|
Adiabatic Accessibility
Adiabatic accessibility denotes a certain relation between two equilibrium states of a thermodynamic system (or of different such systems). The concept was coined by Constantin Carathéodory in 1909 ("adiabatische Erreichbarkeit") and taken up 90 years later by Elliott Lieb and J. Yngvason in their axiomatic approach to the foundations of thermodynamics. It was also used by R. Giles in his 1964 monograph.Robin Giles: "Mathematical Foundations of Thermodynamics", Pergamon, Oxford 1964 Description A system in a state ''Y'' is said to be adiabatically accessible from a state ''X'' if ''X'' can be transformed into ''Y'' without the system suffering transfer of energy as heat or transfer of matter. ''X'' may, however, be transformed to ''Y'' by doing work on ''X''. For example, a system consisting of one kilogram of warm water is adiabatically accessible from a system consisting of one kilogram of cool water, since the cool water may be mechanically stirred to warm it. However, the c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Equilibrium State
Thermodynamic equilibrium is an axiomatic concept of thermodynamics. It is an internal state of a single thermodynamic system, or a relation between several thermodynamic systems connected by more or less permeable or impermeable walls. In thermodynamic equilibrium, there are no net macroscopic flows of matter nor of energy within a system or between systems. In a system that is in its own state of internal thermodynamic equilibrium, no macroscopic change occurs. Systems in mutual thermodynamic equilibrium are simultaneously in mutual thermal, mechanical, chemical, and radiative equilibria. Systems can be in one kind of mutual equilibrium, while not in others. In thermodynamic equilibrium, all kinds of equilibrium hold at once and indefinitely, until disturbed by a thermodynamic operation. In a macroscopic equilibrium, perfectly or almost perfectly balanced microscopic exchanges occur; this is the physical explanation of the notion of macroscopic equilibrium. A thermodynamic sys ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thermodynamic System
A thermodynamic system is a body of matter and/or radiation, confined in space by walls, with defined permeabilities, which separate it from its surroundings. The surroundings may include other thermodynamic systems, or physical systems that are not thermodynamic systems. A wall of a thermodynamic system may be purely notional, when it is described as being 'permeable' to all matter, all radiation, and all forces. A state of a thermodynamic system can be fully described in several different ways, by several different sets of thermodynamic state variables. A widely used distinction is between ''isolated'', ''closed'', and ''open'' thermodynamic systems. An isolated thermodynamic system has walls that are non-conductive of heat and perfectly reflective of all radiation, that are rigid and immovable, and that are impermeable to all forms of matter and all forces. (Some writers use the word 'closed' when here the word 'isolated' is being used.) A closed thermodynamic system is c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Constantin Carathéodory
Constantin Carathéodory ( el, Κωνσταντίνος Καραθεοδωρή, Konstantinos Karatheodori; 13 September 1873 – 2 February 1950) was a Greek mathematician who spent most of his professional career in Germany. He made significant contributions to real and complex analysis, the calculus of variations, and measure theory. He also created an axiomatic formulation of thermodynamics. Carathéodory is considered one of the greatest mathematicians of his era and the most renowned Greek mathematician since antiquity. Origins Constantin Carathéodory was born in 1873 in Berlin to Greek parents and grew up in Brussels. His father Stephanos, a lawyer, served as the Ottoman ambassador to Belgium, St. Petersburg and Berlin. His mother, Despina, née Petrokokkinos, was from the island of Chios. The Carathéodory family, originally from Bosnochori or Vyssa, was well established and respected in Constantinople, and its members held many important governmental positions. Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mathematische Annalen
''Mathematische Annalen'' (abbreviated as ''Math. Ann.'' or, formerly, ''Math. Annal.'') is a German mathematical research journal founded in 1868 by Alfred Clebsch and Carl Neumann. Subsequent managing editors were Felix Klein, David Hilbert, Otto Blumenthal, Erich Hecke, Heinrich Behnke, Hans Grauert, Heinz Bauer, Herbert Amann, Jean-Pierre Bourguignon, Wolfgang Lück, and Nigel Hitchin. Currently, the managing editor of Mathematische Annalen is Thomas Schick. Volumes 1–80 (1869–1919) were published by Teubner. Since 1920 (vol. 81), the journal has been published by Springer. In the late 1920s, under the editorship of Hilbert, the journal became embroiled in controversy over the participation of L. E. J. Brouwer on its editorial board, a spillover from the foundational Brouwer–Hilbert controversy. Between 1945 and 1947 the journal briefly ceased publication. References External links''Mathematische Annalen''homepage at Springer''Mathematische Annalen''archive (1869� ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Jakob Yngvason
Jakob Yngvason (born 23 November 1945) is an Icelandic/Austrian physicist and emeritus professor of mathematical physics at the University of Vienna. He has made important contributions to local quantum field theory, thermodynamics, and the quantum theory of many-body systems, in particular cold atomic gases and Bose–Einstein condensation. He is co-author, together with Elliott H. Lieb, Jan Philip Solovej and Robert Seiringer, of a monograph on Bose gases. Career After graduating from high school in 1964 in Reykjavík, Yngvason studied physics at Göttingen University, obtaining his Diploma in physics in 1969, and a doctorate (''dr.rer.nat'') in 1973. His thesis advisor was Hans-Jürgen Borchers. Yngvason was assistant professor at the University of Göttingen, 1973–1978, 1978–1985 research scientist at the Science Institute of the University of Iceland, and during 1985–1996 professor of theoretical physics at the University of Iceland. In 1996, he became professor of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quasistatic Processes
