potassium peroxide on:
[Wikipedia]
[Google]
[Amazon]
Potassium peroxide is an 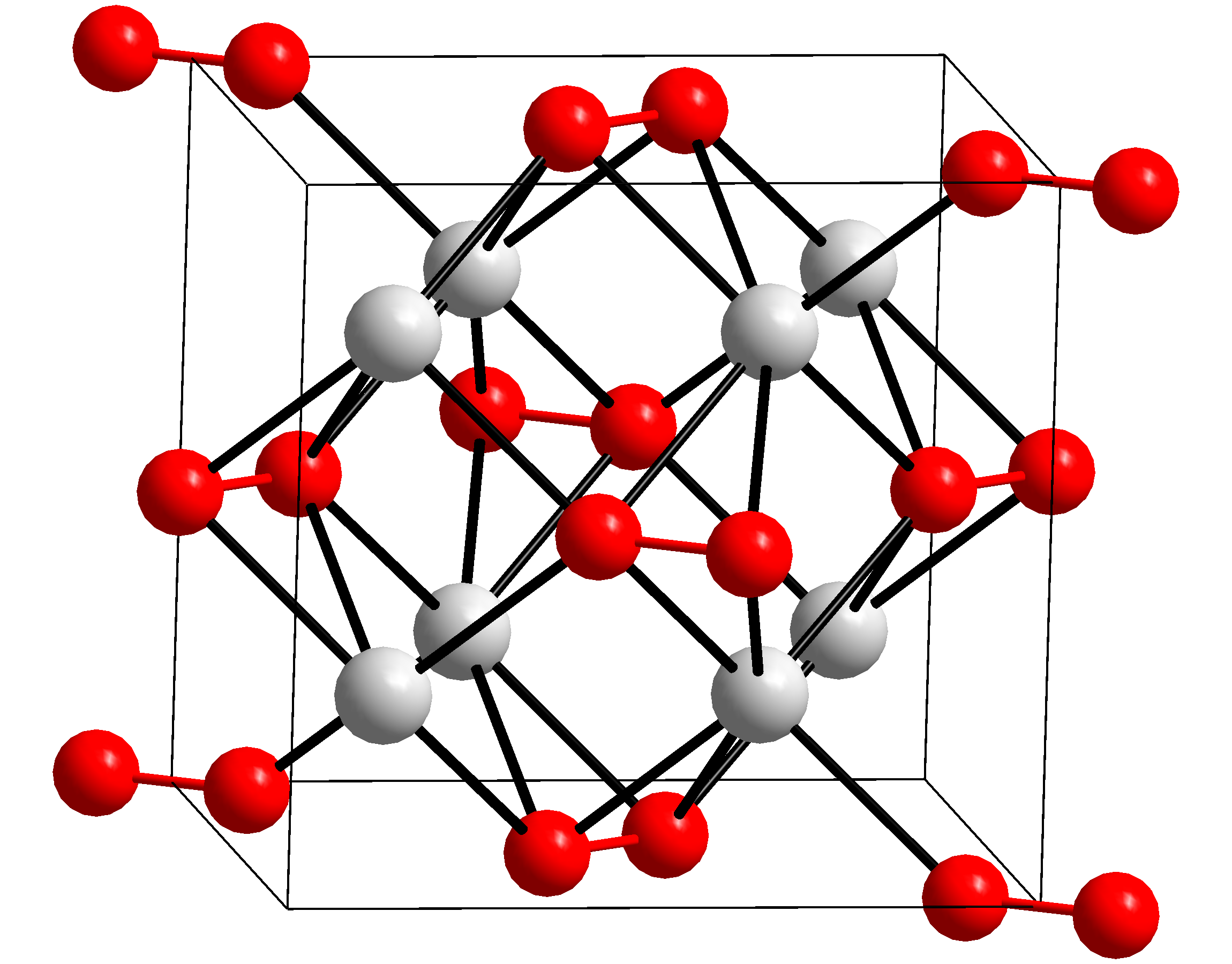 Potassium peroxide reacts with water to form potassium hydroxide and oxygen:
:
Potassium peroxide reacts with water to form potassium hydroxide and oxygen:
: 2K2O2 + 2H2O -> 4KOH + O2 (^)
inorganic compound
In chemistry, an inorganic compound is typically a chemical compound that lacks carbon–hydrogen bonds, that is, a compound that is not an organic compound. The study of inorganic compounds is a subfield of chemistry known as ''inorganic chemistr ...
with the molecular formula K2O2. It is formed as potassium reacts with oxygen in the air, along with potassium oxide
Potassium oxide ( K O) is an ionic compound of potassium and oxygen. It is a base. This pale yellow solid is the simplest oxide of potassium. It is a highly reactive compound that is rarely encountered. Some industrial materials, such as fertili ...
(K2O) and potassium superoxide
Potassium superoxide is an inorganic compound with the formula KO2. It is a yellow paramagnetic solid that decomposes in moist air. It is a rare example of a stable salt of the superoxide anion. It is used as a scrubber, dehumidifier, and gen ...
(KO2).
 Potassium peroxide reacts with water to form potassium hydroxide and oxygen:
:
Potassium peroxide reacts with water to form potassium hydroxide and oxygen:
: Properties
Potassiumperoxide
In chemistry, peroxides are a group of compounds with the structure , where R = any element. The group in a peroxide is called the peroxide group or peroxo group. The nomenclature is somewhat variable.
The most common peroxide is hydrogen p ...
is a highly reactive, oxidizing white to yellowish solid which, while not flammable itself, reacts violently with flammable materials. It decomposes violently on contact with water.
The standard enthalpy of formation of potassium peroxide is ΔH f 0 = −496 kJ/mol.
Usage
Potassium Peroxide is used as an oxidizing agent and bleach (due to theperoxide
In chemistry, peroxides are a group of compounds with the structure , where R = any element. The group in a peroxide is called the peroxide group or peroxo group. The nomenclature is somewhat variable.
The most common peroxide is hydrogen p ...
), and to purify air. References
Peroxides Potassium compounds Oxidizing agents {{inorganic-compound-stub