Elimination reaction on:
[Wikipedia]
[Google]
[Amazon]
An elimination reaction is a type of
 An example of this type of reaction in ''scheme 1'' is the reaction of isobutylbromide with potassium ethoxide in
An example of this type of reaction in ''scheme 1'' is the reaction of isobutylbromide with potassium ethoxide in
 :Only reaction product A results from antiperiplanar elimination. The presence of product B is an indication that an E1 mechanism is occurring.
* It is accompanied by carbocationic
:Only reaction product A results from antiperiplanar elimination. The presence of product B is an indication that an E1 mechanism is occurring.
* It is accompanied by carbocationic 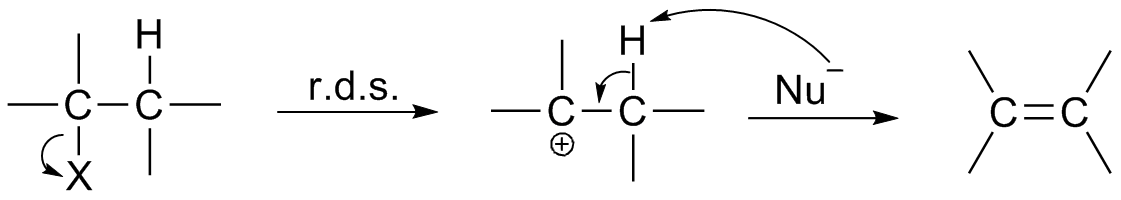 An example in ''scheme 2'' is the reaction of tert-butylbromide with potassium ethoxide in ethanol.
E1 eliminations happen with highly substituted alkyl halides for two main reasons.
* Highly substituted alkyl halides are bulky, limiting the room for the E2 one-step mechanism; therefore, the two-step E1 mechanism is favored.
* Highly substituted carbocations are more stable than methyl or primary substituted cations. Such stability gives time for the two-step E1 mechanism to occur.
* If SN1 and E1 pathways are competing, the E1 pathway can be favored by increasing the heat.
Specific features :
# Rearrangement possible
# Independent of concentration and basicity of base
An example in ''scheme 2'' is the reaction of tert-butylbromide with potassium ethoxide in ethanol.
E1 eliminations happen with highly substituted alkyl halides for two main reasons.
* Highly substituted alkyl halides are bulky, limiting the room for the E2 one-step mechanism; therefore, the two-step E1 mechanism is favored.
* Highly substituted carbocations are more stable than methyl or primary substituted cations. Such stability gives time for the two-step E1 mechanism to occur.
* If SN1 and E1 pathways are competing, the E1 pathway can be favored by increasing the heat.
Specific features :
# Rearrangement possible
# Independent of concentration and basicity of base
 The next most common type of elimination reaction is α-elimination. For a carbon center, the result of α-elimination is the formation of a carbene, which includes "stable carbenes" such as
The next most common type of elimination reaction is α-elimination. For a carbon center, the result of α-elimination is the formation of a carbene, which includes "stable carbenes" such as
organic reaction
Organic reactions are chemical reactions involving organic compounds. The basic organic chemistry reaction types are addition reactions, elimination reactions, substitution reactions, pericyclic reactions, rearrangement reactions, photochemical ...
in which two substituent
A substituent is one or a group of atoms that replaces (one or more) atoms, thereby becoming a moiety in the resultant (new) molecule. (In organic chemistry and biochemistry, the terms ''substituent'' and ''functional group'', as well as '' side ...
s are removed from a molecule in either a one- or two-step mechanism. The one-step mechanism is known as the E2 reaction, and the two-step mechanism is known as the E1 reaction. The numbers refer not to the number of steps in the mechanism, but rather to the kinetics of the reaction: E2 is bimolecular (second-order) while E1 is unimolecular (first-order). In cases where the molecule is able to stabilize an anion but possesses a poor leaving group, a third type of reaction, E1CB, exists. Finally, the pyrolysis of xanthate and acetate esters proceed through an "internal" elimination mechanism, the Ei mechanism.