In thermodynamics, a quasi-static process (also known as a quasi-equilibrium process; from the Latin ''quasi'', meaning ‘as if’), is a thermodynamic process that happens slowly enough for the system to remain in internal physical (but not necessarily chemical) thermodynamic equilibrium. An example of this is quasi-static expansion of a mixture of hydrogen and oxygen gas, where the volume of the system changes so slowly that the pressure remains uniform throughout the system at each instant of time during the process. Such an idealized process is a succession of physical equilibrium states, characterized by infinite slowness.Rajput, R.K. (2010). ''A Textbook of Engineering Thermodynamics'', 4th edition, Laxmi Publications (P) Ltd, New Delhi, pages 21, 45, 58. Only in a quasi-static thermodynamic process can we exactly define intensive quantities (such as pressure, temperature, specific volume, specific entropy) of the system at every instant during the whole process; otherwise ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
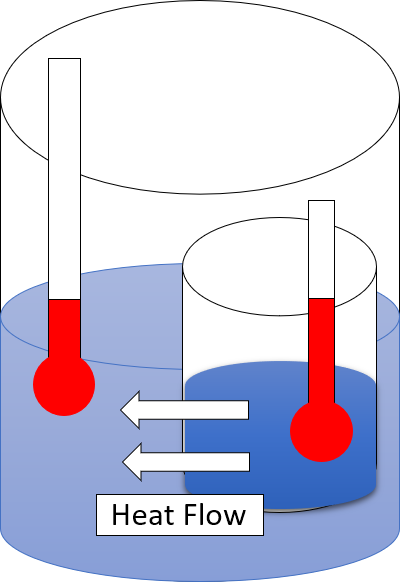
Second Law Of Thermodynamics
The second law of thermodynamics is a physical law based on universal experience concerning heat and Energy transformation, energy interconversions. One simple statement of the law is that heat always moves from hotter objects to colder objects (or "downhill"), unless energy in some form is supplied to reverse the direction of heat flow. Another definition is: "Not all heat energy can be converted into Work (thermodynamics), work in a cyclic process."Young, H. D; Freedman, R. A. (2004). ''University Physics'', 11th edition. Pearson. p. 764. The second law of thermodynamics in other versions establishes the concept of entropy as a physical property of a thermodynamic system. It can be used to predict whether processes are forbidden despite obeying the requirement of conservation of energy as expressed in the first law of thermodynamics and provides necessary criteria for spontaneous processes. The second law may be formulated by the observation that the entropy of isolated systems ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Entropy
Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. The term and the concept are used in diverse fields, from classical thermodynamics, where it was first recognized, to the microscopic description of nature in statistical physics, and to the principles of information theory. It has found far-ranging applications in chemistry and physics, in biological systems and their relation to life, in cosmology, economics, sociology, weather science, climate change, and information systems including the transmission of information in telecommunication. The thermodynamic concept was referred to by Scottish scientist and engineer William Rankine in 1850 with the names ''thermodynamic function'' and ''heat-potential''. In 1865, German physicist Rudolf Clausius, one of the leading founders of the field of thermodynamics, defined it as the quotient of an infinitesimal amount of hea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Equilibrium Chemistry
Equilibrium chemistry is concerned with systems in chemical equilibrium. The unifying principle is that the free energy of a system at equilibrium is the minimum possible, so that the slope of the free energy with respect to the reaction coordinate is zero. This principle, applied to mixtures at equilibrium provides a definition of an equilibrium constant. Applications include acid–base, host–guest, metal–complex, solubility, partition, chromatography and redox equilibria. Thermodynamic equilibrium A chemical system is said to be in equilibrium when the quantities of the chemical entities involved do not and ''cannot'' change in time without the application of an external influence. In this sense a system in chemical equilibrium is in a stable state. The system at chemical equilibrium will be at a constant temperature, pressure or volume and a composition. It will be insulated from exchange of heat with the surroundings, that is, it is a closed system. A change of te ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thermodynamic Cycles
A thermodynamic cycle consists of a linked sequence of thermodynamic processes that involve transfer of heat and work into and out of the system, while varying pressure, temperature, and other state variables within the system, and that eventually returns the system to its initial state. In the process of passing through a cycle, the working fluid (system) may convert heat from a warm source into useful work, and dispose of the remaining heat to a cold sink, thereby acting as a heat engine. Conversely, the cycle may be reversed and use work to move heat from a cold source and transfer it to a warm sink thereby acting as a heat pump. If at every point in the cycle the system is in thermodynamic equilibrium, the cycle is reversible. Whether carried out reversible or irreversibly, the net entropy change of the system is zero, as entropy is a state function. During a closed cycle, the system returns to its original thermodynamic state of temperature and pressure. Process quantities ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thermodynamic Processes
Classical thermodynamics considers three main kinds of thermodynamic process: (1) changes in a system, (2) cycles in a system, and (3) flow processes. (1)A Thermodynamic process is a process in which the thermodynamic state of a system is changed. A change in a system is defined by a passage from an initial to a final state of thermodynamic equilibrium. In classical thermodynamics, the actual course of the process is not the primary concern, and often is ignored. A state of thermodynamic equilibrium endures unchangingly unless it is interrupted by a thermodynamic operation that initiates a thermodynamic process. The equilibrium states are each respectively fully specified by a suitable set of thermodynamic state variables, that depend only on the current state of the system, not on the path taken by the processes that produce the state. In general, during the actual course of a thermodynamic process, the system may pass through physical states which are not describable as thermody ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |