Electromagnetic separation on:
[Wikipedia]
[Google]
[Amazon]
Isotope separation is the process of concentrating specific
 Often done with gases, but also with liquids, the
Often done with gases, but also with liquids, the

 Electromagnetic separation is
Electromagnetic separation is
Utilization of kinetic isotope effects for the concentration of tritium
GM Brown, TJ Meyer et al., 2001.
from the
Annotated bibliography on electromagnetic separation of uranium isotopes form the Alsos Digital Library
German inventions
isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers ( mass num ...
s of a chemical element
A chemical element is a species of atoms that have a given number of protons in their atomic nucleus, nuclei, including the pure Chemical substance, substance consisting only of that species. Unlike chemical compounds, chemical elements canno ...
by removing other isotopes. The use of the nuclides
A nuclide (or nucleide, from nucleus, also known as nuclear species) is a class of atoms characterized by their number of protons, ''Z'', their number of neutrons, ''N'', and their nuclear energy state.
The word ''nuclide'' was coined by Truman ...
produced is varied. The largest variety is used in research (e.g. in chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a natural science that covers the elements that make up matter to the compounds made of atoms, molecules and ions: their composition, structure, proper ...
where atoms of "marker" nuclide are used to figure out reaction mechanisms). By tonnage, separating natural uranium
Natural uranium (NU or Unat) refers to uranium with the same isotopic ratio as found in nature. It contains 0.711% uranium-235, 99.284% uranium-238, and a trace of uranium-234 by weight (0.0055%). Approximately 2.2% of its radioactivity comes ...
into enriched uranium
Enriched uranium is a type of uranium in which the percent composition of uranium-235 (written 235U) has been increased through the process of isotope separation. Naturally occurring uranium is composed of three major isotopes: uranium-238 (238U ...
and depleted uranium
Depleted uranium (DU; also referred to in the past as Q-metal, depletalloy or D-38) is uranium with a lower content of the fissile isotope than natural uranium.: "Depleted uranium possesses only 60% of the radioactivity of natural uranium, hav ...
is the largest application. In the following text, mainly the uranium enrichment is considered. This process is crucial in the manufacture of uranium fuel for nuclear power plant
A nuclear power plant (NPP) is a thermal power station in which the heat source is a nuclear reactor. As is typical of thermal power stations, heat is used to generate steam that drives a steam turbine connected to a generator that produces ...
s, and is also required for the creation of uranium-based nuclear weapon
A nuclear weapon is an explosive device that derives its destructive force from nuclear reactions, either fission (fission bomb) or a combination of fission and fusion reactions ( thermonuclear bomb), producing a nuclear explosion. Both bomb ...
s. Plutonium-based weapons use plutonium
Plutonium is a radioactive chemical element with the symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, and forms a dull coating when oxidized. The element normally exh ...
produced in a nuclear reactor, which must be operated in such a way as to produce plutonium already of suitable isotopic mix or ''grade''.
While different chemical elements can be purified through chemical processes
A chemical substance is a form of matter having constant chemical composition and characteristic properties. Some references add that chemical substance cannot be separated into its constituent elements by physical separation methods, i.e., wit ...
, isotopes of the same element have nearly identical chemical properties, which makes this type of separation impractical, except for separation of deuterium
Deuterium (or hydrogen-2, symbol or deuterium, also known as heavy hydrogen) is one of two stable isotopes of hydrogen (the other being protium, or hydrogen-1). The nucleus of a deuterium atom, called a deuteron, contains one proton and one ...
.
Separation techniques
There are three types of isotope separation techniques: * Those based directly on theatomic weight
Relative atomic mass (symbol: ''A''; sometimes abbreviated RAM or r.a.m.), also known by the deprecated synonym atomic weight, is a dimensionless physical quantity defined as the ratio of the average mass of atoms of a chemical element in a giv ...
of the isotope.
* Those based on the small differences in chemical reaction rates produced by different atomic weights.
* Those based on properties not directly connected to atomic weight, such as nuclear resonances.