E2 mechanism
The E2 mechanism, where E2 stands for bimolecular elimination, involves a one-step mechanism in which ''carbon-hydrogen'' and ''carbon-halogen'' bonds break to form a double bond (''C=C Pi bond''). The specifics of the reaction are as follows: * E2 is a single step elimination, with a singletransition state
In chemistry, the transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest potential energy along this reaction coordinate. It is often marked ...
.
* It is typically undergone by primary substituted alkyl halides, but is possible with some secondary alkyl halides and other compounds.
* The reaction rate
The reaction rate or rate of reaction is the speed at which a chemical reaction takes place, defined as proportional to the increase in the concentration of a product per unit time and to the decrease in the concentration of a reactant per uni ...
is second order, because it's influenced by both the alkyl halide and the base (bimolecular).
* Because the E2 mechanism results in the formation of a pi bond, the two leaving groups (often a hydrogen and a halogen
The halogens () are a group in the periodic table consisting of five or six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). In the modern IUPAC nomenclature, this grou ...
) need to be antiperiplanar. An antiperiplanar transition state
In chemistry, the transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest potential energy along this reaction coordinate. It is often marked ...
has staggered conformation with lower energy than a synperiplanar transition state which is in eclipsed conformation with higher energy. The reaction mechanism involving staggered conformation is more favorable for E2 reactions (unlike E1 reactions).
* E2 typically uses a strong base. It must be strong enough to remove a weakly acidic hydrogen.
* In order for the pi bond to be created, the hybridization
Hybridization (or hybridisation) may refer to:
*Hybridization (biology), the process of combining different varieties of organisms to create a hybrid
*Orbital hybridization, in chemistry, the mixing of atomic orbitals into new hybrid orbitals
*Nu ...
of carbons needs to be lowered from ''sp3'' to ''sp2''.
* The C-H bond is weakened in the rate determining step and therefore a primary deuterium isotope effect
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for ...
much larger than 1 (commonly 2-6) is observed.
* E2 competes with the SN2 reaction mechanism if the base can also act as a nucleophile (true for many common bases).
ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a ...
. The reaction products are isobutene
Isobutylene (or 2-methylpropene) is a hydrocarbon with the chemical formula . It is a four-carbon branched alkene (olefin), one of the four isomers of butylene. It is a colorless flammable gas, and is of considerable industrial value.
Produc ...
, ethanol and potassium bromide
Potassium bromide ( K Br) is a salt, widely used as an anticonvulsant and a sedative in the late 19th and early 20th centuries, with over-the-counter use extending to 1975 in the US. Its action is due to the bromide ion ( sodium bromide is equa ...
.
E1 mechanism
E1 is a model to explain a particular type of chemical elimination reaction. E1 stands for unimolecular elimination and has the following specifications * It is a two-step process of elimination: ''ionization and deprotonation''. **Ionization
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecul ...
: the carbon-halogen bond breaks to give a carbocation
A carbocation is an ion with a positively charged carbon atom. Among the simplest examples are the methenium , methanium and vinyl cations. Occasionally, carbocations that bear more than one positively charged carbon atom are also encount ...
intermediate.
** deprotonation
Deprotonation (or dehydronation) is the removal (transfer) of a proton (or hydron, or hydrogen cation), (H+) from a Brønsted–Lowry acid in an acid–base reaction.Henry Jakubowski, Biochemistry Online Chapter 2A3, https://employees.csbsju. ...
of the carbocation.
* E1 typically takes place with tertiary
Tertiary ( ) is a widely used but obsolete term for the geologic period from 66 million to 2.6 million years ago.
The period began with the demise of the non-avian dinosaurs in the Cretaceous–Paleogene extinction event, at the start ...
alkyl halides, but is possible with some secondary alkyl halides.
