yuan, one kind of square gold coin.
In Roman metallurgy, new methods for extracting gold on a large scale were developed by introducing hydraulic mining methods, especially in Hispania
Hispania ( la, Hispānia , ; nearly identically pronounced in Spanish, Portuguese, Catalan, and Italian) was the Roman name for the Iberian Peninsula and its provinces. Under the Roman Republic, Hispania was divided into two provinces: Hisp ...
from 25 BC onwards and in Dacia
Dacia (, ; ) was the land inhabited by the Dacians, its core in Transylvania, stretching to the Danube in the south, the Black Sea in the east, and the Tisza in the west. The Carpathian Mountains were located in the middle of Dacia. It ...
from 106 AD onwards. One of their largest mines was at Las Medulas in León, where seven long aqueducts
Aqueduct may refer to:
Structures
*Aqueduct (bridge), a bridge to convey water over an obstacle, such as a ravine or valley
*Navigable aqueduct, or water bridge, a structure to carry navigable waterway canals over other rivers, valleys, railw ...
enabled them to sluice most of a large alluvial deposit. The mines at Roşia Montană in Transylvania
Transylvania ( ro, Ardeal or ; hu, Erdély; german: Siebenbürgen) is a historical and cultural region in Central Europe, encompassing central Romania. To the east and south its natural border is the Carpathian Mountains, and to the west the Ap ...
were also very large, and until very recently, still mined by opencast methods. They also exploited smaller deposits in Britain
Britain most often refers to:
* The United Kingdom, a sovereign state in Europe comprising the island of Great Britain, the north-eastern part of the island of Ireland and many smaller islands
* Great Britain, the largest island in the United King ...
, such as placer and hard-rock deposits at Dolaucothi
The Dolaucothi Gold Mines (; cy, Mwynfeydd Aur Dolaucothi) (), also known as the Ogofau Gold Mine, are ancient Roman surface and underground mines located in the valley of the River Cothi, near Pumsaint, Carmarthenshire, Wales. The gold mi ...
. The various methods they used are well described by Pliny the Elder
Gaius Plinius Secundus (AD 23/2479), called Pliny the Elder (), was a Roman author, naturalist and natural philosopher, and naval and army commander of the early Roman Empire, and a friend of the emperor Vespasian. He wrote the encyclopedic ' ...
in his encyclopedia ''Naturalis Historia
The ''Natural History'' ( la, Naturalis historia) is a work by Pliny the Elder. The largest single work to have survived from the Roman Empire to the modern day, the ''Natural History'' compiles information gleaned from other ancient authors. ...
'' written towards the end of the first century AD.
During Mansa Musa's (ruler of the Mali Empire from 1312 to 1337) hajj to Mecca
Mecca (; officially Makkah al-Mukarramah, commonly shortened to Makkah ()) is a city and administrative center of the Mecca Province of Saudi Arabia, and the holiest city in Islam. It is inland from Jeddah on the Red Sea, in a narrow ...
in 1324, he passed through Cairo
Cairo ( ; ar, القاهرة, al-Qāhirah, ) is the Capital city, capital of Egypt and its largest city, home to 10 million people. It is also part of the List of urban agglomerations in Africa, largest urban agglomeration in Africa, List of ...
in July 1324, and was reportedly accompanied by a camel train
A camel train or caravan is a series of camels carrying passengers and goods on a regular or semi-regular service between points. Despite rarely travelling faster than human walking speed, for centuries camels' ability to withstand harsh condi ...
that included thousands of people and nearly a hundred camels where he gave away so much gold that it depressed the price in Egypt for over a decade, causing high inflation
In economics, inflation is an increase in the general price level of goods and services in an economy. When the general price level rises, each unit of currency buys fewer goods and services; consequently, inflation corresponds to a reduct ...
. A contemporary Arab historian remarked:
 The European exploration of the Americas was fueled in no small part by reports of the gold ornaments displayed in great profusion by Native American peoples, especially in
The European exploration of the Americas was fueled in no small part by reports of the gold ornaments displayed in great profusion by Native American peoples, especially in Mesoamerica
Mesoamerica is a historical region and cultural area in southern North America and most of Central America. It extends from approximately central Mexico through Belize, Guatemala, El Salvador, Honduras, Nicaragua, and northern Costa Rica ...
, Peru
, image_flag = Flag of Peru.svg
, image_coat = Escudo nacional del Perú.svg
, other_symbol = Great Seal of the State
, other_symbol_type = National seal
, national_motto = "Firm and Happy f ...
, Ecuador
Ecuador ( ; ; Quechua: ''Ikwayur''; Shuar: ''Ecuador'' or ''Ekuatur''), officially the Republic of Ecuador ( es, República del Ecuador, which literally translates as "Republic of the Equator"; Quechua: ''Ikwadur Ripuwlika''; Shuar: ' ...
and Colombia. The Aztec
The Aztecs () were a Mesoamerican culture that flourished in central Mexico in the post-classic period from 1300 to 1521. The Aztec people included different ethnic groups of central Mexico, particularly those groups who spoke the Nahuatl ...
s regarded gold as the product of the gods, calling it literally "god excrement" (''teocuitlatl'' in Nahuatl), and after Moctezuma II was killed, most of this gold was shipped to Spain. However, for the indigenous peoples of North America
The Indigenous peoples of the Americas are the inhabitants of the Americas before the arrival of the European settlers in the 15th century, and the ethnic groups who now identify themselves with those peoples.
Many Indigenous peoples of the Am ...
gold was considered useless and they saw much greater value in other minerals
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid chemical compound with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed ...
which were directly related to their utility, such as obsidian, flint
Flint, occasionally flintstone, is a sedimentary cryptocrystalline form of the mineral quartz, categorized as the variety of chert that occurs in chalk or marly limestone. Flint was widely used historically to make stone tools and sta ...
, and slate. El Dorado is applied to a legendary story in which precious stones were found in fabulous abundance along with gold coins. The concept of El Dorado underwent several transformations, and eventually accounts of the previous myth were also combined with those of a legendary lost city. El Dorado, was the term used by the Spanish Empire to describe a mythical tribal chief (zipa) of the Muisca native people in Colombia, who, as an initiation rite, covered himself with gold dust and submerged in Lake Guatavita
Lake Guatavita (Spanish: ''Laguna Guatavita'') is located in the Cordillera Oriental of the Colombian Andes in the municipality of Sesquilé in the Almeidas Province, Cundinamarca department of Colombia, northeast of Bogotá, the capital of ...
. The legends surrounding El Dorado changed over time, as it went from being a man, to a city, to a kingdom, and then finally to an empire.
Beginning in the early modern period, European exploration and colonization of West Africa
West Africa or Western Africa is the westernmost region of Africa. The United Nations defines Western Africa as the 16 countries of Benin, Burkina Faso, Cape Verde, The Gambia, Ghana, Guinea, Guinea-Bissau, Ivory Coast, Liberia, Mali, M ...
was driven in large part by reports of gold deposits in the region, which was eventually referred to by Europeans as the "Gold Coast
Gold Coast may refer to:
Places Africa
* Gold Coast (region), in West Africa, which was made up of the following colonies, before being established as the independent nation of Ghana:
** Portuguese Gold Coast (Portuguese, 1482–1642)
** Dutch G ...
". From the late 15th to early 19th centuries, European trade in the region was primarily focused in gold, along with ivory
Ivory is a hard, white material from the tusks (traditionally from elephants) and teeth of animals, that consists mainly of dentine, one of the physical structures of teeth and tusks. The chemical structure of the teeth and tusks of mammals i ...
and slaves. The gold trade in West Africa was dominated by the Ashanti Empire
The Asante Empire (Asante Twi: ), today commonly called the Ashanti Empire, was an Akan state that lasted between 1701 to 1901, in what is now modern-day Ghana. It expanded from the Ashanti Region to include most of Ghana as well as parts of Iv ...
, who initially traded with the Portuguese
Portuguese may refer to:
* anything of, from, or related to the country and nation of Portugal
** Portuguese cuisine, traditional foods
** Portuguese language, a Romance language
*** Portuguese dialects, variants of the Portuguese language
** Portu ...
before branching out and trading with British
British may refer to:
Peoples, culture, and language
* British people, nationals or natives of the United Kingdom, British Overseas Territories, and Crown Dependencies.
** Britishness, the British identity and common culture
* British English, ...
, French, Spanish
Spanish might refer to:
* Items from or related to Spain:
**Spaniards are a nation and ethnic group indigenous to Spain
**Spanish language, spoken in Spain and many Latin American countries
**Spanish cuisine
Other places
* Spanish, Ontario, Can ...
and Danish merchants. British desires to secure control of West African gold deposits played a role in the Anglo-Ashanti wars of the late 19th century, which saw the Ashanti Empire annexed by Britain.
Gold played a role in western culture, as a cause for desire and of corruption, as told in children's fables such as Rumpelstiltskin
"Rumpelstiltskin" ( ; german: Rumpelstilzchen) is a German fairy tale. It was collected by the Brothers Grimm in the 1812 edition of '' Children's and Household Tales''. The story is about a little imp who spins straw into gold in exchange for a ...
—where Rumpelstiltskin turns hay into gold for the peasant's daughter in return for her child when she becomes a princess—and the stealing of the hen that lays golden eggs in Jack and the Beanstalk.
The top prize at the Olympic Games
The modern Olympic Games or Olympics (french: link=no, Jeux olympiques) are the leading international sporting events featuring summer and winter sports competitions in which thousands of athletes from around the world participate in a vari ...
and many other sports competitions is the gold medal.
75% of the presently accounted for gold has been extracted since 1910, two-thirds since 1950.
One main goal of the alchemists was to produce gold from other substances, such as lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
— presumably by the interaction with a mythical substance called the philosopher's stone
The philosopher's stone or more properly philosophers' stone (Arabic: حجر الفلاسفة, , la, lapis philosophorum), is a mythic alchemical substance capable of turning base metals such as mercury into gold (, from the Greek , "gold", ...