The third type of separation is still experimental; practical separation techniques all depend in some way on the atomic mass. It is therefore generally easier to separate isotopes with a larger relative mass difference. For example, deuterium
Deuterium (or hydrogen-2, symbol or deuterium, also known as heavy hydrogen) is one of two stable isotopes of hydrogen (the other being protium, or hydrogen-1). The nucleus of a deuterium atom, called a deuteron, contains one proton and one ...
has twice the mass of ordinary (light) hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-to ...
and it is generally easier to purify it than to separate uranium-235
Uranium-235 (235U or U-235) is an isotope of uranium making up about 0.72% of natural uranium. Unlike the predominant isotope uranium-238, it is fissile, i.e., it can sustain a nuclear chain reaction. It is the only fissile isotope that exi ...
from the more common uranium-238
Uranium-238 (238U or U-238) is the most common isotope of uranium found in nature, with a relative abundance of 99%. Unlike uranium-235, it is non-fissile, which means it cannot sustain a chain reaction in a thermal-neutron reactor. However ...
. On the other extreme, separation of fissile plutonium-239
Plutonium-239 (239Pu or Pu-239) is an isotope of plutonium. Plutonium-239 is the primary fissile isotope used for the production of nuclear weapons, although uranium-235 is also used for that purpose. Plutonium-239 is also one of the three mai ...
from the common impurity plutonium-240
Plutonium-240 ( or Pu-240) is an isotope of plutonium formed when plutonium-239 captures a neutron. The detection of its spontaneous fission led to its discovery in 1944 at Los Alamos and had important consequences for the Manhattan Project.
240 ...
, while desirable in that it would allow the creation of gun-type fission weapon
Gun-type fission weapons are fission-based nuclear weapons whose design assembles their fissile material into a supercritical mass by the use of the "gun" method: shooting one piece of sub-critical material into another. Although this is someti ...
s from plutonium, is generally agreed to be impractical.
Enrichment cascades
All large-scale isotope separation schemes employ a number of similar stages which produce successively higher concentrations of the desired isotope. Each stage enriches the product of the previous step further before being sent to the next stage. Similarly, the tailings from each stage are returned to the previous stage for further processing. This creates a sequential enriching system called acascade
Cascade, Cascades or Cascading may refer to:
Science and technology Science
* Cascade waterfalls, or series of waterfalls
* Cascade, the CRISPR-associated complex for antiviral defense (a protein complex)
* Cascade (grape), a type of fruit
* Bioc ...
.
There are two important factors that affect the performance of a cascade. The first is the separation factor
Separation may refer to:
Films
* ''Separation'' (1967 film), a British feature film written by and starring Jane Arden and directed by Jack Bond
* ''La Séparation'', 1994 French film
* '' A Separation'', 2011 Iranian film
* ''Separation'' (2 ...
, which is a number greater than 1. The second is the number of required stages to get the desired purity.
Commercial materials
To date, large-scale commercial isotope separation of only three elements has occurred. In each case, the rarer of the two most common isotopes of an element has been concentrated for use in nuclear technology: *Uranium
Uranium is a chemical element with the symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium is weak ...
isotopes have been separated to prepare enriched uranium
Enriched uranium is a type of uranium in which the percent composition of uranium-235 (written 235U) has been increased through the process of isotope separation. Naturally occurring uranium is composed of three major isotopes: uranium-238 (238U ...
for use as nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
fuel and in nuclear weapons
A nuclear weapon is an explosive device that derives its destructive force from nuclear reactions, either fission (fission bomb) or a combination of fission and fusion reactions ( thermonuclear bomb), producing a nuclear explosion. Both bom ...
.
* Hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-to ...
isotopes have been separated to prepare heavy water for use as a moderator in nuclear reactors.
** Tritium
Tritium ( or , ) or hydrogen-3 (symbol T or H) is a rare and radioactive isotope of hydrogen with half-life about 12 years. The nucleus of tritium (t, sometimes called a ''triton'') contains one proton and two neutrons, whereas the nucleus of ...
is both a nuisance in the coolant / moderator of water moderated reactors and a valuable product, it is thus sometimes separated from the coolant
* Lithium
Lithium (from el, λίθος, lithos, lit=stone) is a chemical element with the symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard conditions, it is the least dense metal and the least dense soli ...
-6 has been concentrated for use in thermonuclear weapons. Tritium is commonly produced from Lithium-6 which is often enriched for this purpose, too.
Some isotopically purified elements are used in smaller quantities for specialist applications, especially in the semiconductor industry, where purified silicon
Silicon is a chemical element with the symbol Si and atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic luster, and is a tetravalent metalloid and semiconductor. It is a member of group 14 in the periodic ...
is used to improve crystal structure and thermal conductivity
The thermal conductivity of a material is a measure of its ability to conduct heat. It is commonly denoted by k, \lambda, or \kappa.
Heat transfer occurs at a lower rate in materials of low thermal conductivity than in materials of high thermal ...