* The reaction rate
The reaction rate or rate of reaction is the speed at which a chemical reaction takes place, defined as proportional to the increase in the concentration of a product per unit time and to the decrease in the concentration of a reactant per uni ...
is influenced only by the concentration of the alkyl halide because carbocation formation is the slowest step, as known as the rate-determining step. Therefore, first-order kinetics apply (unimolecular).
* The reaction usually occurs in the complete absence of a base or the presence of only a weak base (acidic conditions and high temperature).
* E1 reactions are in competition with SN1 reactions because they share a common carbocationic intermediate.
* A secondary deuterium isotope effect
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for ...
of slightly larger than 1 (commonly 1 - 1.5) is observed.
* There is no antiperiplanar requirement. An example is the pyrolysis
The pyrolysis (or devolatilization) process is the thermal decomposition of materials at elevated temperatures, often in an inert atmosphere. It involves a change of chemical composition. The word is coined from the Greek-derived elements '' ...
of a certain sulfonate ester
In organosulfur chemistry, a sulfonate is a salt or ester of a sulfonic acid. It contains the functional group , where R is an organic group. Sulfonates are the conjugate bases of sulfonic acids. Sulfonates are generally stable in water, non-ox ...
of menthol
Menthol is an organic compound, more specifically a monoterpenoid, made synthetically or obtained from the oils of corn mint, peppermint, or other mints. It is a waxy, clear or white crystalline substance, which is solid at room temperature ...
:
rearrangement reaction
In organic chemistry, a rearrangement reaction is a broad class of organic reactions where the carbon skeleton of a molecule is rearranged to give a structural isomer of the original molecule. Often a substituent moves from one atom to anothe ...
s
 An example in ''scheme 2'' is the reaction of tert-butylbromide with potassium ethoxide in ethanol.
E1 eliminations happen with highly substituted alkyl halides for two main reasons.
* Highly substituted alkyl halides are bulky, limiting the room for the E2 one-step mechanism; therefore, the two-step E1 mechanism is favored.
* Highly substituted carbocations are more stable than methyl or primary substituted cations. Such stability gives time for the two-step E1 mechanism to occur.
* If SN1 and E1 pathways are competing, the E1 pathway can be favored by increasing the heat.
Specific features :
# Rearrangement possible
# Independent of concentration and basicity of base
An example in ''scheme 2'' is the reaction of tert-butylbromide with potassium ethoxide in ethanol.
E1 eliminations happen with highly substituted alkyl halides for two main reasons.
* Highly substituted alkyl halides are bulky, limiting the room for the E2 one-step mechanism; therefore, the two-step E1 mechanism is favored.
* Highly substituted carbocations are more stable than methyl or primary substituted cations. Such stability gives time for the two-step E1 mechanism to occur.
* If SN1 and E1 pathways are competing, the E1 pathway can be favored by increasing the heat.
Specific features :
# Rearrangement possible
# Independent of concentration and basicity of base
Competition among mechanisms
Thereaction rate
The reaction rate or rate of reaction is the speed at which a chemical reaction takes place, defined as proportional to the increase in the concentration of a product per unit time and to the decrease in the concentration of a reactant per uni ...
is influenced by the reactivity of halogen
The halogens () are a group in the periodic table consisting of five or six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). In the modern IUPAC nomenclature, this grou ...
s, iodide and bromide being favored. Fluoride is not a good leaving group, so eliminations with fluoride as the leaving group have slower rates than other halogens .
There is a certain level of competition between the elimination reaction and nucleophilic substitution. More precisely, there are competitions between E2 and SN2 and also between E1 and SN1. Substitution generally predominates and elimination occurs only during precise circumstances. Generally, elimination is favored over substitution when
* steric hindrance
Steric effects arise from the spatial arrangement of atoms. When atoms come close together there is a rise in the energy of the molecule. Steric effects are nonbonding interactions that influence the shape ( conformation) and reactivity of ions ...
around the α-carbon increases.