. Trying to produce gold led the alchemists to systematically find out what can be done with substances, and this laid the foundation for today's chemistry, which can produce gold (albeit uneconomically) by using nuclear transmutation. Their symbol for gold was the circle with a point at its center (☉), which was also the astrological
Astrology is a range of divinatory practices, recognized as pseudoscientific since the 18th century, that claim to discern information about human affairs and terrestrial events by studying the apparent positions of celestial objects. Dif ...
symbol and the ancient Chinese character
Chinese characters () are logograms developed for the writing of Chinese. In addition, they have been adapted to write other East Asian languages, and remain a key component of the Japanese writing system where they are known as ''kanj ...
for the Sun
The Sun is the star at the center of the Solar System. It is a nearly perfect ball of hot plasma, heated to incandescence by nuclear fusion reactions in its core. The Sun radiates this energy mainly as light, ultraviolet, and infrared radi ...
.
The Dome of the Rock is covered with an ultra-thin golden glassier. The Sikh Golden temple, the Harmandir Sahib, is a building covered with gold. Similarly the Wat Phra Kaew emerald Buddhist temple (wat
A wat ( km, វត្ត, ; lo, ວັດ, ; th, วัด, ; khb, 「ᩅᨯ᩠ᨰ」(waD+Dha); nod, 「ᩅ᩠ᨯ᩶」 (w+Da2)) is a type of Buddhist temple and Hindu temple in Cambodia, Laos, East Shan State, Yunnan, the Southern Provi ...
) in Thailand
Thailand ( ), historically known as Siam () and officially the Kingdom of Thailand, is a country in Southeast Asia, located at the centre of the Indochinese Peninsula, spanning , with a population of almost 70 million. The country is b ...
has ornamental gold-leafed statues and roofs. Some European king and queen's crowns were made of gold, and gold was used for the bridal crown since antiquity. An ancient Talmudic text circa 100 AD describes Rachel, wife of Rabbi Akiva, receiving a "Jerusalem of Gold" (diadem). A Greek burial crown made of gold was found in a grave circa 370 BC.
Metropolitan Museum of Art
The Metropolitan Museum of Art of New York City, colloquially "the Met", is the largest art museum in the Americas. Its permanent collection contains over two million works, divided among 17 curatorial departments. The main building at 1000 ...
(New York City)
Earrings from Shulgi.JPG, Pair of Sumerian earrings with cuneiform
Cuneiform is a logo-syllabic script that was used to write several languages of the Ancient Middle East. The script was in active use from the early Bronze Age until the beginning of the Common Era. It is named for the characteristic wedge-sh ...
inscriptions; 2093–2046 BC; Sulaymaniyah Museum (Sulaymaniyah
Sulaymaniyah, also spelled as Slemani ( ku, سلێمانی, Silêmanî, ar, السليمانية, as-Sulaymāniyyah), is a city in the east of the Kurdistan Region of Iraq, not far from the Iran–Iraq border. It is surrounded by the Azmar, G ...
, Iraq)
Statuette of Amun MET DT553.jpg, Ancient Egyptian statuette of Amun; 945–715 BC; gold; ; Metropolitan Museum of Art
Anillo de Sheshonq (46627183381).jpg, Ancient Egyptian signet ring; 664–525 BC; gold; diameter: ; British Museum
The British Museum is a public museum dedicated to human history, art and culture located in the Bloomsbury area of London. Its permanent collection of eight million works is among the largest and most comprehensive in existence. It docum ...
(London)
Gold stater MET DP138743.jpg, Ancient Greek
Ancient Greek includes the forms of the Greek language used in ancient Greece and the ancient world from around 1500 BC to 300 BC. It is often roughly divided into the following periods: Mycenaean Greek (), Dark Ages (), the Archaic p ...
stater
The stater (; grc, , , statḗr, weight) was an ancient coin used in various regions of Greece. The term is also used for similar coins, imitating Greek staters, minted elsewhere in ancient Europe.
History
The stater, as a Greek silver curre ...
; 323–315 BC; ; Metropolitan Museum of Art
Gold funerary wreath MET DP257471.jpg, Etruscan funerary wreath; 4th–3rd century BC; length: ; Metropolitan Museum of Art
Gold aureus of Hadrian MET DP104782b.jpg, Roman aureus
The ''aureus'' ( ''aurei'', 'golden', used as a noun) was a gold coin of ancient Rome originally valued at 25 pure silver ''denarii'' (sin. denarius). The ''aureus'' was regularly issued from the 1st century BC to the beginning of the 4th cen ...
of Hadrian; 134–138 AD; 7.4 g; Metropolitan Museum of Art
Lime Container (Poporo) MET DT1262.jpg, Quimbaya lime container; 5th–9th century; gold; height: ; Metropolitan Museum of Art
Byzantium, 11th century - Scyphate - 2001.25 - Cleveland Museum of Art.tif, Byzantine
The Byzantine Empire, also referred to as the Eastern Roman Empire or Byzantium, was the continuation of the Roman Empire primarily in its eastern provinces during Late Antiquity and the Middle Ages, when its capital city was Constantinopl ...
scyphate Scyphate is a term frequently used in numismatics to refer to the concave or "cup-shaped" Byzantine coins of the 11th–14th centuries.
This usage emerged in the 19th century, when the term ''scyphatus'', attested in south Italian documents of the ...
; 1059–1067; diameter: ; Cleveland Museum of Art
The Cleveland Museum of Art (CMA) is an art museum in Cleveland, Ohio, located in the Wade Park District, in the University Circle neighborhood on the city's east side. Internationally renowned for its substantial holdings of Asian and Egyptian ...
(Cleveland
Cleveland ( ), officially the City of Cleveland, is a city in the U.S. state of Ohio and the county seat of Cuyahoga County. Located in the northeastern part of the state, it is situated along the southern shore of Lake Erie, across the U.S. ...
, Ohio
Ohio () is a state in the Midwestern region of the United States. Of the fifty U.S. states, it is the 34th-largest by area, and with a population of nearly 11.8 million, is the seventh-most populous and tenth-most densely populated. The sta ...
, USA)
Double Bat-Head Figure Pendant MET DT935.jpg, Pre-Columbian pendant with two bat-head warriors who carry spears; 11th–16th century; gold; overall: ; from the Chiriqui Province (Panama
Panama ( , ; es, link=no, Panamá ), officially the Republic of Panama ( es, República de Panamá), is a transcontinental country spanning the southern part of North America and the northern part of South America. It is bordered by Co ...
); Metropolitan Museum of Art
Box with scene depicting Roman hero Gaius Mucius Scaevola before the Etruscan king Lars Porsena MET DP170836 (cropped).jpg, English Neoclassical box; 1741; overall: ; Metropolitan Museum of Art
France, 18th century - Scent Bottle - 1916.315 - Cleveland Museum of Art.tif, French Rococo
Rococo (, also ), less commonly Roccoco or Late Baroque, is an exceptionally ornamental and theatrical style of architecture, art and decoration which combines asymmetry, scrolling curves, gilding, white and pastel colours, sculpted moulding, ...
glass bottle mounted in gold; circa 1775; overall: ; Cleveland Museum of Art
Etymology
 "Gold" is cognate with similar words in many
"Gold" is cognate with similar words in many Germanic languages
The Germanic languages are a branch of the Indo-European language family spoken natively by a population of about 515 million people mainly in Europe, North America, Oceania and Southern Africa. The most widely spoken Germanic language, E ...
, deriving via Proto-Germanic *''gulþą'' from Proto-Indo-European *''ǵʰelh₃-'' ("to shine, to gleam; to be yellow or green").
The symbol ''Au'' is from the la, aurum, the Latin word for "gold". The Proto-Indo-European ancestor of ''aurum'' was ''*h₂é-h₂us-o-'', meaning "glow". This word is derived from the same root (Proto-Indo-European ''*h₂u̯es-'' "to dawn") as ''*h₂éu̯sōs'', the ancestor of the Latin word Aurora
An aurora (plural: auroras or aurorae), also commonly known as the polar lights, is a natural light display in Earth's sky, predominantly seen in high-latitude regions (around the Arctic and Antarctic). Auroras display dynamic patterns of bri ...
, "dawn". This etymological relationship is presumably behind the frequent claim in scientific publications that ''aurum'' meant "shining dawn".Christie, A and Brathwaite, R. (Last updated 2 November 2011Mineral Commodity Report 14 — Gold
Institute of geological and Nuclear sciences Ltd – Retrieved 7 June 2012
Culture
 In popular culture gold is a high standard of excellence, often used in awards. Great achievements are frequently rewarded with gold, in the form of gold medals, gold trophies and other decorations. Winners of athletic events and other graded competitions are usually awarded a gold medal. Many awards such as the
In popular culture gold is a high standard of excellence, often used in awards. Great achievements are frequently rewarded with gold, in the form of gold medals, gold trophies and other decorations. Winners of athletic events and other graded competitions are usually awarded a gold medal. Many awards such as the Nobel Prize
The Nobel Prizes ( ; sv, Nobelpriset ; no, Nobelprisen ) are five separate prizes that, according to Alfred Nobel's will of 1895, are awarded to "those who, during the preceding year, have conferred the greatest benefit to humankind." Alfr ...
are made from gold as well. Other award statues and prizes are depicted in gold or are gold plated
Gold plating is a method of depositing a thin layer of gold onto the surface of another metal, most often copper or silver (to make silver-gilt), by chemical or electrochemical plating. This article covers plating methods used in the modern elec ...
(such as the Academy Awards
The Academy Awards, better known as the Oscars, are awards for artistic and technical merit for the American and international film industry. The awards are regarded by many as the most prestigious, significant awards in the entertainment ind ...
, the Golden Globe Awards, the Emmy Awards, the Palme d'Or
The Palme d'Or (; en, Golden Palm) is the highest prize awarded at the Cannes Film Festival. It was introduced in 1955 by the festival's organizing committee. Previously, from 1939 to 1954, the festival's highest prize was the Grand Prix du Fe ...
, and the British Academy Film Awards).