, and carbon with greater isotopic purity to make diamonds with greater thermal conductivity.
Isotope separation is an important process for both peaceful and military nuclear technology, and therefore the capability that a nation has for isotope separation is of extreme interest to the intelligence community.
Alternatives
The only alternative to isotope separation is to manufacture the required isotope in its pure form. This may be done by irradiation of a suitable target, but care is needed in target selection and other factors to ensure that only the required isotope of the element of interest is produced. Isotopes of other elements are not so great a problem as they can be removed by chemical means. This is particularly relevant in the preparation of high-gradeplutonium-239
Plutonium-239 (239Pu or Pu-239) is an isotope of plutonium. Plutonium-239 is the primary fissile isotope used for the production of nuclear weapons, although uranium-235 is also used for that purpose. Plutonium-239 is also one of the three mai ...
for use in weapons. It is not practical to separate Pu-239 from Pu-240 or Pu-241. Fissile
In nuclear engineering, fissile material is material capable of sustaining a nuclear fission chain reaction. By definition, fissile material can sustain a chain reaction with neutrons of thermal energy. The predominant neutron energy may be t ...
Pu-239 is produced following neutron capture by uranium-238, but further neutron capture will produce Pu-240
Plutonium-240 ( or Pu-240) is an isotope of plutonium formed when plutonium-239 captures a neutron. The detection of its spontaneous fission led to its discovery in 1944 at Los Alamos and had important consequences for the Manhattan Project.
240 ...
which is less fissile and worse, is a fairly strong neutron emitter, and Pu-241 which decays to Am-241
Americium-241 (, Am-241) is an isotope of americium. Like all isotopes of americium, it is radioactive, with a half-life of . is the most common isotope of americium as well as the most prevalent isotope of americium in nuclear waste. It is com ...
, a strong alpha emitter that poses self-heating and radiotoxicity problems. Therefore, the uranium targets used to produce military plutonium must be irradiated for only a short time, to minimise the production of these unwanted isotopes. Conversely, blending plutonium with Pu-240 renders it less suitable for nuclear weapons.
If the desired goal is not an atom bomb but running a nuclear power plant, the alternative to enrichment of uranium for use in a Light Water Reactor
The light-water reactor (LWR) is a type of thermal-neutron reactor that uses normal water, as opposed to heavy water, as both its coolant and neutron moderator; furthermore a solid form of fissile elements is used as fuel. Thermal-neutron react ...
is the use of a neutron moderator
In nuclear engineering, a neutron moderator is a medium that reduces the speed of fast neutrons, ideally without capturing any, leaving them as thermal neutrons with only minimal (thermal) kinetic energy. These thermal neutrons are immensely m ...
with a lower neutron absorption cross section than protium. Options include heavy water as used in CANDU
The CANDU (Canada Deuterium Uranium) is a Canadian pressurized heavy-water reactor design used to generate electric power. The acronym refers to its deuterium oxide ( heavy water) moderator and its use of (originally, natural) uranium fuel. C ...
type reactors or graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on la ...
as used in Magnox
Magnox is a type of nuclear power/production reactor that was designed to run on natural uranium with graphite as the moderator and carbon dioxide gas as the heat exchange coolant. It belongs to the wider class of gas-cooled reactors. The n ...
or RBMK
The RBMK (russian: реактор большой мощности канальный, РБМК; ''reaktor bolshoy moshchnosti kanalnyy'', "high-power channel-type reactor") is a class of graphite-moderated nuclear power reactor designed and buil ...
reactors. Obtaining heavy water however also requires isotope separation, in this case of Hydrogen isotopes, which is easier due to the bigger variation in atomic weight. Both Magnox and RBMK reactors had undesirable properties when run with natural uranium
Natural uranium (NU or Unat) refers to uranium with the same isotopic ratio as found in nature. It contains 0.711% uranium-235, 99.284% uranium-238, and a trace of uranium-234 by weight (0.0055%). Approximately 2.2% of its radioactivity comes ...
, which ultimately led to the replacement of this fuel with low enriched uranium, negating the advantage of foregoing enrichment. Pressurized Heavy Water Reactor
A pressurized heavy-water reactor (PHWR) is a nuclear reactor that uses heavy water ( deuterium oxide D2O) as its coolant and neutron moderator. PHWRs frequently use natural uranium as fuel, but sometimes also use very low enriched uranium. The ...
s such as the CANDU are still in active use and India
India, officially the Republic of India (Hindi: ), is a country in South Asia. It is the List of countries and dependencies by area, seventh-largest country by area, the List of countries and dependencies by population, second-most populous ...
which has limited domestic uranium resources and been under a partial nuclear embargo ever since it became an atom bomb state in particular relies on heavy water moderated reactors for its nuclear power. A big downside of heavy water reactors is the enormous upfront cost of the heavy water.