* a stronger base is used.
* temperature
Temperature is a physical quantity that expresses quantitatively the perceptions of hotness and coldness. Temperature is measured with a thermometer.
Thermometers are calibrated in various temperature scales that historically have relied o ...
increases (increase entropy
Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. The term and the concept are used in diverse fields, from classical thermodyna ...
)
* the base is a poor nucleophile
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they ar ...
. Bases with steric bulk, (such as in Potassium tert-butoxide
Potassium ''tert''-butoxide is the chemical compound with the formula K+(CH3)3CO−. This colourless solid is a strong base (pKa of conjugate acid around 17), which is useful in organic synthesis. It exists as a tetrameric cubane-type cluster. ...
), are often poor nucleophiles.
An example of Elimination predominating substitution is visible from a special case of Williamson synthesis. When 3° haloalkane reacts with an alkoxide, due to strong basic character of the alkoxide and less reactivity of 3° group towards SN2, an alkene will be formed instead of the expected ether with a 3° group.
In one study the kinetic isotope effect
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for ...
(KIE) was determined for the gas phase reaction of several alkyl halides with the chlorate
The chlorate anion has the formula ClO3-. In this case, the chlorine atom is in the +5 oxidation state. "Chlorate" can also refer to chemical compounds containing this anion; chlorates are the salts of chloric acid. "Chlorate", when followed by ...
ion. In accordance with an E2 elimination the reaction with t-butyl chloride results in a KIE of 2.3. The methyl chloride
Chloromethane, also called methyl chloride, Refrigerant-40, R-40 or HCC 40, is an organic compound with the chemical formula . One of the haloalkanes, it is a colorless, odorless, flammable gas. Methyl chloride is a crucial reagent in industria ...
reaction (only SN2 possible) on the other hand has a KIE of 0.85 consistent with a SN2 reaction because in this reaction type the C-H bonds tighten in the transition state. The KIE's for the ethyl (0.99) and isopropyl (1.72) analogues suggest competition between the two reaction modes.
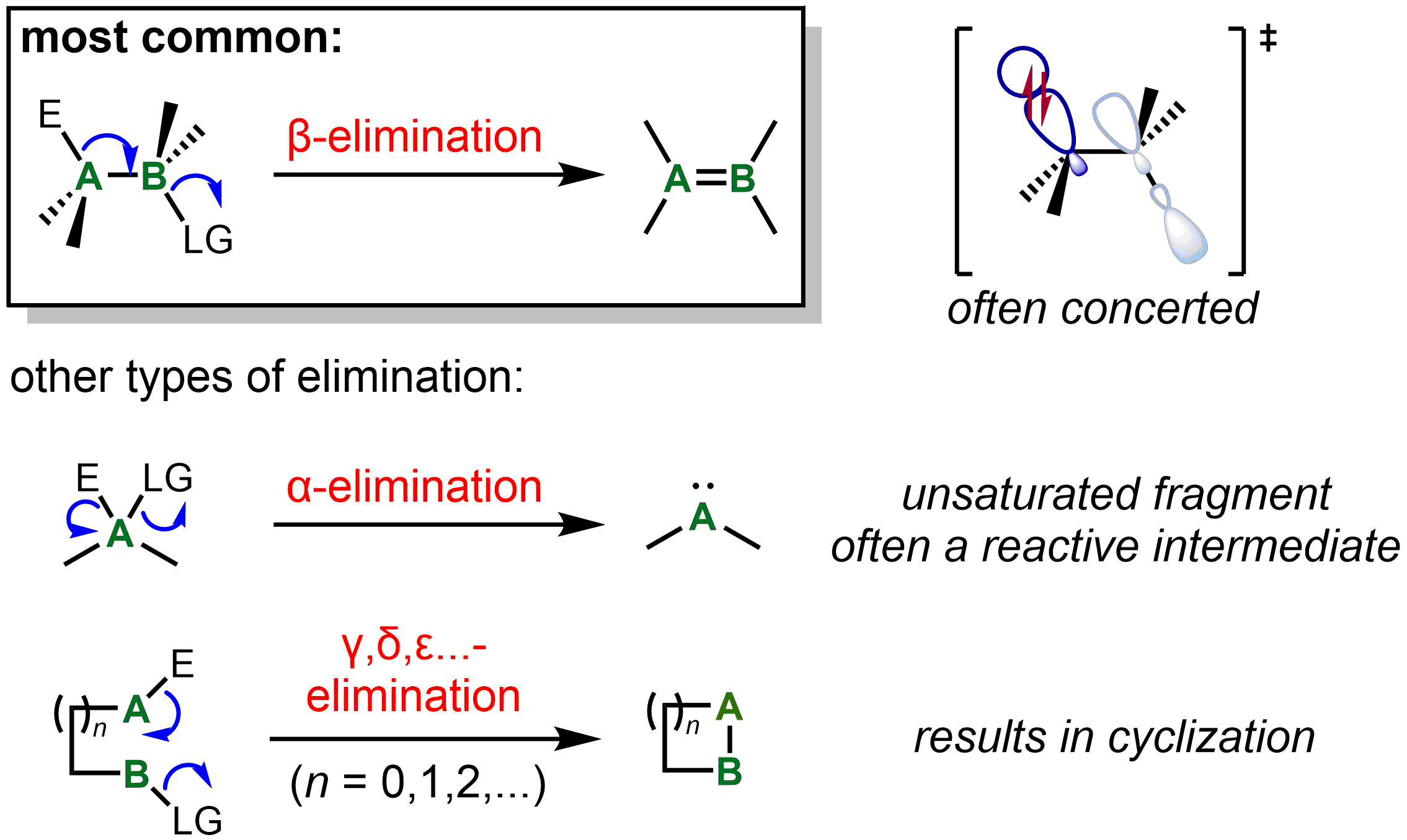