Aristotle
Aristotle (; grc-gre, Ἀριστοτέλης ''Aristotélēs'', ; 384–322 BC) was a Greek philosopher and polymath during the Classical period in Ancient Greece. Taught by Plato, he was the founder of the Peripatetic school of ph ...
in his ethics
Ethics or moral philosophy is a branch of philosophy that "involves systematizing, defending, and recommending concepts of right and wrong behavior".''Internet Encyclopedia of Philosophy'' The field of ethics, along with aesthetics, concer ...
used gold symbolism when referring to what is now known as the golden mean. Similarly, gold is associated with perfect or divine principles, such as in the case of the golden ratio
In mathematics, two quantities are in the golden ratio if their ratio is the same as the ratio of their sum to the larger of the two quantities. Expressed algebraically, for quantities a and b with a > b > 0,
where the Greek letter phi ( ...
and the golden rule. Gold is further associated with the wisdom of aging and fruition. The fiftieth wedding anniversary is golden. A person's most valued or most successful latter years are sometimes considered "golden years". The height of a civilization is referred to as a golden age.
Religion
In some forms of Christianity and Judaism, gold has been associated both with the sacred and evil. In theBook of Exodus
The Book of Exodus (from grc, Ἔξοδος, translit=Éxodos; he, שְׁמוֹת ''Šəmōṯ'', "Names") is the second book of the Bible. It narrates the story of the Exodus, in which the Israelites leave slavery in Biblical Egypt through ...
, the Golden Calf is a symbol of idolatry, while in the Book of Genesis
The Book of Genesis (from Greek ; Hebrew: בְּרֵאשִׁית ''Bəreʾšīt'', "In hebeginning") is the first book of the Hebrew Bible and the Christian Old Testament. Its Hebrew name is the same as its first word, ( "In the beginning" ...
, Abraham was said to be rich in gold and silver, and Moses was instructed to cover the Mercy Seat of the Ark of the Covenant with pure gold. In Byzantine
The Byzantine Empire, also referred to as the Eastern Roman Empire or Byzantium, was the continuation of the Roman Empire primarily in its eastern provinces during Late Antiquity and the Middle Ages, when its capital city was Constantinopl ...
iconography the halos of Christ, Virgin Mary
Mary; arc, ܡܪܝܡ, translit=Mariam; ar, مريم, translit=Maryam; grc, Μαρία, translit=María; la, Maria; cop, Ⲙⲁⲣⲓⲁ, translit=Maria was a first-century Jewish woman of Nazareth, the wife of Joseph and the mother of ...
and the saints are often golden.
In Islam, gold (along with silk
Silk is a natural protein fiber, some forms of which can be woven into textiles. The protein fiber of silk is composed mainly of fibroin and is produced by certain insect larvae to form cocoons. The best-known silk is obtained from the ...
) is often cited as being forbidden for men to wear. Abu Bakr al-Jazaeri, quoting a hadith
Ḥadīth ( or ; ar, حديث, , , , , , , literally "talk" or "discourse") or Athar ( ar, أثر, , literally "remnant"/"effect") refers to what the majority of Muslims believe to be a record of the words, actions, and the silent approva ...
, said that " e wearing of silk and gold are forbidden on the males of my nation, and they are lawful to their women". This, however, has not been enforced consistently throughout history, e.g. in the Ottoman Empire. Further, small gold accents on clothing, such as in embroidery
Embroidery is the craft of decorating fabric or other materials using a needle to apply thread or yarn. Embroidery may also incorporate other materials such as pearls, beads, quills, and sequins. In modern days, embroidery is usually seen ...
, may be permitted.
In ancient Greek religion and mythology
Myth is a folklore genre consisting of narratives that play a fundamental role in a society, such as foundational tales or origin myths. Since "myth" is widely used to imply that a story is not objectively true, the identification of a narra ...
, Theia
In Greek mythology, Theia (; grc, Θεία, Theía, divine, also rendered Thea or Thia), also called Euryphaessa ( grc, Εὐρυφάεσσα) "wide-shining", is one of the twelve Titans, the children of the earth goddess Gaia and the Sky deity ...
was seen as the goddess of gold, silver and other gems.
According to Christopher Columbus, those who had something of gold were in possession of something of great value on Earth and a substance to even help souls to paradise.
Wedding ring
A wedding ring or wedding band is a finger ring that indicates that its wearer is married. It is usually forged from metal, traditionally gold or another precious metal. Rings were used in ancient Rome during marriage, though the modern prac ...
s are typically made of gold. It is long lasting and unaffected by the passage of time and may aid in the ring symbolism of eternal vows before God and the perfection the marriage signifies. In Orthodox Christian wedding ceremonies, the wedded couple is adorned with a golden crown (though some opt for wreaths, instead) during the ceremony, an amalgamation of symbolic rites.
On 24 August 2020, Israel
Israel (; he, יִשְׂרָאֵל, ; ar, إِسْرَائِيل, ), officially the State of Israel ( he, מְדִינַת יִשְׂרָאֵל, label=none, translit=Medīnat Yīsrāʾēl; ), is a country in Western Asia. It is situated ...
i archaeologists discovered a trove of early Islamic gold coins near the central city of Yavne
Yavne ( he, יַבְנֶה) or Yavneh is a city in the Central District of Israel. In many English translations of the Bible, it is known as Jabneh . During Greco-Roman times, it was known as Jamnia ( grc, Ἰαμνία ''Iamníā''; la, Iamnia) ...
. Analysis of the extremely rare collection of 425 gold coins indicated that they were from the late 9th century. Dating to around 1,100 years back, the gold coins were from the Abbasid Caliphate
The Abbasid Caliphate ( or ; ar, الْخِلَافَةُ الْعَبَّاسِيَّة, ') was the third caliphate to succeed the Islamic prophet Muhammad. It was founded by a dynasty descended from Muhammad's uncle, Abbas ibn Abdul-Muttalib ...
.
Production
United States Geological Survey
The United States Geological Survey (USGS), formerly simply known as the Geological Survey, is a scientific agency of the United States government. The scientists of the USGS study the landscape of the United States, its natural resources, ...
in 2016, about of gold has been accounted for, of which 85% remains in active use.
Mining and prospecting

 Since the 1880s, South Africa has been the source of a large proportion of the world's gold supply, and about 22% of the gold presently accounted is from
Since the 1880s, South Africa has been the source of a large proportion of the world's gold supply, and about 22% of the gold presently accounted is from South Africa
South Africa, officially the Republic of South Africa (RSA), is the Southern Africa, southernmost country in Africa. It is bounded to the south by of coastline that stretch along the Atlantic Ocean, South Atlantic and Indian Oceans; to the ...
. Production in 1970 accounted for 79% of the world supply, about 1,480 tonnes. In 2007 China (with 276 tonnes) overtook South Africa as the world's largest gold producer, the first time since 1905 that South Africa had not been the largest.
In 2020, China was the world's leading gold-mining country, followed in order by Russia, Australia, the United States, Canada, and Ghana.
 In South America, the controversial project Pascua Lama aims at exploitation of rich fields in the high mountains of
In South America, the controversial project Pascua Lama aims at exploitation of rich fields in the high mountains of Atacama Desert
The Atacama Desert ( es, Desierto de Atacama) is a desert plateau in South America covering a 1,600 km (990 mi) strip of land on the Pacific coast, west of the Andes Mountains. The Atacama Desert is the driest nonpolar desert in th ...
, at the border between Chile
Chile, officially the Republic of Chile, is a country in the western part of South America. It is the southernmost country in the world, and the closest to Antarctica, occupying a long and narrow strip of land between the Andes to the east a ...
and Argentina
Argentina (), officially the Argentine Republic ( es, link=no, República Argentina), is a country in the southern half of South America. Argentina covers an area of , making it the second-largest country in South America after Brazil, th ...
.
It has been estimated that up to one-quarter of the yearly global gold production originates from artisanal or small scale mining.
The city of Johannesburg
Johannesburg ( , , ; Zulu and xh, eGoli ), colloquially known as Jozi, Joburg, or "The City of Gold", is the largest city in South Africa, classified as a megacity, and is one of the 100 largest urban areas in the world. According to Dem ...
located in South Africa was founded as a result of the Witwatersrand Gold Rush
The Witwatersrand Gold Rush was a gold rush in 1886 that led to the establishment of Johannesburg, South Africa. It was a part of the Mineral Revolution.
Origins
In the modern day province of Mpumalanga, gold miners in the alluvial mines of ...
which resulted in the discovery of some of the largest natural gold deposits in recorded history. The gold fields are confined to the northern and north-western edges of the Witwatersrand basin, which is a thick layer of archean
The Archean Eon ( , also spelled Archaean or Archæan) is the second of four geologic eons of Earth's history, representing the time from . The Archean was preceded by the Hadean Eon and followed by the Proterozoic.
The Earth during the Arc ...
rocks located, in most places, deep under the Free State, Gauteng
Gauteng ( ) is one of the nine provinces of South Africa. The name in Sotho-Tswana languages means 'place of gold'.
Situated on the Highveld, Gauteng is the smallest province by land area in South Africa. Although Gauteng accounts for only ...
and surrounding provinces.Truswell, J.F. (1977). ''The Geological Evolution of South Africa''. pp. 21–28. Purnell, Cape Town. These Witwatersrand rocks are exposed at the surface on the Witwatersrand, in and around Johannesburg, but also in isolated patches to the south-east and south-west of Johannesburg, as well as in an arc around the Vredefort Dome
The Vredefort impact structure is the largest verified impact structure on Earth. The crater, which has since been eroded away, was around across when it was formed. The remaining structure, comprising the deformed underlying bedrock, is loca ...
which lies close to the center of the Witwatersrand basin. From these surface exposures the basin dips extensively, requiring some of the mining to occur at depths of nearly , making them, especially the Savuka and TauTona
The TauTona Mine or Western Deep No.3 Shaft, was a gold mine in South Africa. At approximately deep, it was home to the world's second deepest mining operation, rivalled only by the Mponeng Gold Mine, but was closed in 2018.