Practical methods of separation
Diffusion
 Often done with gases, but also with liquids, the
Often done with gases, but also with liquids, the diffusion
Diffusion is the net movement of anything (for example, atoms, ions, molecules, energy) generally from a region of higher concentration to a region of lower concentration. Diffusion is driven by a gradient in Gibbs free energy or chemical ...
method relies on the fact that in thermal equilibrium, two isotopes with the same energy will have different average velocities. The lighter atoms (or the molecules containing them) will travel more quickly and be more likely to diffuse through a membrane. The difference in speeds is proportional to the square root of the mass ratio, so the amount of separation is small and many cascaded stages are needed to obtain high purity. This method is expensive due to the work needed to push gas through a membrane and the many stages necessary.
The first large-scale separation of uranium isotopes was achieved by the United States
The United States of America (U.S.A. or USA), commonly known as the United States (U.S. or US) or America, is a country Continental United States, primarily located in North America. It consists of 50 U.S. state, states, a Washington, D.C., ...
in large gaseous diffusion separation plants at Oak Ridge Laboratories, which were established as part of the Manhattan Project
The Manhattan Project was a research and development undertaking during World War II that produced the first nuclear weapons. It was led by the United States with the support of the United Kingdom and Canada. From 1942 to 1946, the project w ...
. These used uranium hexafluoride gas as the process fluid. Nickel powder and electro-deposited nickel mesh diffusion barriers were pioneered by Edward Adler and Edward Norris. See gaseous diffusion.
Centrifugal

Centrifugal
Centrifugal (a key concept in rotating systems) may refer to:
*Centrifugal casting (industrial), Centrifugal casting (silversmithing), and Spin casting (centrifugal rubber mold casting), forms of centrifigual casting
*Centrifugal clutch
*Centrifug ...
schemes rapidly rotate the material allowing the heavier isotopes to go closer to an outer radial wall. This too is often done in gaseous form using a Zippe-type centrifuge
The Zippe-type centrifuge is a gas centrifuge designed to enrich the rare fissile isotope uranium-235 (235U) from the mixture of isotopes found in naturally occurring uranium compounds. The isotopic separation is based on the slight difference in ...
.
Centrifuging plasma can separate isotopes as well as separating ranges of elements for radioactive waste reduction, nuclear reprocessing, and other purposes. The process is called "plasma mass separation"; the devices are called "plasma mass filter" or "plasma centrifuge" (not to be confused with medical).
The centrifugal separation of isotopes was first suggested by Aston and Lindemann in 1919 and the first successful experiments were reported by Beams and Haynes on isotopes of chlorine in 1936. However attempts to use the technology during the Manhattan project
The Manhattan Project was a research and development undertaking during World War II that produced the first nuclear weapons. It was led by the United States with the support of the United Kingdom and Canada. From 1942 to 1946, the project w ...
were unproductive. In modern times it is the main method used throughout the world to enrich uranium and as a result remains a fairly secretive process, hindering a more widespread uptake of the technology. In general a feed of UF6 gas is connected to a cylinder that is rotated at high speed. Near the outer edge of the cylinder heavier gas molecules containing U-238 collect, while molecules containing U-235 concentrate at the center and are then fed to another cascade stage. Use of gaseous centrifugal technology to enrich isotopes is desirable as power consumption is greatly reduced when compared to more conventional techniques such as diffusion plants since fewer cascade steps are required to reach similar degrees of separation. In fact, gas centrifuge
A gas centrifuge is a device that performs isotope separation of gases. A centrifuge relies on the principles of centrifugal force accelerating molecules so that particles of different masses are physically separated in a gradient along the radiu ...
s using uranium hexafluoride have largely replaced gaseous diffusion technology for uranium enrichment. As well as requiring less energy to achieve the same separation, far smaller scale plants are possible, making them an economic possibility for a small nation attempting to produce a nuclear weapon
A nuclear weapon is an explosive device that derives its destructive force from nuclear reactions, either fission (fission bomb) or a combination of fission and fusion reactions ( thermonuclear bomb), producing a nuclear explosion. Both bomb ...
. Pakistan is believed to have used this method in developing its nuclear weapons.