Elimination reactions other than β-elimination
β-Elimination, with loss of electrofuge and nucleofuge on vicinal carbons, is by far the most common type of elimination. The ability to form a stable product containing a C=C or C=X bond, as well as orbital alignment considerations, strongly favors β-elimination over other elimination processes. However, other types are known, generally for systems where β-elimination cannot occur. The next most common type of elimination reaction is α-elimination. For a carbon center, the result of α-elimination is the formation of a carbene, which includes "stable carbenes" such as
The next most common type of elimination reaction is α-elimination. For a carbon center, the result of α-elimination is the formation of a carbene, which includes "stable carbenes" such as carbon monoxide
Carbon monoxide ( chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simpl ...
or isocyanides. For instance, α-elimination the elements of HCl from chloroform (CHCl3) in the presence of strong base is a classic approach for the generation of dichlorocarbene, :CCl2, as a reactive intermediate. On the other hand, formic acid undergoes α-elimination to afford the stable products water and carbon monoxide under acidic conditions. α-Elimination may also occur on a metal center, one particularly common result of which is lowering of both the metal oxidation state and coordination number by 2 units in a process known as reductive elimination. (Confusingly, in organometallic terminology, the terms ''α-elimination'' and ''α-abstraction'' refer to processes that result in formation of a metal-carbene complex. In these reactions, it is the carbon adjacent to the metal that undergoes α-elimination.)
In certain special cases, γ- and higher eliminations to form three-membered or larger rings is also possible in both organic and organometallic processes. For instance, certain Pt(II) complexes undergo γ- and δ-elimination to give metallocycles. More recently, γ-silyl elimination of a silylcyclobutyl tosylate has been used to prepare strained bicyclic systems.
History
Many of the concepts and terminology related to elimination reactions were proposed by Christopher Kelk Ingold in the 1920s.See also
* E1cB-elimination reactionReferences
External links
* * {{DEFAULTSORT:Elimination Reaction Olefination reactions Reaction mechanisms