The mine was one o ...
mines to the south-west of Johannesburg, the deepest mines on earth. The gold is found only in six areas where archean
The Archean Eon ( , also spelled Archaean or Archæan) is the second of four geologic eons of Earth's history, representing the time from . The Archean was preceded by the Hadean Eon and followed by the Proterozoic.
The Earth during the Arc ...
rivers from the north and north-west formed extensive pebbly Braided river deltas before draining into the "Witwatersrand sea" where the rest of the Witwatersrand sediments were deposited.
The Second Boer War
The Second Boer War ( af, Tweede Vryheidsoorlog, , 11 October 189931 May 1902), also known as the Boer War, the Anglo–Boer War, or the South African War, was a conflict fought between the British Empire and the two Boer Republics (the South ...
of 1899–1901 between the British Empire
The British Empire was composed of the dominions, colonies, protectorates, mandates, and other territories ruled or administered by the United Kingdom and its predecessor states. It began with the overseas possessions and trading posts e ...
and the Afrikaner Boer
Boers ( ; af, Boere ()) are the descendants of the Dutch-speaking Free Burghers of the eastern Cape frontier in Southern Africa during the 17th, 18th, and 19th centuries. From 1652 to 1795, the Dutch East India Company controlled this are ...
s was at least partly over the rights of miners and possession of the gold wealth in South Africa.
 During the 19th century,
During the 19th century, gold rush
A gold rush or gold fever is a discovery of gold—sometimes accompanied by other precious metals and rare-earth minerals—that brings an onrush of miners seeking their fortune. Major gold rushes took place in the 19th century in Australia, New ...
es occurred whenever large gold deposits were discovered. The first documented discovery of gold in the United States was at the Reed Gold Mine
The Reed Gold Mine is located in Midland, Cabarrus County, North Carolina, and is the site of the first documented commercial gold find in the United States. It has been designated a National Historic Landmark because of its importance and ...
near Georgeville, North Carolina in 1803. The first major gold strike in the United States occurred in a small north Georgia town called Dahlonega
The city of Dahlonega () is the county seat of Lumpkin County, Georgia, United States. As of the 2010 census, the city had a population of 5,242, and in 2018 the population was estimated to be 6,884.
Dahlonega is located at the north end of ...
. Further gold rushes occurred in California
California is a state in the Western United States, located along the Pacific Coast. With nearly 39.2million residents across a total area of approximately , it is the most populous U.S. state and the 3rd largest by area. It is also the m ...
, Colorado
Colorado (, other variants) is a state in the Mountain states, Mountain West subregion of the Western United States. It encompasses most of the Southern Rocky Mountains, as well as the northeastern portion of the Colorado Plateau and the wes ...
, the Black Hills
The Black Hills ( lkt, Ȟe Sápa; chy, Moʼȯhta-voʼhonáaeva; hid, awaxaawi shiibisha) is an isolated mountain range rising from the Great Plains of North America in western South Dakota and extending into Wyoming, United States. Black ...
, Otago in New Zealand, a number of locations across Australia, Witwatersrand in South Africa, and the Klondike in Canada.
Grasberg mine
The Grasberg mine has one of the largest gold mining, reserves of gold and copper extraction, copper in the world. It is located in Mimika Regency, Central Papua, Indonesia near Puncak Jaya. It is operated by PT Freeport Indonesia (PTFI, ''s ...
located in Papua, Indonesia
Indonesia, officially the Republic of Indonesia, is a country in Southeast Asia and Oceania between the Indian and Pacific oceans. It consists of over 17,000 islands, including Sumatra, Java, Sulawesi, and parts of Borneo and New Guine ...
is the largest gold mine Gold Mine may refer to:
* Gold Mine (board game)
*Gold Mine (Long Beach), an arena
*"Gold Mine", a song by Joyner Lucas from the 2020 album ''ADHD
Attention deficit hyperactivity disorder (ADHD) is a neurodevelopmental disorder characteri ...
in the world.
Extraction and refining
Gold extraction
Gold extraction refers to the processes required to extract gold from its ores. The great majority of gold is extracted from dilute ores using a combination of chemical processes. About 2000 tons are obtained from the earth annually, plus anoth ...
is most economical in large, easily mined deposits. Ore grades as little as 0.5 parts per million (ppm) can be economical. Typical ore grades in open-pit
Open-pit mining, also known as open-cast or open-cut mining and in larger contexts mega-mining, is a surface mining technique of extracting rock or minerals from the earth from an open-air pit, sometimes known as a borrow.
This form of mining ...
mines are 1–5 ppm; ore grades in underground or hard rock mines are usually at least 3 ppm. Because ore grades of 30 ppm are usually needed before gold is visible to the naked eye, in most gold mines the gold is invisible.
The average gold mining and extraction costs were about $317 per troy ounce in 2007, but these can vary widely depending on mining type and ore quality; global mine production amounted to 2,471.1 tonnes.
After initial production, gold is often subsequently refined industrially by the Wohlwill process which is based on electrolysis or by the Miller process, that is chlorination in the melt. The Wohlwill process results in higher purity, but is more complex and is only applied in small-scale installations. Other methods of assaying and purifying smaller amounts of gold include parting and inquartation as well as cupellation
Cupellation is a refining process in metallurgy where ores or alloyed metals are treated under very high temperatures and have controlled operations to separate noble metals, like gold and silver, from base metals, like lead, copper, zinc, arse ...
, or refining methods based on the dissolution of gold in aqua regia.
As of 2020, the amount of carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
produced in mining a kilogram of gold is 16 tonnes, while recycling a kilogram of gold produces 53 kilograms of equivalent. Approximately 30 percent of the global gold supply is recycled and not mined as of 2020.
Corporations are starting to adopt gold recycling including jewelry companies such as Generation Collection and computer companies including Dell.
Consumption
The consumption of gold produced in the world is about 50% in jewelry, 40% in investments, and 10% in industry. According to theWorld Gold Council
The World Gold Council is the market development organisation for the gold industry. It works across all parts of the industry, from gold mining to investment, with the aim of stimulating and sustaining demand for gold.
They frequently publish ...
, China was the world's largest single consumer of gold in 2013, overtaking India.
Pollution
Gold production is associated with contribution to hazardous pollution. Low-grade gold ore may contain less than one ppm gold metal; such ore is ground and mixed withsodium cyanide
Sodium cyanide is a poisonous compound with the formula Na C N. It is a white, water-soluble solid. Cyanide has a high affinity for metals, which leads to the high toxicity of this salt. Its main application, in gold mining, also exploits its hi ...
to dissolve the gold. Cyanide is a highly poisonous chemical, which can kill living creatures when exposed in minute quantities. Many cyanide spills from gold mines have occurred in both developed and developing countries which killed aquatic life in long stretches of affected rivers. Environmentalists consider these events major environmental disasters. Up to thirty tons of used ore can dumped as waste for producing one troy ounce of gold.Behind gold's glitter, torn lands and pointed questions, ''The New York Times'', 24 October 2005 Gold ore dumps are the source of many heavy elements such as cadmium, lead, zinc, copper,
arsenic
Arsenic is a chemical element with the symbol As and atomic number 33. Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a metalloid. It has various allotropes, ...
, selenium
Selenium is a chemical element with the symbol Se and atomic number 34. It is a nonmetal (more rarely considered a metalloid) with properties that are intermediate between the elements above and below in the periodic table, sulfur and tellurium, ...
and mercury. When sulfide-bearing minerals in these ore dumps are exposed to air and water, the sulfide transforms into sulfuric acid which in turn dissolves these heavy metals facilitating their passage into surface water and ground water. This process is called acid mine drainage
Acid mine drainage, acid and metalliferous drainage (AMD), or acid rock drainage (ARD) is the outflow of acidic water from metal mines or coal mines.
Acid rock drainage occurs naturally within some environments as part of the rock weathering ...
. These gold ore dumps are long-term, highly hazardous wastes second only to nuclear waste
Radioactive waste is a type of hazardous waste that contains radioactive material. Radioactive waste is a result of many activities, including nuclear medicine, nuclear research, nuclear power generation, rare-earth mining, and nuclear weapons ...
dumps.
It was once common to use mercury to recover gold from ore, but today the use of mercury is largely limited to small-scale individual miners. Minute quantities of mercury compounds can reach water bodies, causing heavy metal contamination. Mercury can then enter into the human food chain in the form of methylmercury. Mercury poisoning in humans causes incurable brain function damage and severe retardation.
Gold extraction is also a highly energy-intensive industry, extracting ore from deep mines and grinding the large quantity of ore for further chemical extraction requires nearly 25 kWh of electricity per gram of gold produced.
Monetary use
 Gold has been widely used throughout the world as
Gold has been widely used throughout the world as money
Money is any item or verifiable record that is generally accepted as payment for goods and services and repayment of debts, such as taxes, in a particular country or socio-economic context. The primary functions which distinguish money are as ...
, for efficient indirect exchange (versus barter
In trade, barter (derived from ''baretor'') is a system of exchange in which participants in a transaction directly exchange goods or services for other goods or services without using a medium of exchange, such as money. Economists disti ...
), and to store wealth in hoards. For exchange purposes, mints
A mint or breath mint is a food item often consumed as an after-meal refreshment or before business and social engagements to improve breath odor. Mints are commonly believed to soothe the stomach given their association with natural byproducts ...
produce standardized gold bullion coins
A coin is a small, flat (usually depending on the country or value), round piece of metal or plastic used primarily as a medium of exchange or legal tender. They are standardized in weight, and produced in large quantities at a mint in order t ...
, bars and other units of fixed weight and purity.
The first known coins containing gold were struck in Lydia, Asia Minor, around 600 BC. The '' talent'' coin of gold in use during the periods of Grecian history both before and during the time of the life of Homer weighed between 8.42 and 8.75 grams. From an earlier preference in using silver, European economies re-established the minting of gold as coinage during the thirteenth and fourteenth centuries.