Vortex tube
The vortex tube, also known as the Ranque-Hilsch vortex tube, is a mechanical device that separates a compressed gas into hot and cold streams. The gas emerging from the hot end can reach temperatures of , and the gas emerging from the cold end ...
s were used by South Africa
South Africa, officially the Republic of South Africa (RSA), is the southernmost country in Africa. It is bounded to the south by of coastline that stretch along the South Atlantic and Indian Oceans; to the north by the neighbouring coun ...
in their Helikon vortex separation process
The Helikon vortex separation process is an aerodynamic uranium enrichment process designed around a device called a vortex tube. Paul Dirac thought of the idea for isotope separation and tried creating such a device in 1934 in the lab of Peter K ...
. The gas is injected tangentially into a chamber with special geometry that further increases its rotation to a very high rate, causing the isotopes to separate. The method is simple because vortex tubes have no moving parts, but energy intensive, about 50 times greater than gas centrifuges. A similar process, known as ''jet nozzle'', was created in Germany, with a demonstration plant built in Brazil, and they went as far as developing a site to fuel the country's nuclear plants.
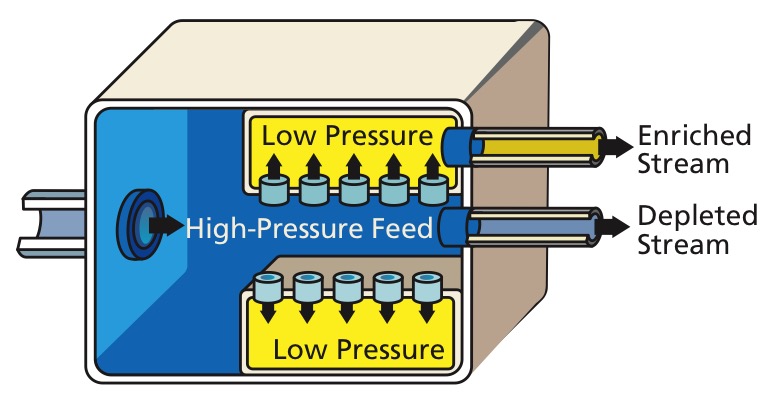
Electromagnetic
 Electromagnetic separation is
Electromagnetic separation is mass spectrometry
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a '' mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is u ...
on a large scale, so it is sometimes referred to as mass spectrometry. It uses the fact that charged particles are deflected in a magnetic field
A magnetic field is a vector field that describes the magnetic influence on moving electric charges, electric currents, and magnetic materials. A moving charge in a magnetic field experiences a force perpendicular to its own velocity and to ...
and the amount of deflection depends upon the particle's mass. It is very expensive for the quantity produced, as it has an extremely low throughput, but it can allow very high purities to be achieved. This method is often used for processing small amounts of pure isotopes for research or specific use (such as isotopic tracer
Isotopic labeling (or isotopic labelling) is a technique used to track the passage of an isotope (an atom with a detectable variation in neutron count) through a reaction, metabolic pathway, or cell. The reactant is 'labeled' by replacing specific ...
s), but is impractical for industrial use.
At Oak Ridge and at the University of California, Berkeley
The University of California, Berkeley (UC Berkeley, Berkeley, Cal, or California) is a public land-grant research university in Berkeley, California. Established in 1868 as the University of California, it is the state's first land-grant un ...
, Ernest O. Lawrence
Ernest Orlando Lawrence (August 8, 1901 – August 27, 1958) was an American nuclear physicist and winner of the Nobel Prize in Physics in 1939 for his invention of the cyclotron. He is known for his work on uranium-isotope separation fo ...
developed electromagnetic separation for much of the uranium used in the first United States atomic bomb (see Manhattan Project
The Manhattan Project was a research and development undertaking during World War II that produced the first nuclear weapons. It was led by the United States with the support of the United Kingdom and Canada. From 1942 to 1946, the project w ...
). Devices using his principle are named calutron
A calutron is a mass spectrometer originally designed and used for separating the isotopes of uranium. It was developed by Ernest Lawrence during the Manhattan Project and was based on his earlier invention, the cyclotron. Its name was deri ...
s. After the war the method was largely abandoned as impractical. It had only been undertaken (along with diffusion and other technologies) to guarantee there would be enough material for use, whatever the cost. Its main eventual contribution to the war effort was to further concentrate material from the gaseous diffusion plants to higher levels of purity.