Bills (that mature into gold coin) and gold certificates
Gold certificates were issued by the United States Treasury as a form of representative money from 1865 to 1933. While the United States observed a gold standard, the certificates offered a more convenient way to pay in gold than the use of coin ...
(convertible into gold coin at the issuing bank) added to the circulating stock of gold standard money in most 19th century industrial economies.
In preparation for World War I
World War I (28 July 1914 11 November 1918), often abbreviated as WWI, was one of the deadliest global conflicts in history. Belligerents included much of Europe, the Russian Empire, the United States, and the Ottoman Empire, with fightin ...
the warring nations moved to fractional gold standards, inflating their currencies to finance the war effort.
Post-war, the victorious countries, most notably Britain, gradually restored gold-convertibility, but international flows of gold via bills of exchange remained embargoed; international shipments were made exclusively for bilateral trades or to pay war reparations.
After World War II
World War II or the Second World War, often abbreviated as WWII or WW2, was a world war that lasted from 1939 to 1945. It involved the vast majority of the world's countries—including all of the great powers—forming two opposing ...
gold was replaced by a system of nominally convertible currencies related by fixed exchange rates following the Bretton Woods system. Gold standards and the direct convertibility of currencies to gold have been abandoned by world governments, led in 1971 by the United States' refusal to redeem its dollars in gold. Fiat currency now fills most monetary roles. Switzerland was the last country to tie its currency to gold; this was ended by a referendum in 1999.
Central banks continue to keep a portion of their liquid reserves as gold in some form, and metals exchanges such as the London Bullion Market Association still clear transactions denominated in gold, including future delivery contracts. Today, gold mining
Gold mining is the extraction of gold resources by mining. Historically, mining gold from alluvial deposits used manual separation processes, such as gold panning. However, with the expansion of gold mining to ores that are not on the surface, ...
output is declining. With the sharp growth of economies in the 20th century, and increasing foreign exchange, the world's gold reserves and their trading market have become a small fraction of all markets and fixed exchange rates of currencies to gold have been replaced by floating prices for gold and gold future contract. Though the gold stock grows by only 1% or 2% per year, very little metal is irretrievably consumed. Inventory above ground would satisfy many decades of industrial and even artisan uses at current prices.
The gold proportion (fineness) of alloys is measured by karat
The fineness of a precious metal object (coin, bar, jewelry, etc.) represents the weight of ''fine metal'' therein, in proportion to the total weight which includes alloying base metals and any impurities. Alloy metals are added to increase hardne ...
(k). Pure gold (commercially termed ''fine'' gold) is designated as 24 karat, abbreviated 24k. English gold coins intended for circulation from 1526 into the 1930s were typically a standard 22k alloy called crown gold
Crown gold is a 22 karat (kt) gold alloy used in the crown coin introduced in England in 1526 (by Henry VIII). In this alloy, the proportion of gold is 22 parts out of 24 (91.667% gold)—and is appreciably less prone to wear than the softer 23 k ...
, for hardness (American gold coins for circulation after 1837 contain an alloy of 0.900 fine gold, or 21.6 kt).
Although the prices of some platinum group metals can be much higher, gold has long been considered the most desirable of precious metals, and its value has been used as the standard for many currencies
A currency, "in circulation", from la, currens, -entis, literally meaning "running" or "traversing" is a standardization of money in any form, in use or circulation as a medium of exchange, for example banknotes and coins.
A more general def ...
. Gold has been used as a symbol for purity, value, royalty, and particularly roles that combine these properties. Gold as a sign of wealth and prestige was ridiculed by Thomas More
Sir Thomas More (7 February 1478 – 6 July 1535), venerated in the Catholic Church as Saint Thomas More, was an English lawyer, judge, social philosopher, author, statesman, and noted Renaissance humanist. He also served Henry VIII as Lord ...
in his treatise '' Utopia''. On that imaginary island, gold is so abundant that it is used to make chains for slaves, tableware, and lavatory seats. When ambassadors from other countries arrive, dressed in ostentatious gold jewels and badges, the Utopians mistake them for menial servants, paying homage instead to the most modestly dressed of their party.
The ISO 4217 currency code of gold is XAU. Many holders of gold store it in form of bullion coins or bars as a hedge against inflation
In economics, inflation is an increase in the general price level of goods and services in an economy. When the general price level rises, each unit of currency buys fewer goods and services; consequently, inflation corresponds to a reduct ...
or other economic disruptions, though its efficacy as such has been questioned; historically, it has not proven itself reliable as a hedging instrument. Modern bullion coin
Bullion is non-ferrous metal that has been refined to a high standard of elemental purity. The term is ordinarily applied to bulk metal used in the production of coins and especially to precious metals such as gold and silver. It comes fro ...
s for investment or collector purposes do not require good mechanical wear properties; they are typically fine gold at 24k, although the American Gold Eagle
The American Gold Eagle is an official gold bullion coin of the United States. Authorized under the Gold Bullion Coin Act of 1985, it was first released by the United States Mint in 1986. Because the term "eagle" also is the official United S ...
and the British gold sovereign
The sovereign is a British gold coin with a nominal value of one pound sterling (£1) and contains 0.2354 troy oz of pure gold. Struck since 1817, it was originally a circulating coin that was accepted in Britain and elsewhere in the wor ...
continue to be minted in 22k (0.92) metal in historical tradition, and the South African Krugerrand
The Krugerrand (; ) is a South African coin, first minted on 3 July 1967 to help market South African gold and produced by Rand Refinery and the South African Mint. The name is a compound of ''Paul Kruger'', the former President of the South A ...
, first released in 1967, is also 22k (0.92).
The ''special issue'' Canadian Gold Maple Leaf
The Canadian Gold Maple Leaf (GML) is a gold bullion coin that is issued annually by the Government of Canada. It is produced by the Royal Canadian Mint.
The Gold Maple Leaf is legal tender with a face value of 50 Canadian dollars. The market ...
coin contains the highest purity gold of any bullion coin
Bullion is non-ferrous metal that has been refined to a high standard of elemental purity. The term is ordinarily applied to bulk metal used in the production of coins and especially to precious metals such as gold and silver. It comes fro ...
, at 99.999% or 0.99999, while the ''popular issue'' Canadian Gold Maple Leaf coin has a purity of 99.99%. In 2006, the United States Mint
The United States Mint is a bureau of the Department of the Treasury responsible for producing coinage for the United States to conduct its trade and commerce, as well as controlling the movement of bullion. It does not produce paper money; tha ...
began producing the American Buffalo
American Buffalo may refer to:
*American Buffalo (play), ''American Buffalo'' (play), a play by David Mamet
*American Buffalo (film), ''American Buffalo'' (film), a 1996 film of Mamet's play directed by Michael Corrente
*American Buffalo (coin), a ...
gold bullion coin with a purity of 99.99%. The Australian Gold Kangaroos were first coined in 1986 as the Australian Gold Nugget
The Australian Gold Nugget is a gold bullion coin minted by the Perth Mint. The coins have been minted in denominations of oz, oz, oz, oz, 1 oz, 2 oz, 10 oz, and 1 kg of 24 carat gold. They have legal tender status in Australia and are one ...
but changed the reverse design in 1989. Other modern coins include the Austria
Austria, , bar, Östareich officially the Republic of Austria, is a country in the southern part of Central Europe, lying in the Eastern Alps. It is a federation of nine states, one of which is the capital, Vienna, the most populous ...
n Vienna Philharmonic bullion coin and the Chinese Gold Panda
The Chinese Gold Panda () is a series of gold bullion coins issued by the People's Republic of China. The Official Mint of the People's Republic of China introduced the panda gold bullion coins in 1982. The panda design changes every year (with ...
.
Price
 , gold is valued at around $42 per gram ($1,300 per troy ounce).
Like other precious metals, gold is measured by troy weight and by grams. The proportion of gold in the alloy is measured by ''
, gold is valued at around $42 per gram ($1,300 per troy ounce).
Like other precious metals, gold is measured by troy weight and by grams. The proportion of gold in the alloy is measured by ''karat
The fineness of a precious metal object (coin, bar, jewelry, etc.) represents the weight of ''fine metal'' therein, in proportion to the total weight which includes alloying base metals and any impurities. Alloy metals are added to increase hardne ...
'' (k), with 24 karat (24k) being pure gold (100%), and lower karat numbers proportionally less (18k = 75%). The purity of a gold bar or coin can also be expressed as a decimal figure ranging from 0 to 1, known as the millesimal fineness
The fineness of a precious metal object (coin, bar, jewelry, etc.) represents the weight of ''fine metal'' therein, in proportion to the total weight which includes alloying base metals and any impurities. Alloy metals are added to increase hardne ...
, such as 0.995 being nearly pure.
The price of gold is determined through trading in the gold and derivatives markets, but a procedure known as the Gold Fixing
The London Gold Fixing (or Gold Fix) is the setting of the price of gold that takes place via a dedicated conference line. It was formerly held on the London premises of Nathan Mayer Rothschild & Sons by the members of The London Gold Market Fixi ...
in London
London is the capital and List of urban areas in the United Kingdom, largest city of England and the United Kingdom, with a population of just under 9 million. It stands on the River Thames in south-east England at the head of a estuary dow ...
, originating in September 1919, provides a daily benchmark price to the industry. The afternoon fixing was introduced in 1968 to provide a price when US markets are open.
History
Historically goldcoinage
Coinage may refer to:
* Coins, standardized as currency
* Neologism, coinage of a new word
* '' COINage'', numismatics magazine
* Tin coinage, a tax on refined tin
* Protologism
''Protologism'' is a term coined in 2003 by the American literary ...
was widely used as currency; when paper money
A banknote—also called a bill (North American English), paper money, or simply a note—is a type of negotiable promissory note, made by a bank or other licensed authority, payable to the bearer on demand.
Banknotes were originally issued ...
was introduced, it typically was a receipt redeemable for gold coin or bullion. In a monetary system known as the gold standard, a certain weight
In science and engineering, the weight of an object is the force acting on the object due to gravity.
Some standard textbooks define weight as a vector quantity, the gravitational force acting on the object. Others define weight as a scalar qua ...
of gold was given the name of a unit of currency. For a long period, the United States government set the value of the US dollar so that one troy ounce
Troy weight is a system of units of mass that originated in 15th-century England, and is primarily used in the precious metals industry. The troy weight units are the grain, the pennyweight (24 grains), the troy ounce (20 pennyweights), and th ...
was equal to $20.67 ($0.665 per gram), but in 1934 the dollar was devalued to $35.00 per troy ounce ($0.889/g). By 1961, it was becoming hard to maintain this price, and a pool of US and European banks agreed to manipulate the market to prevent further currency devaluation against increased gold demand.
On 17 March 1968, economic circumstances caused the collapse of the gold pool, and a two-tiered pricing scheme was established whereby gold was still used to settle international accounts at the old $35.00 per troy ounce ($1.13/g) but the price of gold on the private market was allowed to fluctuate; this two-tiered pricing system was abandoned in 1975 when the price of gold was left to find its free-market level. Central bank
A central bank, reserve bank, or monetary authority is an institution that manages the currency and monetary policy of a country or monetary union,
and oversees their commercial banking system. In contrast to a commercial bank, a central b ...
s still hold historical gold reserves as a store of value
A store of value is any commodity or asset that would normally retain purchasing power into the future and is the function of the asset that can be saved, retrieved and exchanged at a later time, and be predictably useful when retrieved.
The mos ...
although the level has generally been declining. The largest gold depository in the world is that of the U.S. Federal Reserve Bank in New York, which holds about 3% of the gold known to exist and accounted for today, as does the similarly laden U.S. Bullion Depository at Fort Knox.
In 2005 the World Gold Council
The World Gold Council is the market development organisation for the gold industry. It works across all parts of the industry, from gold mining to investment, with the aim of stimulating and sustaining demand for gold.
They frequently publish ...
estimated total global gold supply to be 3,859 tonnes and demand to be 3,754 tonnes, giving a surplus of 105 tonnes.
After 15 August 1971 Nixon shock, the price began to greatly increase, and between 1968 and 2000 the price of gold ranged widely, from a high of $850 per troy ounce ($27.33/g) on 21 January 1980, to a low of $252.90 per troy ounce ($8.13/g) on 21 June 1999 (London Gold Fixing). Prices increased rapidly from 2001, but the 1980 high was not exceeded until 3 January 2008, when a new maximum of $865.35 per troy ounce
Troy weight is a system of units of mass that originated in 15th-century England, and is primarily used in the precious metals industry. The troy weight units are the grain, the pennyweight (24 grains), the troy ounce (20 pennyweights), and th ...
was set. Another record price was set on 17 March 2008, at $1023.50 per troy ounce ($32.91/g).
In late 2009, gold markets experienced renewed momentum upwards due to increased demand and a weakening US dollar. On 2 December 2009, gold reached a new high closing at $1,217.23. Gold further rallied hitting new highs in May 2010 after the European Union debt crisis prompted further purchase of gold as a safe asset. On 1 March 2011, gold hit a new all-time high of $1432.57, based on investor
An investor is a person who allocates financial capital with the expectation of a future return (profit) or to gain an advantage (interest). Through this allocated capital most of the time the investor purchases some species of property. Type ...
concerns regarding ongoing unrest in North Africa
North Africa, or Northern Africa is a region encompassing the northern portion of the African continent. There is no singularly accepted scope for the region, and it is sometimes defined as stretching from the Atlantic shores of Mauritania in ...
as well as in the Middle East
The Middle East ( ar, الشرق الأوسط, ISO 233: ) is a geopolitical region commonly encompassing Arabia (including the Arabian Peninsula and Bahrain), Asia Minor (Asian part of Turkey except Hatay Province), East Thrace (Europ ...
.
From April 2001 to August 2011, spot gold prices more than quintupled in value against the US dollar, hitting a new all-time high of $1,913.50 on 23 August 2011, prompting speculation that the long secular bear market had ended and a bull market
A market trend is a perceived tendency of financial markets to move in a particular direction over time. Analysts classify these trends as ''secular'' for long time-frames, ''primary'' for medium time-frames, and ''secondary'' for short time-fram ...
had returned. However, the price then began a slow decline towards $1200 per troy ounce in late 2014 and 2015.
In August 2020, the gold price picked up to US$2060 per ounce after a complexive growth of 59% from August 2018 to October 2020, a period during which it outplaced the Nasdaq total return of 54%.
Gold futures are traded on the COMEX exchange. These contacts are priced in USD per troy ounce (1 troy ounce = 31.1034768 grams). Below are the CQG contract specifications outlining the futures contracts:
Medicinal uses
Medicinal applications of gold and its complexes have a long history dating back thousands of years. Several gold complexes have been applied to treatrheumatoid arthritis
Rheumatoid arthritis (RA) is a long-term autoimmune disorder that primarily affects joints. It typically results in warm, swollen, and painful joints. Pain and stiffness often worsen following rest. Most commonly, the wrist and hands are invol ...
, the most frequently used being aurothiomalate, aurothioglucose
Aurothioglucose, also known as gold thioglucose, is a chemical compound with the formula AuSC6H11O5. This derivative of the sugar glucose was formerly used to treat rheumatoid arthritis.
History
Throughout history, gold was used to cure diseases ...
, and auranofin
Auranofin is a gold salt classified by the World Health Organization as an antirheumatic agent. It has the brand name Ridaura.
Use
Auranofin is used to treat rheumatoid arthritis. It improves arthritis symptoms including painful or tender and ...
. Both gold(I) and gold(III) compounds have been investigated as possible anti-cancer drugs. For gold(III) complexes, reduction to gold(0/I) under physiological conditions has to be considered. Stable complexes can be generated using different types of bi-, tri-, and tetradentate ligand systems, and their efficacy has been demonstrated in vitro and in vivo.
Other applications
Jewelry

 Because of the softness of pure (24k) gold, it is usually
Because of the softness of pure (24k) gold, it is usually alloy
An alloy is a mixture of chemical elements of which at least one is a metal. Unlike chemical compounds with metallic bases, an alloy will retain all the properties of a metal in the resulting material, such as electrical conductivity, ductilit ...
ed with base metals for use in jewelry, altering its hardness and ductility, melting point, color and other properties. Alloys with lower karat rating, typically 22k, 18k, 14k or 10k, contain higher percentages of copper or other base metals or silver or palladium in the alloy.Jewellery AlloysWorld Gold Council Nickel is toxic, and its release from nickel white gold is controlled by legislation in Europe. Palladium-gold alloys are more expensive than those using nickel. High-karat white gold alloys are more resistant to corrosion than are either pure silver or
sterling silver
Sterling silver is an alloy of silver containing 92.5% by weight of silver and 7.5% by weight of other metals, usually copper. The sterling silver standard has a minimum millesimal fineness of 925.
'' Fine silver'', which is 99.9% pure silver, i ...
. The Japanese craft of Mokume-gane
is a Japanese metalworking procedure which produces a mixed-metal laminate with distinctive layered patterns; the term is also used to refer to the resulting laminate itself. The term translates closely to "wood grain metal" or "wood eye metal" ...
exploits the color contrasts between laminated colored gold alloys to produce decorative wood-grain effects.
By 2014, the gold jewelry industry was escalating despite a dip in gold prices. Demand in the first quarter of 2014 pushed turnover to $23.7 billion according to a World Gold Council
The World Gold Council is the market development organisation for the gold industry. It works across all parts of the industry, from gold mining to investment, with the aim of stimulating and sustaining demand for gold.
They frequently publish ...
report.
Gold solder
Solder (; NA: ) is a fusible metal alloy used to create a permanent bond between metal workpieces. Solder is melted in order to wet the parts of the joint, where it adheres to and connects the pieces after cooling. Metals or alloys suitable ...
is used for joining the components of gold jewelry by high-temperature hard soldering or brazing. If the work is to be of hallmark
A hallmark is an official mark or series of marks struck on items made of metal, mostly to certify the content of noble metals—such as platinum, gold, silver and in some nations, palladium. In a more general sense, the term '' hallmark'' can a ...
ing quality, the gold solder alloy must match the fineness (purity) of the work, and alloy formulas are manufactured to color-match yellow and white gold. Gold solder is usually made in at least three melting-point ranges referred to as Easy, Medium and Hard. By using the hard, high-melting point solder first, followed by solders with progressively lower melting points, goldsmiths can assemble complex items with several separate soldered joints. Gold can also be made into thread and used in embroidery
Embroidery is the craft of decorating fabric or other materials using a needle to apply thread or yarn. Embroidery may also incorporate other materials such as pearls, beads, quills, and sequins. In modern days, embroidery is usually seen ...
.
Electronics
Only 10% of the world consumption of new gold produced goes to industry, but by far the most important industrial use for new gold is in fabrication of corrosion-free electrical connectors in computers and other electrical devices. For example, according to the World Gold Council, a typical cell phone may contain 50 mg of gold, worth about 2 dollars 82 cents. But since nearly one billion cell phones are produced each year, a gold value of US$2.82 in each phone adds to US$2.82 billion in gold from just this application. (Prices updated to November 2022) Though gold is attacked by free chlorine, its good conductivity and general resistance to oxidation and corrosion in other environments (including resistance to non-chlorinated acids) has led to its widespread industrial use in the electronic era as a thin-layer coating on electrical connectors, thereby ensuring good connection. For example, gold is used in the connectors of the more expensive electronics cables, such as audio, video and USB cables. The benefit of using gold over other connector metals such as tin in these applications has been debated; gold connectors are often criticized by audio-visual experts as unnecessary for most consumers and seen as simply a marketing ploy. However, the use of gold in other applications in electronic sliding contacts in highly humid or corrosive atmospheres, and in use for contacts with a very high failure cost (certain computers, communications equipment,spacecraft
A spacecraft is a vehicle or machine designed to fly in outer space. A type of artificial satellite, spacecraft are used for a variety of purposes, including communications, Earth observation, meteorology, navigation, space colonization, p ...