Laser
In this method alaser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The ...
is tuned to a wavelength which excites only one isotope of the material and ionizes those atoms preferentially. The resonant absorption of light for an isotope is dependent upon its mass and certain hyperfine
In atomic physics, hyperfine structure is defined by small shifts in otherwise degenerate energy levels and the resulting splittings in those energy levels of atoms, molecules, and ions, due to electromagnetic multipole interaction between the ...
interactions between electrons and the nucleus, allowing finely tuned lasers to interact with only one isotope. After the atom is ionized it can be removed from the sample by applying an electric field
An electric field (sometimes E-field) is the physical field that surrounds electrically charged particles and exerts force on all other charged particles in the field, either attracting or repelling them. It also refers to the physical field ...
. This method is often abbreviated as AVLIS ( atomic vapor laser isotope separation). This method has only recently been developed as laser technology has improved, and is currently not used extensively. However, it is a major concern to those in the field of nuclear proliferation
Nuclear proliferation is the spread of nuclear weapons, fissionable material, and weapons-applicable nuclear technology and information to nations not recognized as " Nuclear Weapon States" by the Treaty on the Non-Proliferation of Nuclear Wea ...
because it may be cheaper and more easily hidden than other methods of isotope separation. Tunable laser
A tunable laser is a laser whose wavelength of operation can be altered in a controlled manner. While all laser gain media allow small shifts in output wavelength, only a few types of lasers allow continuous tuning over a significant wavelength ran ...
s used in AVLIS include the dye laser
A dye laser is a laser that uses an organic dye as the lasing medium, usually as a liquid solution. Compared to gases and most solid state lasing media, a dye can usually be used for a much wider range of wavelengths, often spanning 50 to 100 ...
and more recently diode laser
The laser diode chip removed and placed on the eye of a needle for scale
A laser diode (LD, also injection laser diode or ILD, or diode laser) is a semiconductor device similar to a light-emitting diode in which a diode pumped directly with e ...
s.
A second method of laser separation is known as molecular laser isotope separation (MLIS). In this method, an infrared laser is directed at uranium hexafluoride gas, exciting molecules that contain a U-235
Uranium-235 (235U or U-235) is an isotope of uranium making up about 0.72% of natural uranium. Unlike the predominant isotope uranium-238, it is fissile, i.e., it can sustain a nuclear chain reaction. It is the only fissile isotope that exist ...
atom. A second laser frees a fluorine
Fluorine is a chemical element with the symbol F and atomic number 9. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. As the most electronegative reactive element, it is extremely reactiv ...
atom, leaving uranium pentafluoride which then precipitates out of the gas. Cascading the MLIS stages is more difficult than with other methods because the UF5 must be fluorinated back to UF6 before being introduced into the next MLIS stage. Alternative MLIS schemes are currently being developed (using a first laser in the near-infrared or visible region) where an enrichment of over 95% can be obtained in a single stage, but the methods have not (yet) reached industrial feasibility. This method is called OP-IRMPD (Overtone Pre-excitation— IR Multiple Photon Dissociation).
Finally, the ' Separation of isotopes by laser excitation' (SILEX) process, developed by Silex Systems
Silex is any of various forms of ground stone. In modern contexts the word refers to a finely ground, nearly pure form of silica or silicate.
In the late 16th century, it meant powdered or ground up "flints" (i.e. stones, generally meaning the cl ...
in Australia, has been licensed to General Electric for the development of a pilot enrichment plant. The method uses uranium hexafluoride as a feedstock, and uses magnets to separate the isotopes after one isotope is preferentially ionized. Further details of the process are not disclosed.
Quite recently yet another scheme has been proposed for the deuterium
Deuterium (or hydrogen-2, symbol or deuterium, also known as heavy hydrogen) is one of two stable isotopes of hydrogen (the other being protium, or hydrogen-1). The nucleus of a deuterium atom, called a deuteron, contains one proton and one ...
separation using Trojan wavepackets in circularly polarized electromagnetic field. The process of Trojan wave packet formation by the adiabatic-rapid passage depends in ultra-sensitive way on the reduced electron and nucleus mass which with the same field frequency further leads to excitation of Trojan or anti-Trojan wavepacket depending on the kind of the isotope. Those and their giant, rotating electric dipole moment
The electric dipole moment is a measure of the separation of positive and negative electrical charges within a system, that is, a measure of the system's overall polarity. The SI unit for electric dipole moment is the coulomb- meter (C⋅m). ...
s are then -shifted in phase and the beam of such atoms splits in the gradient of the electric field in the analogy to Stern–Gerlach experiment
The Stern–Gerlach experiment demonstrated that the spatial orientation of angular momentum is quantized. Thus an atomic-scale system was shown to have intrinsically quantum properties. In the original experiment, silver atoms were sent throug ...
.
Chemical methods
Although isotopes of a single element are normally described as having the same chemical properties, this is not strictly true. In particular,reaction rate
The reaction rate or rate of reaction is the speed at which a chemical reaction takes place, defined as proportional to the increase in the concentration of a product per unit time and to the decrease in the concentration of a reactant per uni ...
s are very slightly affected by atomic mass.
Techniques using this are most effective for light atoms such as hydrogen. Lighter isotopes tend to react or evaporate
Evaporation is a type of vaporization that occurs on the surface of a liquid as it changes into the gas phase. High concentration of the evaporating substance in the surrounding gas significantly slows down evaporation, such as when humi ...
more quickly than heavy isotopes, allowing them to be separated. This is how heavy water is produced commercially, see Girdler sulfide process for details. Lighter isotopes also disassociate more rapidly under an electric field. This process in a large cascade
Cascade, Cascades or Cascading may refer to:
Science and technology Science
* Cascade waterfalls, or series of waterfalls
* Cascade, the CRISPR-associated complex for antiviral defense (a protein complex)
* Cascade (grape), a type of fruit
* Bioc ...
was used at the heavy water production plant at Rjukan
Rjukan () is a town and the administrative centre of Tinn municipality in Telemark, Norway. It is situated in Vestfjorddalen, between Møsvatn and Lake Tinn, and got its name after Rjukan Falls west of the town. The Tinn municipality council gra ...
.
One candidate for the largest kinetic isotopic effect ever measured at room temperature, 305, may eventually be used for the separation of tritium
Tritium ( or , ) or hydrogen-3 (symbol T or H) is a rare and radioactive isotope of hydrogen with half-life about 12 years. The nucleus of tritium (t, sometimes called a ''triton'') contains one proton and two neutrons, whereas the nucleus of ...
(T). The effects for the oxidation of tritiated formate
Formate (IUPAC name: methanoate) is the conjugate base of formic acid. Formate is an anion () or its derivatives such as ester of formic acid. The salts and esters are generally colorless.Werner Reutemann and Heinz Kieczka "Formic Acid" in ''Ull ...
anions to HTO were measured as:
:
Gravity
Isotopes of carbon, oxygen, and nitrogen can be purified by chilling these gases or compounds nearly to their liquefaction temperature in very tall () columns. The heavier isotopes sink and the lighter isotopes rise, where they are easily collected. The process was developed in the late 1960s by scientists at Los Alamos National Laboratory. This process is also called " cryogenic distillation".The SWU (separative work unit)
Separative Work Unit (SWU) is a complex unit which is a function of the amount of uranium processed and the degree to which it is enriched, ''i.e.'' the extent of increase in the concentration of the U-235 isotope relative to the remainder. The unit is strictly: Kilogram Separative Work Unit, and it measures the quantity of separative work (indicative of energy used in enrichment) when feed and product quantities are expressed in kilograms. The effort expended in separating a mass ''F'' of feed of assay ''xf'' into a mass ''P'' of product assay xp and waste of mass ''W'' and assay ''xw'' is expressed in terms of the number of separative work units needed, given by the expression SWU = ''WV''(''xw'') + ''PV''(''xp'') - ''FV''(''xf''), where ''V''(''x'') is the "value function," defined as ''V''(''x'') = (1 - 2''x'') ln ((1 - ''x'') /''x''). Separative work is expressed in SWUs, kg SW, or kg UTA (from the German ''Urantrennarbeit'' ) * 1 SWU = 1 kg SW = 1 kg UTA * 1 kSWU = 1.0 t SW = 1 t UTA * 1 MSWU = 1 kt SW = 1 kt UTA If, for example, you begin with 100 kilograms (220 pounds) of natural uranium, it takes about 60 SWU to produce 10 kilograms (22 pounds) of uranium enriched in U-235 content to 4.5%.Isotope separators for research
Radioactive beams of specific isotopes are widely used in the fields of experimental physics, biology and materials science. The production and formation of these radioactive atoms into an ionic beam for study is an entire field of research carried out at many laboratories throughout the world. The first isotope separator was developed at the Copenhagen Cyclotron by Bohr and coworkers using the principle of electromagnetic separation. Today, there are many laboratories around the world that supply beams of radioactive ions for use. Arguably the principal Isotope Separator On Line (ISOL) is ISOLDE at CERN, which is a joint European facility spread across the Franco-Swiss border near the city of Geneva. This laboratory uses mainly proton spallation of uranium carbide targets to produce a wide range of radioactive fission fragments that are not found naturally on earth. During spallation (bombardment with high energy protons), a uranium carbide target is heated to several thousand degrees so that radioactive atoms produced in the nuclear reaction are released. Once out of the target, the vapour of radioactive atoms travels to an ionizer cavity. This ionizer cavity is a thin tube made of a refractory metal with a highwork function
In solid-state physics, the work function (sometimes spelt workfunction) is the minimum thermodynamic work (i.e., energy) needed to remove an electron from a solid to a point in the vacuum immediately outside the solid surface. Here "immediately ...