, jet aircraft engines) remains very common.
Besides sliding electrical contacts, gold is also used in electrical contacts because of its resistance to corrosion
Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials (usually a metal) by chemical or electrochemical reaction with their environment. Corrosion engi ...
, electrical conductivity, ductility
Ductility is a mechanical property commonly described as a material's amenability to drawing (e.g. into wire). In materials science, ductility is defined by the degree to which a material can sustain plastic deformation under tensile str ...
and lack of toxicity. Switch contacts are generally subjected to more intense corrosion stress than are sliding contacts. Fine gold wires are used to connect semiconductor devices to their packages through a process known as wire bonding
Wire bonding is the method of making interconnections between an integrated circuit (IC) or other semiconductor device and its packaging during semiconductor device fabrication. Although less common, wire bonding can be used to connect an IC ...
.
The concentration of free electrons in gold metal is 5.91×1022 cm−3. Gold is highly conductive
In physics and electrical engineering, a conductor is an object or type of material that allows the flow of charge (electric current) in one or more directions. Materials made of metal are common electrical conductors. Electric current is gene ...
to electricity, and has been used for electrical wiring in some high-energy applications (only silver and copper are more conductive per volume, but gold has the advantage of corrosion resistance). For example, gold electrical wires were used during some of the Manhattan Project
The Manhattan Project was a research and development undertaking during World War II that produced the first nuclear weapons. It was led by the United States with the support of the United Kingdom and Canada. From 1942 to 1946, the project w ...
's atomic experiments, but large high-current silver wires were used in the calutron isotope separator magnets in the project.
It is estimated that 16% of the world's presently-accounted-for gold and 22% of the world's silver is contained in electronic technology in Japan.
Medicine
Metallic and gold compounds have long been used for medicinal purposes. Gold, usually as the metal, is perhaps the most anciently administered medicine (apparently by shamanic practitioners) and known toDioscorides
Pedanius Dioscorides ( grc-gre, Πεδάνιος Διοσκουρίδης, ; 40–90 AD), “the father of pharmacognosy”, was a Greek physician, pharmacologist, botanist, and author of '' De materia medica'' (, On Medical Material) —a 5-vo ...
. In medieval times, gold was often seen as beneficial for the health, in the belief that something so rare and beautiful could not be anything but healthy. Even some modern esotericists and forms of alternative medicine assign metallic gold a healing power.
In the 19th century gold had a reputation as an anxiolytic
An anxiolytic (; also antipanic or antianxiety agent) is a medication or other intervention that reduces anxiety. This effect is in contrast to anxiogenic agents which increase anxiety. Anxiolytic medications are used for the treatment of anxiet ...
, a therapy for nervous disorders. Depression, epilepsy
Epilepsy is a group of non-communicable neurological disorders characterized by recurrent epileptic seizures. Epileptic seizures can vary from brief and nearly undetectable periods to long periods of vigorous shaking due to abnormal electrica ...
, migraine, and glandular problems such as amenorrhea
Amenorrhea is the absence of a menstrual period in a woman of reproductive age. Physiological states of amenorrhoea are seen, most commonly, during pregnancy and lactation (breastfeeding). Outside the reproductive years, there is absence of menses ...
and impotence
Erectile dysfunction (ED), also called impotence, is the type of sexual dysfunction in which the penis fails to become or stay erect during sexual activity. It is the most common sexual problem in men.Cunningham GR, Rosen RC. Overview of mal ...
were treated, and most notably alcoholism
Alcoholism is, broadly, any drinking of alcohol that results in significant mental or physical health problems. Because there is disagreement on the definition of the word ''alcoholism'', it is not a recognized diagnostic entity. Predomi ...
(Keeley, 1897).
The apparent paradox of the actual toxicology of the substance suggests the possibility of serious gaps in the understanding of the action of gold in physiology. Only salts and radioisotopes of gold are of pharmacological value, since elemental (metallic) gold is inert to all chemicals it encounters inside the body (e.g., ingested gold cannot be attacked by stomach acid). Some gold salts do have anti-inflammatory
Anti-inflammatory is the property of a substance or treatment that reduces inflammation or swelling. Anti-inflammatory drugs, also called anti-inflammatories, make up about half of analgesics. These drugs remedy pain by reducing inflammation as o ...
properties and at present two are still used as pharmaceuticals in the treatment of arthritis and other similar conditions in the US (sodium aurothiomalate
Sodium aurothiomalate ( INN, known in the United States as gold sodium thiomalate) is a gold compound that is used for its immunosuppressive anti-rheumatic effects. Along with an orally-administered gold salt, auranofin, it is one of only two ...
and auranofin
Auranofin is a gold salt classified by the World Health Organization as an antirheumatic agent. It has the brand name Ridaura.
Use
Auranofin is used to treat rheumatoid arthritis. It improves arthritis symptoms including painful or tender and ...
). These drugs have been explored as a means to help to reduce the pain and swelling of rheumatoid arthritis
Rheumatoid arthritis (RA) is a long-term autoimmune disorder that primarily affects joints. It typically results in warm, swollen, and painful joints. Pain and stiffness often worsen following rest. Most commonly, the wrist and hands are invol ...
, and also (historically) against tuberculosis
Tuberculosis (TB) is an infectious disease usually caused by '' Mycobacterium tuberculosis'' (MTB) bacteria. Tuberculosis generally affects the lungs, but it can also affect other parts of the body. Most infections show no symptoms, i ...
and some parasites.
Gold alloys are used in restorative dentistry
Restorative dentistry is the study, diagnosis and integrated management of diseases of the teeth and their supporting structures and the rehabilitation of the dentition to functional and aesthetic requirements of the individual. Restorative dentis ...
, especially in tooth restorations, such as crowns and permanent bridges. The gold alloys' slight malleability facilitates the creation of a superior molar mating surface with other teeth and produces results that are generally more satisfactory than those produced by the creation of porcelain crowns. The use of gold crowns in more prominent teeth such as incisors is favored in some cultures and discouraged in others.
Colloidal gold preparations (suspensions of gold nanoparticle
Colloidal gold is a sol or colloidal suspension of nanoparticles of gold in a fluid, usually water. The colloid is usually either wine-red coloured (for spherical particles less than 100 nm) or blue/purple (for larger spherical particles ...
s) in water are intensely red-color
Color (American English) or colour (British English) is the visual perceptual property deriving from the spectrum of light interacting with the photoreceptor cells of the eyes. Color categories and physical specifications of color are assoc ...
ed, and can be made with tightly controlled particle sizes up to a few tens of nanometers across by reduction of gold chloride with citrate
Citric acid is an organic compound with the chemical formula HOC(CO2H)(CH2CO2H)2. It is a colorless weak organic acid. It occurs naturally in citrus fruits. In biochemistry, it is an intermediate in the citric acid cycle, which occurs in the ...
or ascorbate ions. Colloidal gold is used in research applications in medicine, biology and materials science. The technique of immunogold labeling exploits the ability of the gold particles to adsorb protein molecules onto their surfaces. Colloidal gold particles coated with specific antibodies can be used as probes for the presence and position of antigens on the surfaces of cells. In ultrathin sections of tissues viewed by electron microscopy, the immunogold labels appear as extremely dense round spots at the position of the antigen.
Gold, or alloys of gold and palladium, are applied as conductive coating to biological specimens and other non-conducting materials such as plastics and glass to be viewed in a scanning electron microscope. The coating, which is usually applied by sputtering
In physics, sputtering is a phenomenon in which microscopic particles of a solid material are ejected from its surface, after the material is itself bombarded by energetic particles of a plasma or gas. It occurs naturally in outer space, and ca ...
with an argon
Argon is a chemical element with the symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as ...
plasma, has a triple role in this application. Gold's very high electrical conductivity drains electrical charge
Electricity is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as described ...
to earth, and its very high density provides stopping power for electrons in the electron beam, helping to limit the depth to which the electron beam penetrates the specimen. This improves definition of the position and topography of the specimen surface and increases the spatial resolution
In physics and geosciences, the term spatial resolution refers to distance between independent measurements, or the physical dimension that represents a pixel of the image. While in some instruments, like cameras and telescopes, spatial resolut ...
of the image. Gold also produces a high output of secondary electrons when irradiated by an electron beam, and these low-energy electrons are the most commonly used signal source used in the scanning electron microscope.
The isotope gold-198 ( half-life 2.7 days) is used in nuclear medicine
Nuclear medicine or nucleology is a medical specialty involving the application of radioactive substances in the diagnosis and treatment of disease. Nuclear imaging, in a sense, is " radiology done inside out" because it records radiation emi ...
, in some cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
treatments and for treating other diseases.
Cuisine
* Gold can be used in food and has the E number 175. In 2016, theEuropean Food Safety Authority
The European Food Safety Authority (EFSA) is the agency of the European Union (EU) that provides independent scientific advice and communicates on existing and emerging risks associated with the food chain. EFSA was established in February 2002, ...
published an opinion on the re-evaluation of gold as a food additive. Concerns included the possible presence of minute amounts of gold nanoparticles in the food additive, and that gold nanoparticles have been shown to be genotoxic in mammalian cells in vitro
''In vitro'' (meaning in glass, or ''in the glass'') studies are performed with microorganisms, cells, or biological molecules outside their normal biological context. Colloquially called " test-tube experiments", these studies in biology ...
.
* Gold leaf, flake or dust is used on and in some gourmet foods, notably sweets and drinks as decorative ingredient. Gold flake was used by the nobility in medieval Europe as a decoration in food and drinks, in the form of leaf, flakes or dust, either to demonstrate the host's wealth or in the belief that something that valuable and rare must be beneficial for one's health.
* Danziger Goldwasser (German: Gold water of Danzig) or Goldwasser ( en, Goldwater) is a traditional German herbal liqueur produced in what is today Gdańsk, Poland
Poland, officially the Republic of Poland, is a country in Central Europe. It is divided into 16 administrative provinces called voivodeships, covering an area of . Poland has a population of over 38 million and is the fifth-most populou ...