allowing for collisions with the walls to liberate a single electron from a free atom (surface ionization
Thermal ionization, also known as surface ionization or contact ionization, is a physical process whereby the atoms are desorbed from a hot surface, and in the process are ionized.
Thermal ionization is used to make simple ion sources, for mass ...
effect). Once ionized, the radioactive species are accelerated by an electrostatic field and injected into an electromagnetic separator. As ions entering the separator are of approximately equal energy, those ions with a smaller mass will be deflected by the magnetic field by a greater amount than those with a heavier mass. This differing radius of curvature allows for isobaric purification to take place. Once purified isobarically, the ion beam is then sent to the individual experiments. In order to increase the purity of the isobaric beam, laser ionization can take place inside the ionizer cavity to selectively ionize a single element chain of interest. At CERN, this device is called the Resonance Ionization Laser Ion Source (RILIS). Currently over 60% of all experiments opt to use the RILIS to increase the purity of radioactive beams.
Beam production capability of ISOL facilities
As the production of radioactive atoms by the ISOL technique depends on the free atom chemistry of the element to be studied, there are certain beams which cannot be produced by simple proton bombardment of thick actinide targets.Refractory
In materials science, a refractory material or refractory is a material that is resistant to decomposition by heat, pressure, or chemical attack, and retains strength and form at high temperatures. Refractories are polycrystalline, polyphase, ...
metals such as tungsten and rhenium do not emerge from the target even at high temperatures due to their low vapour pressure. In order to produce these types of beams, a thin target is required. The Ion Guide Isotope Separator On Line (IGISOL) technique was developed in 1981 at the University of Jyväskylä cyclotron
A cyclotron is a type of particle accelerator invented by Ernest O. Lawrence in 1929–1930 at the University of California, Berkeley, and patented in 1932. Lawrence, Ernest O. ''Method and apparatus for the acceleration of ions'', filed: Jan ...
laboratory in Finland
Finland ( fi, Suomi ; sv, Finland ), officially the Republic of Finland (; ), is a Nordic country in Northern Europe. It shares land borders with Sweden to the northwest, Norway to the north, and Russia to the east, with the Gulf of Bot ...
. In this technique, a thin uranium target is bombarded with protons and nuclear reaction products recoil out of the target in a charged state. The recoils are stopped in a gas cell and then exit through a small hole in the side of the cell where they are accelerated electrostatically and injected into a mass separator. This method of production and extraction takes place on a shorter timescale compared to the standard ISOL technique and isotopes with short half-lives (sub millisecond) can be studied using an IGISOL. An IGISOL has also been combined with a laser ion source at the Leuven Isotope Separator On Line (LISOL) in Belgium. Thin target sources generally provide significantly lower quantities of radioactive ions than thick target sources and this is their main drawback.
As experimental nuclear physics progresses, it is becoming more and more important to study the most exotic of radioactive nuclei. In order to do so, more inventive techniques are required to create nuclei with extreme proton/neutron ratios. An alternative to the ISOL techniques described here is that of fragmentation beams, where the radioactive ions are produced by fragmentation reactions on a fast beam of stable ions impinging on a thin target (usually of beryllium atoms). This technique is used, for example, at the National Superconducting Cyclotron Laboratory (NSCL) at Michigan State University and at the Radioactive Isotope Beam Factory (RIBF) at RIKEN, in Japan.
References
{{ReflistExternal links
Utilization of kinetic isotope effects for the concentration of tritium
GM Brown, TJ Meyer et al., 2001.
from the
World Nuclear Association
World Nuclear Association is the international organization that promotes nuclear power and supports the companies that comprise the global nuclear industry. Its members come from all parts of the nuclear fuel cycle, including uranium mining, u ...
Annotated bibliography on electromagnetic separation of uranium isotopes form the Alsos Digital Library
German inventions