, and Schwabach
Schwabach () is a German city of about 40,000 inhabitants near Nuremberg in the centre of the region of Franconia in the north of Bavaria. The city is an autonomous administrative district (''kreisfreie Stadt''). Schwabach is also the name of th ...
, Germany, and contains flakes of gold leaf. There are also some expensive (c. $1000) cocktails which contain flakes of gold leaf. However, since metallic gold is inert to all body chemistry, it has no taste, it provides no nutrition, and it leaves the body unaltered.
* Vark
Vark (also varak or warq) is a fine filigree foil sheet of pure metal, typically silver but sometimes gold,
is a foil
Foil may refer to:
Materials
* Foil (metal), a quite thin sheet of metal, usually manufactured with a rolling mill machine
* Metal leaf, a very thin sheet of decorative metal
* Aluminium foil, a type of wrapping for food
* Tin foil, metal foil ...
composed of a pure metal that is sometimes gold, and is used for garnishing sweets in South Asian cuisine.
Miscellanea

 * Gold produces a deep, intense red color when used as a coloring agent in
* Gold produces a deep, intense red color when used as a coloring agent in cranberry glass
Cranberry glass or Gold Ruby glass is a red glass made by adding gold salts or colloidal gold to molten glass. Tin, in the form of stannous chloride, is sometimes added in tiny amounts as a reducing agent. The glass is used primarily in exp ...
.
* In photography, gold toners are used to shift the color of silver bromide black-and-white prints towards brown or blue tones, or to increase their stability. Used on sepia-toned prints, gold toners produce red tones. Kodak published formulas for several types of gold toners, which use gold as the chloride.
* Gold is a good reflector of electromagnetic radiation such as infrared and visible light, as well as radio waves
Radio waves are a type of electromagnetic radiation with the longest wavelengths in the electromagnetic spectrum, typically with frequencies of 300 gigahertz ( GHz) and below. At 300 GHz, the corresponding wavelength is 1 mm (s ...
. It is used for the protective coatings on many artificial satellite
A satellite or artificial satellite is an object intentionally placed into orbit in outer space. Except for passive satellites, most satellites have an electricity generation system for equipment on board, such as solar panels or radioi ...
s, in infrared protective faceplates in thermal-protection suits and astronauts' helmets, and in electronic warfare
Electronic warfare (EW) is any action involving the use of the electromagnetic spectrum (EM spectrum) or directed energy to control the spectrum, attack an enemy, or impede enemy assaults. The purpose of electronic warfare is to deny the opponen ...
planes such as the EA-6B Prowler.
* Gold is used as the reflective layer on some high-end CDs.
* Automobiles may use gold for heat shielding. McLaren uses gold foil in the engine compartment of its F1 model.
* Gold can be manufactured so thin that it appears semi-transparent. It is used in some aircraft cockpit windows for de-icing
Deicing is the process of removing snow, ice or frost from a surface. Anti-icing is the application of chemicals that not only deice but also remain on a surface and continue to delay the reformation of ice for a certain period of time, or prev ...
or anti-icing by passing electricity through it. The heat produced by the resistance of the gold is enough to prevent ice from forming.
* Gold is attacked by and dissolves in alkaline solutions of potassium or sodium cyanide, to form the salt gold cyanide—a technique that has been used in extracting metallic gold from ores in the cyanide process
Gold cyanidation (also known as the cyanide process or the MacArthur-Forrest process) is a hydrometallurgical technique for extracting gold from low-grade ore by converting the gold to a water-soluble coordination complex. It is the most commonl ...
. Gold cyanide is the electrolyte used in commercial electroplating of gold onto base metals and electroforming
Electroforming is a metal forming process in which parts are fabricated through electrodeposition on a model, known in the industry as a mandrel. Conductive (metallic) mandrels are treated to create a mechanical parting layer, or are chemically p ...
.
* Gold chloride ( chloroauric acid) solutions are used to make colloidal gold by reduction with citrate
Citric acid is an organic compound with the chemical formula HOC(CO2H)(CH2CO2H)2. It is a colorless weak organic acid. It occurs naturally in citrus fruits. In biochemistry, it is an intermediate in the citric acid cycle, which occurs in the ...
or ascorbate ions
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conven ...
. Gold chloride and gold oxide are used to make cranberry or red-colored glass, which, like colloidal gold suspensions, contains evenly sized spherical gold nanoparticle
Colloidal gold is a sol or colloidal suspension of nanoparticles of gold in a fluid, usually water. The colloid is usually either wine-red coloured (for spherical particles less than 100 nm) or blue/purple (for larger spherical particles ...
s.
* Gold, when dispersed in nanoparticles, can act as a heterogeneous catalyst of chemical reactions.
Toxicity
Pure metallic (elemental) gold is non-toxic and non-irritating when ingested and is sometimes used as a food decoration in the form of gold leaf. Metallic gold is also a component of the alcoholic drinks Goldschläger, Gold Strike, and Goldwasser. Metallic gold is approved as a food additive in the EU ( E175 in the Codex Alimentarius). Although the gold ion is toxic, the acceptance of metallic gold as a food additive is due to its relative chemical inertness, and resistance to being corroded or transformed into soluble salts (gold compounds) by any known chemical process which would be encountered in the human body. Soluble compounds ( gold salts) such asgold chloride Gold chloride can refer to:
* Gold(I) chloride (gold monochloride), AuCl
* Gold(I,III) chloride (gold dichloride, tetragold octachloride), Au4Cl8
* Gold(III) chloride (gold trichloride, digold hexachloride), Au2Cl6
* Chloroauric acid
Chloroauri ...
are toxic to the liver and kidneys. Common cyanide salts of gold such as potassium gold cyanide, used in gold electroplating, are toxic by virtue of both their cyanide and gold content. There are rare cases of lethal gold poisoning from potassium gold cyanide. Gold toxicity can be ameliorated with chelation therapy with an agent such as dimercaprol.
Gold metal was voted Allergen of the Year in 2001 by the American Contact Dermatitis Society; gold contact allergies affect mostly women. Despite this, gold is a relatively non-potent contact allergen, in comparison with metals like nickel
Nickel is a chemical element with symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive but large pieces are slow ...
.
A sample of the fungus ''Aspergillus niger
''Aspergillus niger'' is a mold classified within the ''Nigri'' section of the ''Aspergillus'' genus. The ''Aspergillus'' genus consists of common molds found throughout the environment within soil and water, on vegetation, in fecal matter, on de ...
'' was found growing from gold mining solution; and was found to contain cyano metal complexes, such as gold, silver, copper, iron and zinc. The fungus also plays a role in the solubilization of heavy metal sulfides.
See also
 * Bulk leach extractable gold, for sampling ores
*
* Bulk leach extractable gold, for sampling ores
* Chrysiasis
Chrysiasis is a dermatological condition induced by the parenteral administration of gold salts, usually for the treatment of rheumatoid arthritis.James, William; Berger, Timothy; Elston, Dirk (2005). ''Andrews' Diseases of the Skin: Clinical Derma ...
(dermatological condition)
* Digital gold currency
Digital gold currency (or DGC) is a form of electronic money (or digital currency) based on mass units of gold. It is a kind of representative money, like a US paper gold certificate at the time (from 1873 to 1933) that these were exchangeable ...
, form of electronic currency
* GFMS business consultancy
* Gold fingerprinting, use impurities to identify an alloy
* Gold standard in banking
* List of countries by gold production
* Tumbaga, alloy of gold and copper
* Iron pyrite, fool's gold
* Nordic gold, non-gold copper alloy
References
Further reading
* Bachmann, H. G. ''The lure of gold : an artistic and cultural history'' (2006online
* Bernstein, Peter L. ''The Power of Gold: The History of an Obsession'' (2000
online
* Brands, H.W. ''The Age of Gold: The California Gold Rush and the New American Dream'' (2003
excerpt
* Buranelli, Vincent. ''Gold : an illustrated history'' (1979
online
wide-ranging popular history * Cassel, Gustav. "The restoration of the gold standard." ''Economica'' 9 (1923): 171–185
online
* Eichengreen, Barry. ''Golden Fetters: The Gold Standard and the Great Depression, 1919–1939'' (Oxford UP, 1992). * Ferguson, Niall. ''The Ascent of Money - Financial History of the World'' (2009
online
* Hart, Matthew
Gold: The Race for the World's Most Seductive Metal
''Gold : the race for the world's most seductive metal"], New York: Simon & Schuster, 2013. '' * Johnson, Harry G. "The gold rush of 1968 in retrospect and prospect." ''American Economic Review'' 59.2 (1969): 344–348
online
* Kwarteng, Kwasi. ''War and Gold: A Five-Hundred-Year History of Empires, Adventures, and Debt'' (2014
online
* Vilar, Pierre. '' A History of Gold and Money, 1450 to 1920'' (1960)
online
* Vilches, Elvira. ''New World Gold: Cultural Anxiety and Monetary Disorder in Early Modern Spain'' (2010).
External links
*Chemistry in its element podcast
(MP3) from the
Royal Society of Chemistry
The Royal Society of Chemistry (RSC) is a learned society (professional association) in the United Kingdom with the goal of "advancing the chemical sciences". It was formed in 1980 from the amalgamation of the Chemical Society, the Royal Instit ...
's Chemistry WorldGold
www.rsc.org
at ''
The Periodic Table of Videos
''Periodic Videos'' (also known as ''The Periodic Table of Videos'') is a video project and YouTube channel on chemistry. It consists of a series of videos about chemical elements and the periodic table, with additional videos on other topics i ...
'' (University of Nottingham)
''Getting Gold'' 1898 book
www.lateralscience.co.uk * , www.epa.gov
Gold element information
- rsc.org {{Authority control Chemical elements Transition metals Noble metals Precious metals Cubic minerals Minerals in space group 225 Dental materials Electrical conductors Native element minerals E-number additives Symbols of Alaska Symbols of California Chemical elements with face-centered cubic structure